Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech financings
Spanish biotech Ona announced a $86.6 million Series B round, ranking among the largest venture financings in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funds will expand Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical pipeline.

FDA Clears Genentech’s Tecentriq for ctDNA-Guided Adjuvant Bladder Cancer Therapy
Genentech announced FDA approval of Tecentriq and Tecentriq Hybreza as adjuvant therapy for muscle‑invasive bladder cancer patients with ctDNA‑detected minimal residual disease. The Phase III IMvigor011 trial showed a 36% drop in disease recurrence or death and a 41% reduction in overall mortality, marking the first ctDNA‑guided oncology approval in the United States.

Inside the Expert Network Guiding TGN-S15 to Market
Toragen’s CEO Sandra Coufal detailed the formation of a Scientific Advisory Board designed to steer the HPV‑targeted therapy TGN‑S15 through development. The board is anchored by Dr. Aldo Venuti, a leading authority on the HPV E5 protein, who supplies the rare W12...

Bayer's Asundexian Receives FDA Priority Review for Secondary Stroke Prevention
Bayer AG announced that the U.S. FDA has granted priority review to its investigational oral Factor XIa inhibitor Asundexian for secondary stroke prevention. The decision follows positive Phase 3 OCEANIC‑STROKE data that met both efficacy and safety endpoints, positioning the...
BioMarin Drug Acquired in Buyout Misses Goal in Rare Disease Study
BioMarin Pharmaceutical’s BMN 401, an enzyme‑replacement therapy for ENPP1 deficiency, met its primary biochemical endpoint by raising plasma PPi levels but failed the co‑primary skeletal health goal in a Phase 3 trial of 27 children. The miss on clinical benefit and all...

Lessons Learned From Drug Development Programs in Autism: Implications for Future Programs
The article synthesizes expert insights on why autism drug development has lagged, highlighting the lack of approved therapies for core symptoms and the challenges posed by biological and clinical heterogeneity. It outlines six key learnings, including the need to measure...

Wave Aims for Monthly Dosing with RNA Editing Treatment for AATD
Wave Life Sciences announced an updated read‑out from its early‑stage trial of an RNA‑editing therapy for alpha‑1 antitrypsin deficiency (AATD). The data indicate that the treatment can restore functional protein levels with a dosing schedule that could be moved to...

Relay Doubles the Bar, Outpacing Novartis with a 60% Response in Rare Disease
Relay’s oral PI3Kα inhibitor zovegalisib posted a 60% volumetric response in a Phase 2 trial of patients with vascular malformations, far outpacing Novartis’ 11% result with Vijoice. The data, presented at the ISVAA World Congress 2026, came from 20 evaluable patients,...

STAT+: A New Ranking of Pharma Companies by R&D Performance
Eli Lilly topped IDEA Pharma’s annual R&D ranking, ranking first in both the Innovation and Invention categories—a first in the study’s history. The Innovation score reflects revenue from newly launched drugs, while the Invention score weighs the size of the pipeline...

EMA Marketing Authorization of New Drugs in April 2026
In April 2026 the European Medicines Agency granted marketing authorizations and CHMP positive opinions for a slate of new therapies spanning infectious disease, neurology, immunology, endocrinology and rare disorders. Merck’s Enflonsia monoclonal antibody reduced RSV‑related hospitalizations by 84% in infants,...
FDA Approves Eli Lilly’s Foundayo, Pitting First Oral Obesity Pill Against Novo Nordisk’s Wegovy
Eli Lilly’s oral obesity medication Foundayo received FDA approval on April 1, becoming the first pill of its kind and setting up an immediate head‑to‑head battle with Novo Nordisk’s oral Wegovy. The two drugs differ in efficacy, manufacturing ease and safety profile,...

Accro Bioscience Secures $50 Million Series C Led by OrbiMed to Advance Ulcerative Colitis Drug
Accro Bioscience announced a $50 million Series C round headed by OrbiMed, with participation from TCGX, LAV, Cenova Capital and existing backers. The capital will finance a Phase IIb study of AC-101, a RIPK2 inhibitor targeting ulcerative colitis, underscoring private‑equity interest...

Sourav K. Mishra, M.D. / All India Institute of Medical Sciences - 724881 - 04/29/2026
The FDA issued a warning letter to Dr. Sourav K. Mishra of AIIMS Bhubaneswar for serious violations in a bioequivalence study of doxorubicin hydrochloride liposome injection conducted for Qilu Pharmaceutical. The investigator administered the prohibited CYP3A4 inhibitor aprepitant to all...
SERB Pays €115 Million for Hansa Biopharma’s Idefirix Rights in Europe and MENA
SERB Pharmaceuticals agreed to buy exclusive rights to Hansa Biopharma's kidney‑transplant drug Idefirix across Europe and the MENA region for €115 million ($124 million). The deal includes an upfront €110 million payment and a €5 million contingent payment tied to EMA approval, positioning SERB...
Sanofi's Efdoralprin Alfa Shows Triple Alpha‑1 Levels in Phase 2 AATD Emphysema Trial
Sanofi announced that its experimental drug efdoralprin alfa met the primary endpoint of its Phase 2 ElevAATe trial, delivering mean alpha‑1 antitrypsin trough levels more than three times higher than weekly plasma‑derived augmentation therapy. The result strengthens Sanofi’s rare‑disease portfolio and...

RAS Inhibition Enters Its Second Wave
RAS inhibition has moved into a second wave of drug development that goes beyond the KRAS G12C breakthrough. After sotorasib and adagrasib secured accelerated approvals for non‑small cell lung cancer and later for KRAS G12C‑mutated colorectal cancer, companies are targeting more prevalent...

Vincentage Pharma Reports Topline P-III Trial Data on VCT220 for Weight Management
Vincentage Pharma announced topline results from its pivotal Phase III trial of VCT220, a GLP‑1 agonist, in 840 Chinese adults with obesity or overweight plus a comorbidity. Over 52 weeks, the 120 mg and 160 mg doses produced mean weight losses of 12.2%...
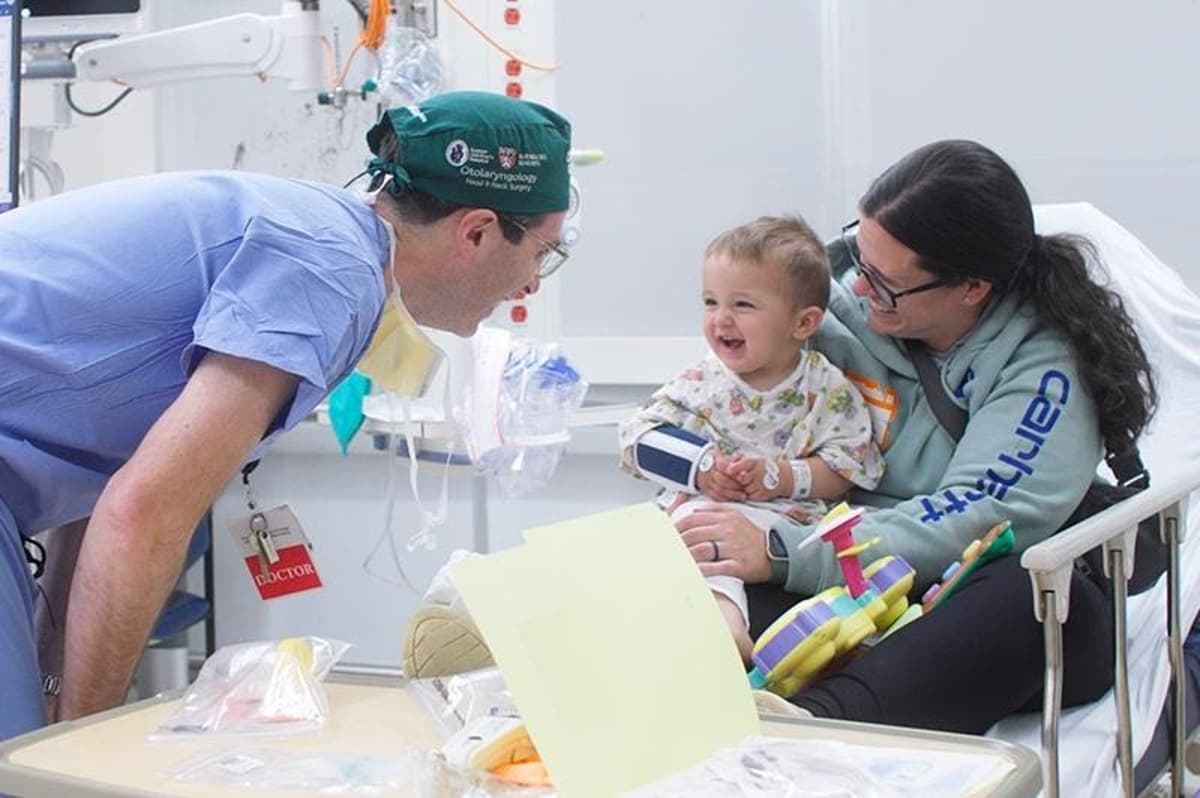
FDA Approves First-Ever Gene Therapy For Deafness, Opening Door To New Era
The U.S. Food and Drug Administration has approved the world’s first gene therapy for deafness, targeting the OTOF mutation that blocks sound transmission in the inner ear. In a pivotal trial of 20 children and teens, 80% experienced improved hearing...

In the Clinic for May 19, 2026
BioWorld’s May 19, 2026 clinic roundup aggregates a wide array of industry snapshots, special reports, and infographics covering biopharma, med‑tech, and emerging health topics. The collection highlights trends such as dynamic digital data analysis, the 2026 med‑tech outlook, mRNA vaccine research, and...

BioMarin Suffers Another Blow to Rare Disease Portfolio in Phase 3 Flop
BioMarin’s investigational enzyme replacement therapy BMN 401 lowered plasma inorganic pyrophosphate (PPi) in the Phase 3 ENERGY 3 trial for ENPP1 deficiency, but it did not translate into clinical benefit. The study enrolled almost 30 children aged 1‑12 and missed the primary Radiographic...

Bayer’s Hyrnuo (Sevabertinib) Gains the US FDA Priority Review for 1L Treatment of HER2-Mutated NSCLC
Bayer’s Hyrnuo (sevabertinib) received FDA priority review for first‑line treatment of adults with locally advanced or metastatic non‑small cell lung cancer (NSCLC) harboring HER2 kinase‑domain activating mutations and no prior systemic therapy. The review follows promising interim data from Cohort F...

What It Takes to Run Rare Pediatric Disease Trials Across Borders: Insights From Polaryx Therapeutics
Polaryx Therapeutics is gearing up to test its oral candidate PLX-200 in a Phase II SOTERIA basket trial for four ultra‑rare lysosomal storage disorders—CLN2, CLN3, Krabbe and Sandhoff. The FDA has granted Fast Track designation for all four indications, promising closer...

Brepocitinib Approval May Be ‘Highlight of the Derm-Rheum World’
The FDA has set an Aug. 29 PDUFA date for brepocitinib, a first‑in‑class oral TYK2/JAK1 inhibitor, as a potential therapy for dermatomyositis. In the 52‑week VALOR phase‑3 trial, the drug outperformed placebo on skin and muscle outcomes and enabled significant glucocorticoid...

First Healthy Volunteers Receive TRIV-573 Doses in Triveni Bio’s Phase I Trial
Trivena Bio has dosed its first healthy volunteers in a Phase I trial of TRIV‑573, a half‑life‑extended bispecific antibody that simultaneously inhibits kallikreins 5/7 and blocks interleukin‑13. The dual‑target approach is designed to repair the skin barrier while curbing inflammation in moderate‑to‑severe...
Common Asthma Drug May Turn Off Tumor 'Switch' Tied to Immunotherapy Resistance
A Northwestern Medicine study published in Nature Cancer shows that blocking the cysteinyl leukotriene receptor 1 (CysLTR1) with the asthma drug montelukast can reverse immunotherapy resistance in several aggressive cancers. Experiments in mouse models and analyses of human tumor samples demonstrated...

Scaling Scientific R&D with AI Supercomputing Infrastructure — with Thomas Fuchs of Eli Lilly
In this episode, Thomas Fuchs, Chief AI Officer at Eli Lilly, explains how the company is building an AI‑ready supercomputing platform—based on NVIDIA’s DGX SuperPod B300 architecture—to accelerate drug discovery, development, and manufacturing. He highlights that the massive compute power lets...
Vaccine Experts Debate Options to Combat Outbreak of Unusual Ebola Strain
The World Health Organization convened a closed meeting of vaccine experts after the Bundibugyo ebolavirus outbreak in the Democratic Republic of the Congo was declared a public‑health emergency. The outbreak has produced roughly 500 suspected cases and more than 130...
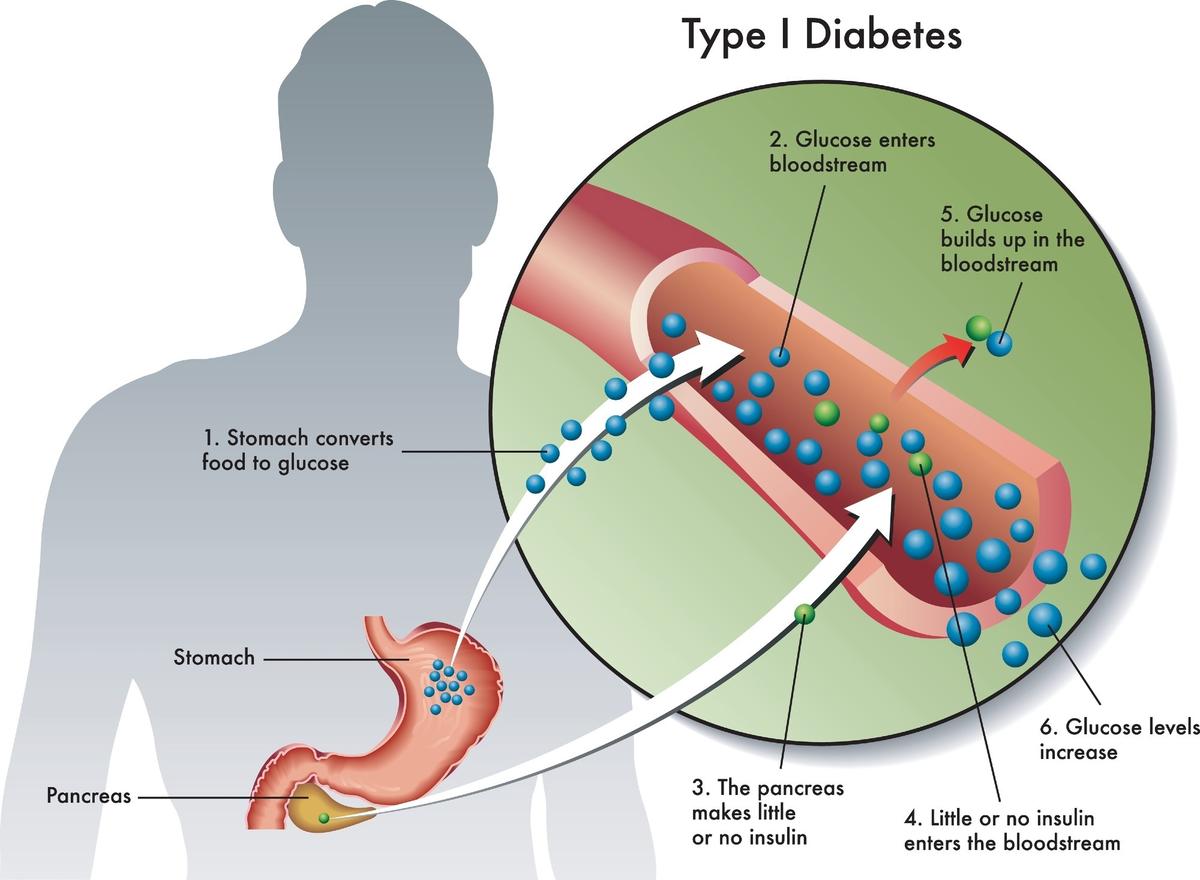
New Therapies Could Help Type 1 Diabetes Care Move Beyond Insulin Alone
A recent review in The Journal of Clinical Investigation outlines emerging disease-modifying therapies for type 1 diabetes that aim to preserve beta‑cell function alongside insulin. The anti‑CD3 antibody teplizumab showed a single 14‑day course can postpone clinical onset by up to...

VERTANICAL’s VER‑01 Gets FDA Breakthrough Therapy Designation for Chronic Low‑Back Pain
VERTANICAL announced that the U.S. Food and Drug Administration has granted Breakthrough Therapy Designation to its oral pain drug VER‑01 for chronic low‑back pain. The designation speeds development and underscores the drug’s potential to address a massive unmet need for...

Testosterone Shots: SubQ or IM?
A 2022 head‑to‑head trial of 234 hypogonadal men compared weekly intramuscular testosterone cypionate with subcutaneous testosterone enanthate. Both routes achieved equivalent total testosterone levels, but the subcutaneous group showed significantly lower post‑therapy estradiol and hematocrit. The flatter absorption curve of...

RFK Jr. Cites Danish Vaccine Study to Justify $2.6 B Gavi Funding Cut
U.S. Health and Human Services Secretary Robert F. Kennedy Jr. referenced a 2017 Danish study on non‑specific vaccine effects to justify a $2.6 billion cut to Gavi support, prompting renewed scrutiny of the research and its policy impact. Critics argue the...

Neurocrine Biosciences Completes $2.9 Billion Soleno Acquisition
Neurocrine Biosciences has closed a $2.9 billion cash deal to acquire Soleno, bringing the hyperphagia drug VYKAT XR into its portfolio. The transaction was funded with a $53 per‑share tender offer and a new $1 billion revolving credit facility, positioning Neurocrine for growth...
AstraZeneca's Baxfendy Wins FDA Approval, Offering First‑In‑Class Hypertension Therapy
AstraZeneca announced FDA approval of Baxfendy (baxdrostat), the first aldosterone synthase inhibitor for hypertension. The drug cut systolic blood pressure by up to 9.8 mmHg in Phase III trials, targeting roughly 23 million U.S. patients whose blood pressure remains uncontrolled despite multiple medicines.

What’s Next for FDA
The Biden administration swiftly removed most of the ideological appointees installed by former FDA commissioner Marty Makary, reinstating career officials to restore institutional norms. HHS advisor Chris Klomp led the overnight purge, signaling a shift from political loyalty to administrative competence....
Immune Checkpoint Regulation in Cancer Therapy and Evasion
The article reviews the multilayered regulation of immune checkpoint molecules, detailing genetic, epigenetic, transcriptional, post‑transcriptional, translational, and post‑translational mechanisms that shape checkpoint expression in tumors and immune cells. It highlights the recent clinical integration of LAG3‑targeted therapies as the newest...

MHRA’s Pound on Aligning Clinical Trial Regulation with Innovation
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) is rolling out clinical‑trial reforms aimed at more than just faster approvals. Interim director of innovation James Pound says the agency is pursuing international alignment and horizon‑scanning to anticipate emerging therapies....
New Math Method Inflates Alzheimer’s Drug Success by 29x
A Brown University research letter published in JAMA Neurology warns that the statistical technique known as quantile aggregation can dramatically overstate the efficacy of emerging Alzheimer’s drugs. Simulations showed the method inflates the perceived link between amyloid clearance and cognitive...

SCOTUS Rejects Pharma Companies’ Petitions To Review IRA Drug Program
The U.S. Supreme Court on May 18 denied six pharmaceutical companies’ petitions to revisit lower‑court rulings that upheld the Inflation Reduction Act’s (IRA) Medicare drug‑price negotiation program. The decision ends the companies’ multi‑year legal challenge, leaving the federal government’s authority...
SciBase Shows Nevisense Detects Age‑Related Skin Barrier Changes with 0.69 Correlation
SciBase Holding AB announced that its Nevisense electrical impedance spectroscopy platform can identify age‑related skin barrier alterations, delivering a composite score that correlates with age at a Spearman rho of 0.69. The findings were unveiled at the 2026 Society for...
Outlook Therapeutics Narrows Q2 Loss, Awaits FDA Decision on LYTENAVA in May 2026
Outlook Therapeutics announced a second‑quarter net loss of $4.45 million, a dramatic improvement from $46.35 million a year earlier. The biotech also signaled that the FDA will decide on its lead ophthalmic drug, LYTENAVA, in May 2026, a milestone that could reshape...

EU Approves Pfizer’s HYMPAVZI Gene Therapy for Hemophilia A/B with Inhibitors
The European Commission granted marketing authorization for Pfizer’s HYMPAVZI (marstacimab) to treat adults and adolescents with hemophilia A or B who have developed inhibitors. The approval follows a Phase 3 trial that showed a 93% drop in annualized bleeding rates versus...

Coya 302 Gets Fast-Track Designation for ALS Treatment
Coya Therapeutics received FDA fast‑track designation for its experimental ALS drug Coya 302, a subcutaneous blend of low‑dose IL‑2 and CTLA‑4 immunoglobulin. The therapy is designed to boost regulatory T‑cells while suppressing inflammatory monocytes, addressing the immune imbalance that accelerates motor‑neuron...
More Than 5 Million US Adults Could Benefit From Lp(a)-Targeted Therapies
New analysis presented at ACC.26 estimates over 5.3 million U.S. adults with ASCVD and Lp(a) ≥ 70 mg/dL could qualify for emerging Lp(a)-lowering therapies. Modeling suggests a 10‑30% relative risk reduction could prevent 123,000‑368,000 recurrent cardiovascular events over five years, roughly 25,000‑74,000 per year....

Atara Bounces Back with FDA
Atara Biotherapeutics saw its shares nearly double on May 7 after the FDA outlined a path for resubmitting its cell‑therapy candidate, tabelecleucel. The agency agreed to accept the single‑arm trial data in exchange for additional long‑term follow‑up, easing the regulatory deadlock...

Kazia’s Paxalisib Shows >50% CTC Drop, Early Responses
Kazia Therapeutics ($KZIA) is a catalyst-driven setup centered on upcoming ASCO data from their Paxalisib drug development platform. The update is following earlier disclosures from their ongoing Phase 1b ABC-Pax study evaluating paxalisib in metastatic triple-negative breast cancer (TNBC) showing...

Developing a Drug To Reverse Heart Disease
Repair Biotechnologies’ lead candidate, REP-0004, is an mRNA‑based lipid nanoparticle designed to deliver a cholesterol‑degrading protein exclusively to hepatocytes. The FDA granted the drug orphan‑drug status and the company targets a Phase 1 start by mid‑2027, pending GMP manufacturing and IND‑enabling...

Design Therapeutics to Unveil RESTORE-FA Gene Therapy Data as Shares Edge Higher
Design Therapeutics (DSGN) announced a webcast on May 18, 2026, to present Phase 1/2 RESTORE-FA trial data for its DT‑216P2 gene‑therapy candidate targeting Friedreich's ataxia. The move has drawn heightened investor attention as the company seeks to prove its novel...

Merck's China-Licensed ADC Succeeds in First Global Trial
Merck ADC, licensed from China, hits mark in first big global trial https://t.co/tAvLkw6pYm @ByJonGardner $MRK $GILD $AZN
Genentech Secures FDA Approval for Tecentriq in Muscle‑Invasive Bladder Cancer
Roche’s Genentech has won U.S. FDA approval for its immunotherapy Tecentriq as an adjuvant treatment for patients with muscle‑invasive bladder cancer guided by circulating tumor DNA. The decision adds a new therapeutic line for a disease with limited options and...

PASSAGE Trial Shows Tezepelumab Benefit in Diverse Asthma Groups: Njira L. Lugogo, MD, MS
The phase 4 PASSAGE trial evaluated tezepelumab in severe, uncontrolled asthma and reported a 70% reduction in annualized asthma exacerbation rate overall, with 54‑77% reductions across all phenotypes. The study included underrepresented groups such as smokers, patients with COPD, Black individuals,...

MSD's Sac-TMT Delivers in First Phase 3 Readout
Merck (MSD) reported that its TROP2‑targeting antibody‑drug conjugate sacituzumab tirumotecan (sac‑TMT), licensed from China’s Kelun‑Biotech, improved progression‑free and overall survival in a phase 3 trial for advanced or recurrent endometrial cancer. The TroFuse‑005 readout marks the first pivotal data for sac‑TMT...