Today's Pharma Pulse

Ona lands $86.6M Series B, one of Spain’s biggest biotech financings
Spanish biotech Ona announced a $86.6 million Series B round, ranking among the largest venture financings in Spain’s life‑science sector this year. The round was led by Seventure Partners with participation from existing backers and new strategic investors. The funds will expand Ona’s AI‑driven drug discovery platform and accelerate its pre‑clinical pipeline.

Adenine Base Editing Demonstrates Profound Impact on Rare Disease
Researchers at The Jackson Laboratory used adenine base editing to repair the SCN1A R613X mutation that causes Dravet syndrome in mice. A single brain injection corrected roughly 60% of the defective DNA, restoring normal gene expression and dramatically reducing seizures. Treated mice lived significantly longer, with benefits observed both when the edit was delivered at birth and at day 12. The work showcases a potential one‑time genetic cure for a rare, fatal childhood epilepsy and aligns with the FDA’s new Plausible Mechanism Framework for rare‑disease therapies.

FDA Approves Baxdrostat for Uncontrolled Hypertension in First-in-Class Entry
The FDA has granted approval to Baxfendy (baxdrostat), AstraZeneca’s first aldosterone synthase inhibitor, for adults whose blood pressure remains uncontrolled despite existing therapies. The approval is based on the phase 3 BaxHTN trial, where a 2 mg dose lowered systolic pressure by...

Merck's ADC Sac-TMT Gets Its First Global Phase 3 Win Ahead of Schedule
Merck and China‑based Kelun‑Biotech announced that their experimental antibody‑drug conjugate sac‑TMT achieved its primary endpoint in a global Phase 3 trial, completing ahead of schedule. The study enrolled roughly 650 patients with advanced solid tumours across 12 countries and demonstrated a...

Sotatercept Reduced Morbidity in CTD-PAH Analysis: Rogerio Souza, MD, PhD
A pooled analysis of the phase‑3 STELLAR, ZENITH and HYPERION trials shows that sotatercept significantly lowers the risk of first major morbidity or mortality events in patients with connective tissue disease‑associated pulmonary arterial hypertension (CTD‑PAH). The benefit persisted despite most...

FDA Approves Baxdrostat for Inadequately Controlled Hypertension
AstraZeneca’s Baxdrostat (Baxfendy) received FDA approval as the first aldosterone synthase inhibitor for hypertension in the United States. The drug is indicated for patients whose blood pressure remains uncontrolled despite taking at least two other antihypertensive agents. Phase III trials (BaxHTN...
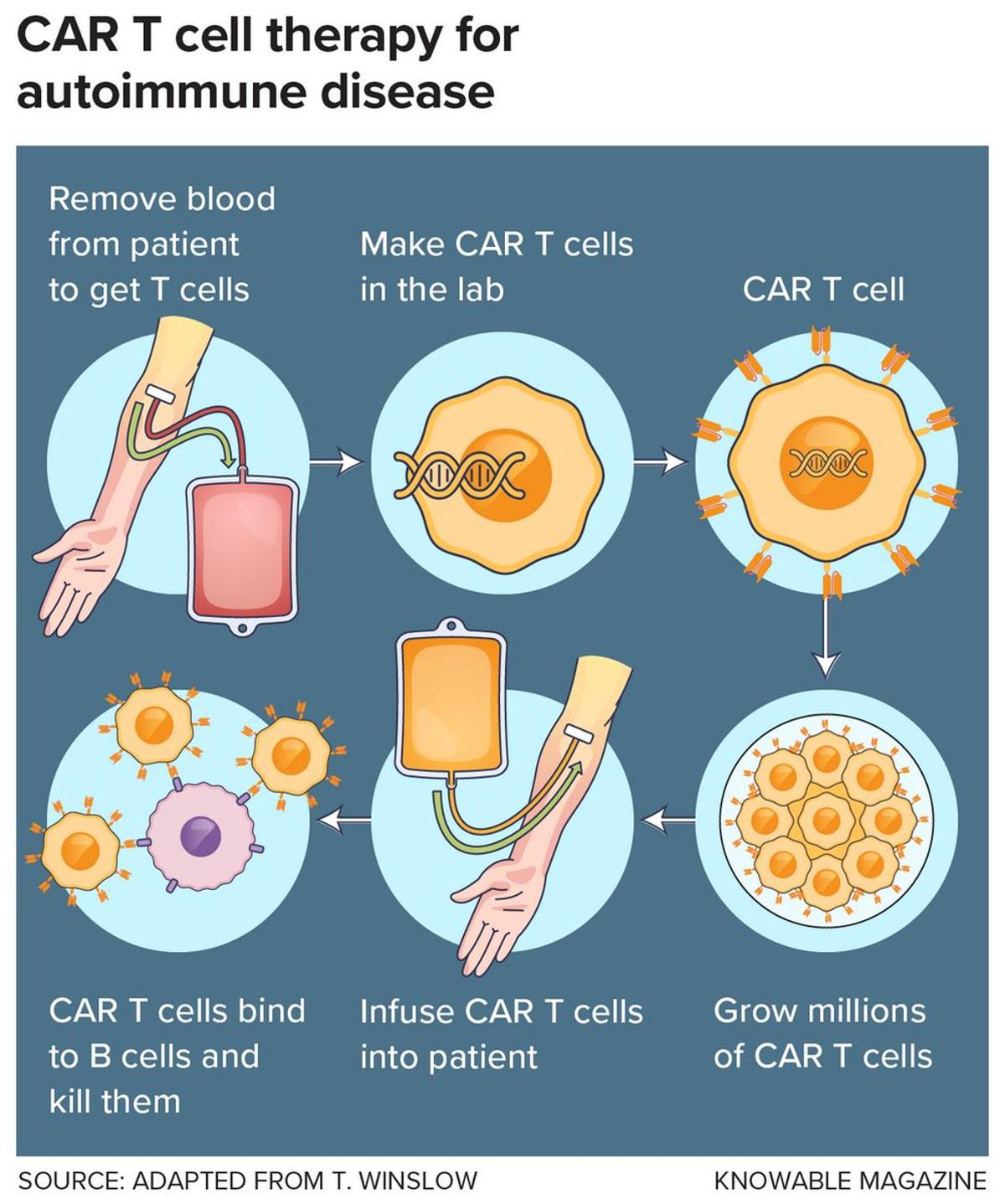
CAR‑T Therapy Shows Promise for Autoimmune Diseases
CAR T therapy, originally developed to fight blood cancers, is now being tested as a potential way to “reset” the immune system in autoimmune diseases like MS, lupus, Graves’ disease and stiff person syndrome. Early results are promising, but risks remain,...

Once‑night Pill Proves Effective for Severe Sleep Apnea
A once a night pill for severe obstructive sleep apnea effective in a Phase 3 randomized trial https://t.co/zq2Ew65WHK https://t.co/ovQEh7taeN https://t.co/N8vXzYincy

Novartis' Pluvicto Cuts PSA Progression Risk in Phase 3 Prostate Cancer Trial
Novartis announced that its radioligand therapy Pluvicto, when added to standard of care, reduced the risk of prostate-specific antigen progression in the Phase III PSMAddition trial for PSMA‑positive metastatic hormone‑sensitive prostate cancer. The data were unveiled at the American Urological Association...
Nanobiotix's Phase‑2 Lung Cancer Trial Shows 85.7% Response Rate
Nanobiotix S.A., with Johnson & Johnson as sponsor, presented Phase‑2 CONVERGE data showing an 85.7% overall response rate and 57.1% complete response in seven stage III inoperable NSCLC patients, suggesting the nanoparticle radioenhancer NBTXR3 could improve outcomes when combined with chemoradiotherapy...

Regeneron Shares Tumble 6% After Phase 3 Melanoma Trial Misses Statistical Significance
Regeneron Pharmaceuticals reported that its Phase 3 trial of fianlimab plus cemiplimab did not achieve statistical significance for progression‑free survival versus pembrolizumab, prompting a 6.23% drop in the stock during after‑hours trading. The high‑dose arm showed a numeric 5.1‑month PFS...

How Advanced Analytics Partnerships Enhance the Biopharma Value Chain
Strategic biopharma alliances are increasingly embedding multimodal AI across the entire drug‑development pipeline. Partnerships leverage foundation models to sharpen biomarker discovery and patient selection for complex modalities such as antibody‑drug conjugates. Large‑scale genomic and clinical datasets enable virtual cell modeling...

Roche’s $1.05 B PathAI Deal Highlights Massachusetts AI Hub Momentum
Roche agreed to buy Boston‑based PathAI for $750 million upfront, with a potential $300 million earn‑out, a deal valued at up to $1.05 billion. The transaction, praised by coalition members including Whoop, signals deepening venture capital confidence in Massachusetts’ AI cluster.

FDA Approves Enhertu for Neoadjuvant and Adjuvant HER2‑Positive Early Breast Cancer
The U.S. Food and Drug Administration has approved Enhertu (fam‑trastuzumab deruxtecan‑nxki) for two new HER2‑positive early‑breast‑cancer indications – neoadjuvant treatment of stage 2‑3 disease and adjuvant therapy for patients with residual invasive disease after standard therapy. The approvals rest on phase 3...

Module 4, Section 3: Selectivity and Site
The fourth lecture in the Protein Structure & Target Pharmacology module examines how drug discovery teams achieve selectivity. It outlines strategies for designing ligands that discriminate across the entire proteome, within closely related protein families, and even between proteins that...

New Antidepressants: Tackling Treatment Resistant Depression
New rapid‑acting antidepressants are reshaping treatment for the roughly one‑third of patients with treatment‑resistant depression who do not benefit from SSRIs or SNRIs. FDA‑approved agents such as esketamine and the newer oral combo Auvelity provide relief within minutes to weeks,...

Novartis Reveals More Data Behind Pluvicto Expansion Bid
Novartas reported new PSMAddition trial data showing that adding its radioligand therapy Pluvicto to standard hormonal treatment cuts PSA progression by 58% in hormone‑sensitive metastatic prostate cancer. Deep PSA reductions below 0.2 ng/mL were achieved by 87.4% of patients versus 74.9%...
Amgen’s Rare Disease Drug Tavneos Tied to 20 Deaths in Japan
Amgen’s rare‑disease drug Tavneos has been linked to 20 deaths among roughly 8,500 Japanese patients, primarily due to vanishing bile duct syndrome, a severe form of drug‑induced liver injury. The Japanese distributor Kissei Pharmaceutical has warned doctors against initiating new...

Regeneron Inks a ~$2.3B Deal with Parabilis Medicines to Advance Antibody-Helicon Conjugates Across Multiple Therapeutic Areas
Regeneron has struck a strategic research collaboration with Parabilis Medicines to develop antibody‑Helicon conjugates (AHCs) across several therapeutic areas. The agreement provides Parabilis with $125 million, including $50 million upfront and a $75 million equity‑financing commitment, and makes it eligible for roughly $2.2 billion...

Sandoz Reports the EC Approval of Bysumlog and Dazparda (Biosimilars, Humalog and NovoRapid)
The European Commission has granted approval for Sandoz's biosimilar insulin pens Bysumlog (insulin lispro) and Dazparda (insulin aspart). Both products are authorized as prefilled pens for diabetes treatment in adults, children and patients as young as one year, demonstrating efficacy...
Regeneron Misses Again as Melanoma Combo Bows to Merck’s Keytruda in Phase 3
Regeneron reported that its investigational LAG‑3 antibody fianlimab, combined with the PD‑1 blocker Libtayo, failed to show statistically significant survival benefits versus Merck’s Keytruda in a Phase 3 first‑line melanoma trial. Median progression‑free survival was 11.5 months for the high‑dose combo...

Magic Mushrooms Could Be Effective Treatment for Cocaine Addiction, Study Shows
Researchers published a small clinical trial showing that a single dose of psilocybin increased the likelihood of abstaining from cocaine compared with a placebo. The study involved 19 participants receiving psilocybin and 17 receiving diphenhydramine, all of whom engaged in...

BIO Supports and Seeks Refinements to FDA’s Plausible Mechanism Framework
The FDA has issued draft guidance introducing a Plausible Mechanism Framework to evaluate safety and efficacy of individualized, disease‑targeted therapies when traditional trials are infeasible. The Biotechnology Innovation Organization (BIO) issued a supportive comment letter, praising the agency’s direction while...

What Psychedelic Clinical Trials Could Teach Psychiatry
Treatment‑resistant depression (TRD) affects roughly one‑third of U.S. adults on antidepressants and remains a high‑need, low‑treatment area. Compass Pathways’ synthetic psilocybin, COMP360, recently achieved its primary endpoints in two Phase III trials—258 patients in COMP005 and 581 in COMP006—showing rapid, durable...

Supercharging Immune Cells May Help Control HIV Long-Term
Scientists have repurposed CAR‑T cell therapy, originally used for cancer, to target HIV. In a small Phase 1 trial, two participants who received the engineered T cells remained off antiretroviral drugs with undetectable viral loads for nearly two years and one...

AstraZeneca Reports the US FDA Approval of Baxfendy (Baxdrostat) To Treat Hard-to-Control Hypertension
AstraZeneca’s Baxfendy (baxdrostat) received U.S. FDA approval for use with other antihypertensives in patients with uncontrolled or treatment‑resistant hypertension. The approval is based on the Phase III BaxHTN trial, which enrolled 796 participants and showed significant blood‑pressure reductions versus placebo. In...
Michigan Teen Gets FDA‑Approved Therapy to Delay Insulin Use in Type‑1 Diabetes
Fourteen‑year‑old Grayson Visco of Hudsonville, Michigan, started a teplizumab infusion that can postpone the need for insulin by a median of 2.7 years. The therapy, sold as TZield, received broader FDA clearance in April to treat children as young as...

The Future of Radiopharma Is Being Built by Nuclear Geeks in SLC
Nusco, a Salt Lake City physics firm, is building a 190,000‑sq‑ft radioisotope production facility featuring a novel ion‑source linear accelerator capable of producing up to 12 isotopes simultaneously. The plant, designed for 24/7 operation and earthquake resilience, aims to meet...

Japan’s MHLW Approves Boehringer’s Jascayd for IPF and PPF
Boehringer Ingelheim’s Jascayd (nerandomilast) has been approved by Japan’s Ministry of Health, Labour and Welfare for treating idiopathic pulmonary fibrosis (IPF) and progressive pulmonary fibrosis (PPF). It is the first phosphodiesterase 4B inhibitor with antifibrotic and immunomodulatory properties to receive such...
ABBV's BLA for Pivekimab Sunirine Remains Pending
$ABBV announced BLA submission for pivekimab sunirine back in Sep 2025, but no news since on whether this was accepted/Pdufa date set.
FDA Greenlights Enhertu for Early HER2‑positive Breast Cancer
#AZN Enhertu approved by US FDA both before and after surgery for treating patients with HER2-positive early breast cancer.

Lotte Biologics Expands Antibody Manufacturing Agreement with Ottimo Pharma
Lotte Biologics announced on May 18, 2026 an expanded collaboration with South Korean biotech Ottimo Pharma to manufacture its PD‑1/VEGFR2 dual‑paratopic antibody, OTP‑01. The agreement builds on a June‑2025 deal and adds commercial process development support at Lotte’s Syracuse Bio Campus in...

Genentech Reports FDA Approval of Tecentriq and Tecentriq Hybreza for Adjuvant Muscle-Invasive Bladder Cancer With ctDNA-Guided Treatment
Genentech’s Tecentriq and Tecentriq Hybreza received FDA approval as adjuvant therapies for adults with muscle‑invasive bladder cancer (MIBC) who show circulating tumor DNA (ctDNA) minimal residual disease after cystectomy. The approval is linked to Natera’s Signatera CDx assay, which was...

AstraZeneca and Daiichi Sankyo’s Enhertu Receives US FDA Approval in Early Breast Cancer for Two Indications
The U.S. FDA approved AstraZeneca and Daiichi Sankyo’s antibody‑drug conjugate Enhertu (trastuzumab deruxtecan) for two early‑stage HER2‑positive breast‑cancer indications. In the neoadjuvant setting, Enhertu combined with trastuzumab‑pertuzumab (THP) earned approval after the DESTINY‑Breast11 trial showed a 67.3% pathologic complete response...

Can Revolution’s 'Miracle' Pancreatic Cancer Drug Be Topped? Immuneering, Actuate Say Yes
Revolution Medicines reported Phase 3 data showing its oral RAS inhibitor daraxonrasib more than doubled median overall survival for advanced pancreatic cancer to 13.2 months versus 6.7 months on chemotherapy. The results triggered a 40 % share surge and will support global regulatory filings,...
4 Spaces Primed for the Next Wave of Gene Therapies
The FDA’s recent approval of Regeneron’s Otarmeni for hereditary hearing loss marks the first non‑oncology gene‑therapy clearance, underscoring a shift toward treating central‑nervous‑system, ophthalmic, cardiovascular and muscular disorders. Companies such as Lexeo, Lilly/AskBio, uniQure, REGENXBIO/AbbVie, Tenaya, Medera and Sarepta are...
This Startup Aims to Upend Biologic Drug Production With Implantable ‘Cell Factories’
Duracyte, a Rice‑University spin‑out, is developing an implantable "cell factory" that produces therapeutic proteins inside patients, eliminating the need for traditional biologic manufacturing and injection pens. The capsule‑sized device houses genetically engineered human cells, supplies them with nutrients from the...

Isomorphic Labs Raises $2.1 B to Expand AI-Driven Drug Discovery Platform
Isomorphic Labs announced a $2.1 billion financing round led by Thrive Capital, with participation from Alphabet, GV, CapitalG, MGX, Temasek and the UK Sovereign AI Fund. The capital will accelerate development of its IsoDDE AI drug design engine and aim to...
Nanoparticle Therapy Erases Alzheimer Plaques in Mice, Restores Cognition
Researchers from the Institute for Bioengineering of Catalonia, West China Hospital Sichuan University and UK partners reported that a single‑dose nanotechnology therapy cut brain amyloid‑β by up to 60% within an hour and restored memory in Alzheimer‑model mice after three...
Denali Therapeutics Secures FDA Approval for AVLAYAH, First Gene Therapy for Hunter Syndrome
Denali Therapeutics announced that the FDA granted accelerated approval to AVLAYAH, the first gene‑therapy targeting the neurologic manifestations of Hunter syndrome. The company said early commercial activity is already exceeding its launch forecasts, signaling strong physician and payer interest in...

Ridglan Farms Agrees to Sell 1,500 Beagles, Sparking New Debate on Animal Testing in Pharma
Ridglan Farms, a Wisconsin beagle breeder for scientific research, reached an agreement on April 30 to sell 1,500 dogs to rescue organizations, marking a rare victory for animal‑rights activists. The deal, brokered by Big Dog Rescue and the Center for...

Kailera Therapeutics IPO Priced at $16, Opens at $26 Targeting Obesity Market
Kailera Therapeutics priced its initial public offering at $16 per share and opened at $26 on April 17, raising capital to advance its GLP‑1 obesity drug pipeline. The move places the company directly against industry giants Eli Lilly and Novo Nordisk as...

FDA Authorizes Lenire® Device, Expanding Non‑Invasive Treatment for Chronic Tinnitus
The Tinnitus & Hearing Center of Arizona announced that Lenire®, a bimodal neuromodulation system, received FDA authorization as a non‑invasive treatment for chronic tinnitus. The device combines sound and tongue stimulation to retrain neural pathways, providing a drug‑free alternative for...

A Revolutionary Cancer Treatment Could Transform Autoimmune Disease
Researchers are expanding CAR T cell therapy, originally a cancer breakthrough, to treat autoimmune diseases such as multiple sclerosis, lupus, and stiff‑person syndrome. Early trials, including a 2025 Nebraska study and a 2025 Kyverna trial of 26 stiff‑person patients, report functional...
Ipsen's Corabotase Shows 66% Improvement in Glabellar Lines and 82% Patient Satisfaction
Ipsen presented Phase II data for its recombinant neuroinhibitor corabotase at the Scale Symposium, reporting a 66% composite response at week 4 and a 60.8% sustained effect at week 24 in moderate‑to‑severe glabellar lines. Patient satisfaction reached 82.8%, positioning the drug as...

Regeneron’s Phase 3 Melanoma Trial Misses PFS Goal, Shows 5‑Month Numeric Gain
Regeneron Pharmaceuticals reported that its Phase 3 trial of fianlimab (LAG‑3 inhibitor) combined with cemiplimab (PD‑1 inhibitor) did not achieve statistical significance for progression‑free survival versus pembrolizumab, despite a 5.1‑month median PFS advantage. The data reshape expectations for LAG‑3‑targeted regimens...

Alteogen Wins PTAB Ruling, Halting Halozyme's Challenge to ALT‑B4 Manufacturing Patent
Alteogen secured a victory at the U.S. Patent Trial and Appeal Board as Halozyme Therapeutics' petition to invalidate the company's ALT‑B4 manufacturing technology patent was denied. The PTAB found Halozyme's prior‑art arguments insufficient, keeping the core process for producing PH20...
Alumis Reports 68% PASI‑90 in Phase 3 Envudeucitinib Trial, Eyeing 2026 NDA
Alumis Therapeutics said 68% of participants in its Phase 3 envudeucitinib trial reached PASI‑90 by week 24, outpacing the TYK2 benchmark set by Bristol Myers Squibb. The data, presented at the American Academy of Dermatology meeting, supports an NDA filing targeted...

FDA OKs Daiichi Sankyo/AstraZeneca’s Enhertu for Two HER2‑Positive Breast Cancer Indications
The U.S. Food and Drug Administration granted approval for fam‑trastuzumab deruxtecan‑nxki, marketed as Enhertu, to treat two HER2‑positive breast cancer indications. The antibody‑drug conjugate, co‑developed by Daiichi Sankyo and AstraZeneca, now covers early‑stage and metastatic disease, widening its market reach.

Enveric Biosciences Posts $1.6M Q1 Loss, Raises $16.7M as EB-003 Enters Preclinical Trials
Enveric Biosciences announced a $1.63 million net loss for Q1 2026 but bolstered its balance sheet with $16.7 million from a private offering and placement. The capital infusion supports the pre‑clinical advancement of EB-003, its lead non‑hallucinogenic neuroplastogen targeting neurological disorders.
Wedbush Sets $165 Target on Oruka Ahead of 2026 Psoriasis Data
Wedbush analyst Martin Fan reiterated an Outperform rating on Oruka, raising the 12‑month price target to $165 from $63.90. The upgrade hinges on expected psoriasis trial readouts in the second half of 2026, which could unlock a multi‑billion‑dollar market. Investors...