
FDA Warning Letter Breakdown: A Manufacturer Tells Investigators Its AI Agent Never Said Process Validation Was Required
The FDA’s Center for Drug Evaluation and Research issued a warning letter to a Michigan‑based homeopathic drug manufacturer after an October 2025 inspection uncovered multiple CGMP violations, including unsanitary conditions, missing microbiological testing, and inadequate component verification. The letter’s standout section flagged the firm’s inappropriate reliance on artificial‑intelligence agents to draft specifications, procedures, and batch records, claiming the AI never indicated the need for process validation. FDA officials rejected that defense, emphasizing that AI‑generated outputs must be reviewed and approved by a qualified human quality unit under 21 CFR 211.22(c). This marks the first FDA warning letter explicitly addressing AI use in pharmaceutical manufacturing.

Pharma Pulse: The Ember Cube 2 and a $1 Trillion Prescription Spending Projection for 2026
Ember LifeSciences unveiled the Ember Cube 2, a modular cold‑chain solution with real‑time monitoring, GPS tracking and cloud visibility, earning a Red Dot “Best of the Best” award. At the Asembia AXS26 summit, policy experts warned that new pricing legislation is compressing post‑launch...

FDA Moves to Speed Review Support for Psychedelic-Related Treatments
The FDA announced a new fast‑track mechanism for psychedelic‑based therapies, issuing Commissioner’s National Priority Vouchers to three programs targeting depression and PTSD with psilocybin or methylone. It also granted an IND for DemeRx NB to study noribogaine hydrochloride as a...

Varenicline
Varenicline (Chantix) received FDA approval in 2006 as a partial α4β2 nicotinic receptor agonist, offering a middle‑ground approach between nicotine replacement and bupropion. Its mechanism delivers enough receptor activation to ease cravings while antagonizing nicotine’s rewarding effects. The drug quickly...

Merck Announces First Dose in Phase 3 Study with Enpatoran for Lupus Patients with Active Skin Manifestations
Merck announced the first patient has been dosed in its global Phase 3 ELOWEN program, testing the oral TLR7/8 inhibitor enpatoran in lupus patients with active skin manifestations. The double‑blind, placebo‑controlled studies—ELOWEN‑1 and ELOWEN‑2—will enroll roughly 200 participants each across 266...

CGT Global Appoints Charlotte Ivancic to Board of Advisors, Strengthening Strategic Leadership in Cell and Gene Therapy Policy and Market...
CGT Global announced the appointment of Charlotte Ivancic to its Board of Advisors, bringing over 25 years of federal health policy and legislative experience. Ivancic, a partner at FGS Global and former senior aide to Senate Majority Leader Bill Frist,...

What We Know (and Don’t Know) About Peptide Safety
Peptide safety is neither automatically assured by mimicking endogenous hormones nor inherently hazardous because research is incomplete. The risk profile depends on the specific peptide, the depth of clinical evidence, product purity, and real‑world usage conditions. Early‑phase trials provide maximum...

The FDA Real Time Clinical Trial Announcement Quietly Dissolves Phase Gates, Breaks Biotech Capital Markets Plumbing, and Opens a Founder...
The FDA announced on April 28 that real‑time clinical trials (RTCTs) are now live, with AstraZeneca’s phase‑2 lymphoma study and Amgen’s phase‑1b small‑cell lung cancer trial streaming data through Paradigm Health. The agency claims a 20‑40 % reduction in development timelines, but...

Hemab Therapeutics (COAG) IPO Deck
Hemab Therapeutics, a clinical‑stage biotechnology firm, announced its IPO deck on April 30, 2026. The company focuses on innovative therapies for rare blood‑coagulation disorders, including both bleeding and thrombotic conditions. Its pipeline features two Phase 2 candidates targeting hemophilia and antiphospholipid syndrome, and...

Xtalks Featured Member: Parminder Singh Bhasin, Senior Director, Head of Analytical Research and Project Management, Intech Biopharm. Ltd
Parminder Singh Bhasin, Senior Director at Intech Biopharm Ltd., has been highlighted as an Xtalks Featured Community Member. With more than 26 years in pharmaceutical R&D, he oversees analytical research, method validation, and project management for complex generics, pMDI, nasal...

A Combination Treatment Is Claimed to Produce Sizable Life Extension in Aged Mice
Seragon funded a pre‑clinical trial of SRN‑901, a proprietary oral cocktail that blends urolithin A, quercetin, nicotinamide riboside, alpha‑lipoic acid and the company’s SRN‑820. In 18‑month‑old mice on a Western diet, the regimen extended median remaining lifespan by 33% and cut...

Seaport Therapeutics (SPTX) IPO Deck
Seaport Therapeutics, a clinical‑stage biotech firm, is preparing an initial public offering to fund its oral drug program targeting major neuropsychiatric conditions such as depression and anxiety. The company’s IPO deck emphasizes a “proven path” in neuroscience, highlighting pre‑clinical data...

The Biotech Bi-Weekly: A Virtual Biology Initiative, a New Discovery Grant and a Protein Supplier to Watch in Cancer Research...
The biotech bi‑weekly highlights a wave of new funding and tools, starting with Biohub’s $500 million five‑year Virtual Biology Initiative to generate global multimodal datasets for predictive biology. Zymo Research launched the Fecal Microbiome Discovery Grant to support early‑stage researchers, while...

Qubit Pharmaceuticals Aims for Quadratic Speedup in Simulations
Qubit Pharmaceuticals and Singapore’s Centre for Quantum Technologies have deployed the first quantum Markov Chain Monte Carlo (qMCMC) algorithm on gate‑based quantum hardware. The two‑year partnership blends Qubit’s quantum chemistry know‑how with CQT’s expertise in circuit design, using Quantinuum’s H2...
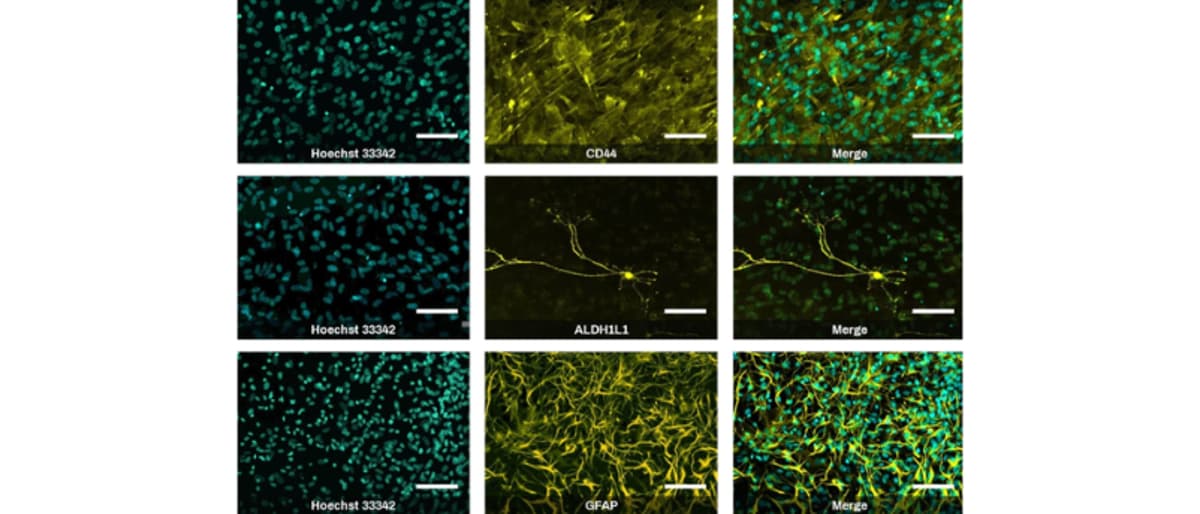
New iPSC Differentiation Kits for Neuroscience Research
AMSBIO introduced the Quick‑Glia™ product line, iPSC‑derived glial cell kits designed for neuroscience research. The kits convert human induced pluripotent stem cells into functional astrocytes or microglia in 1–2 weeks, delivering high‑purity, cryopreserved cells ready for disease modeling and drug...
A Study in Regulatory Conservatism versus Clinical Innovation
At today’s ODAC meeting, the FDA will review AstraZeneca’s oral selective estrogen receptor degrader camizestrant for hormone‑receptor‑positive, HER2‑negative breast cancer with ESR1 mutations. Late‑stage data showed a 56% reduction in risk of disease progression and consistent safety, along with improved...

Cardiovascular Health 2026
Recent studies highlight pitavastatin’s pleiotropic benefits beyond LDL‑C reduction, including functional HDL elevation, enhanced cholesterol‑efflux capacity, and antioxidative actions in dyslipidemic patients. Pre‑clinical work shows the drug strengthens blood‑brain barrier integrity and mitigates lipopolysaccharide‑induced BBB dysfunction, suggesting neuroprotective potential. Real‑world...

FDA-Approved Marijuana Products and Those Subject to State-Issued Licenses Rescheduled; Marijuana Hearing Calendared; Dispensary Applications Accepted
Acting Attorney General Todd Blanche and the DEA issued a final rule that instantly reschedules FDA‑approved THC drug products and marijuana sold under state medical licenses from Schedule I to Schedule III of the Controlled Substances Act. The rule aligns federal regulation...

Your Weekly TechBio News: High-Throughput Screening
High‑Throughput Screening (HTS) remains a cornerstone of modern drug discovery, allowing researchers to evaluate millions of chemical compounds against biological targets in a single campaign. Recent advances in robotics, miniaturized assay formats, and cloud‑based data pipelines have dramatically increased throughput...

Generally Good Indian Pharma Companies
An audit of top‑tier Indian generic manufacturers evaluated options for 20 therapeutic compounds, ranking firms by market capitalization, API vertical integration, ANDA volume, and GMP compliance. Cipla, Abbott, Sun Pharma, Zydus, Biocon and others were highlighted for specific products such...

Pharmaceutical Executive Daily: Rocket Pharmaceuticals Sells Pediatric Disease Priority Review Voucher
Rocket Pharmaceuticals agreed to sell its rare‑pediatric disease priority‑review voucher for $180 million in cash, providing non‑dilutive funding as the PRV program was reauthorized in early 2026. Teva Pharmaceutical announced a definitive agreement to acquire Emalex Biosciences for up to $900 million,...

Senate Report: Early COVID Vaccine Warning Signs Were Overlooked
A Senate Permanent Subcommittee report alleges the FDA ignored early COVID‑19 vaccine safety signals uncovered by an alternative data‑mining method in 2021. Analyst Dr. Ana Szarfman identified dozens of statistically significant adverse‑event signals, including cardiac death and Bell’s palsy, but...

The Pharma Sovereignty Playbook: How I’m Playing the $1.5 Trillion Drug Supply Crisis
A 2023 FDA inspection of Intas Pharmaceuticals’ plant in India halted half of the U.S. cisplatin supply, triggering nationwide shortages of both cisplatin and its substitute carboplatin. The crisis highlighted that only 24% of active pharmaceutical ingredient (API) manufacturing for...

AssureCare and Pillr Health Partner to Help Health Systems Tackle Specialty Pharmacy Costs
AssureCare and Pillr Health announced a strategic partnership to embed AssureCare's Akumen analytics platform into Pillr Health's Pharmacy Accelerator solution. The integration gives hospitals real‑time data and AI‑driven predictive modeling to manage specialty pharmacy costs, which are expected to exceed...

March 2026 Patent Highlights
The March 2026 Patent Highlights page aggregates the latest drug‑discovery milestones, from 38 first‑time small‑molecule approvals by Europe’s EMA, China’s NMPA and Japan’s PMDA to a deep dive on protein‑structure advances and machine‑learning tools. It spotlights a newly optimized HPK1 inhibitor...

Teva Enters $900 Million Agreement to Acquire Emalex
Teva Pharmaceutical Industries announced a $900 million acquisition of Emalex Biosciences, securing the late‑stage pediatric Tourette syndrome candidate ecopipam. The deal includes a $700 million upfront payment and up to $200 million in milestone fees, with the transaction slated to close in Q3 2026....

FDA Knew of Better Method to Detect COVID Vaccine Safety Signals — But Refused to Use It
Senator Ron Johnson released a report showing that FDA officials during the Biden administration knew a superior empirical Bayesian data‑mining method existed for detecting COVID‑19 vaccine safety signals in VAERS but ordered staff to continue using the older, flawed tool....
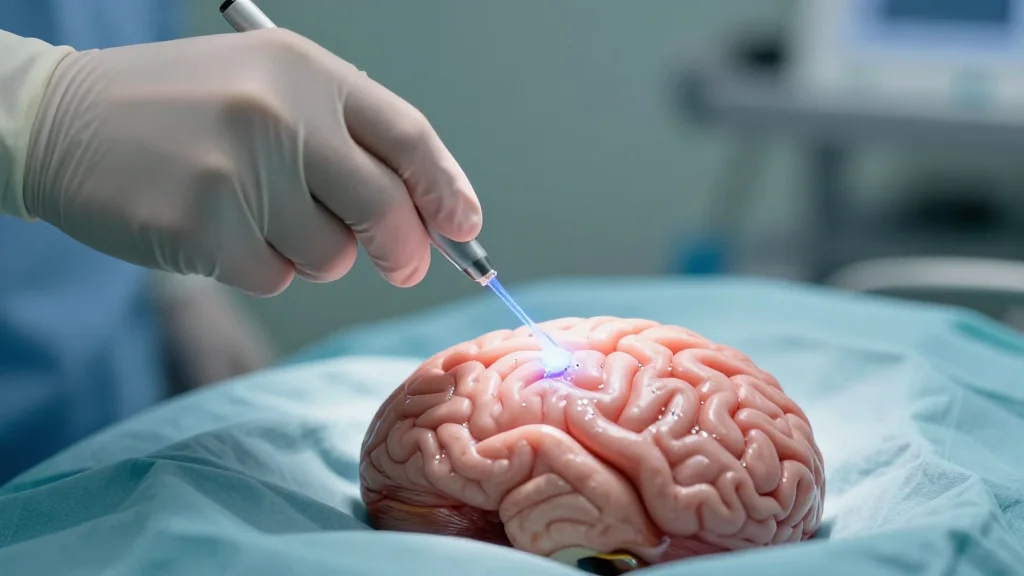
Nanozymes Against Brain Tumors
Researchers at Empa and HOCH Health Ostschweiz are developing biocompatible nanozymes that can be applied directly during brain‑tumor surgery to attack astrocytoma cells. The nanozymes act like enzymes, generating reactive‑oxygen species and activating drug precursors, and they are triggered by...

Reduced Ghrelin Receptor Activity Improves Mitochondrial Function and Muscle Function in Aged Mice
Researchers demonstrated that reducing activity of the ghrelin receptor (GHSR‑1a) improves muscle endurance and mitochondrial function in aged mice. Both genetic knockout and the inverse‑agonist PF‑5190457 increased markers of mitochondrial biogenesis and mitophagy, enhancing fatigue resistance. The interventions did not...

Word Games: How Moderna Is Selling Its Newest Vaccine without Using the “V” Word
Moderna’s $776 million federal award for a bird‑flu vaccine is under scrutiny after U.S. officials targeted mRNA technology, prompting the company to warn it may halt late‑stage vaccine programs. Simultaneously, Moderna and Merck are advancing an mRNA‑based cancer treatment, which Merck...

CRL Release Update – No Litigation War yet, but New Citizen Petition Beats the Drums
The FDA has begun posting Complete Response Letters (CRLs) for unapproved NDAs, ANDAs, and BLAs, releasing 127 documents to date, including 36 that had never been publicly disclosed. A Washington‑based law firm filed a Citizen Petition on April 20, 2026,...

Peptides / Bioregulators
The AGI House in San Francisco hosted the inaugural California Peptide Club, drawing over 100 invite‑only attendees to discuss self‑optimization peptides. Organiser Julius Ritter highlighted the fragmented information landscape and showcased stacks ranging from growth‑hormone releasers to cognitive enhancers. Days later, HHS...

FOXO4-DRI Is Fascinating, but Was Never Intended for Human Use, What Are the Takeaways?
FOXO4‑DRI is an experimental senolytic peptide that selectively eliminates senescent cells by disrupting the FOXO4‑p53 interaction, prompting p53‑mediated apoptosis. Pre‑clinical studies across vascular, reproductive, musculoskeletal and renal models report improved endothelial function, restored testosterone production, chondrocyte rejuvenation, and reduced frailty....

Peptides / Bioregulators
A new study examined 6,441 gray‑market peptide samples covering 14 compounds, measuring purity, dose accuracy, and endotoxin levels. Between 41.6% and 71.1% of the products failed basic pharmaceutical standards, and 2.4% contained no active peptide at all. Endotoxin contamination appeared...

Semiglutide Regenerates Cartilage Loss Through Weight Loss Independent Metabolic Restoration Mechanism
Semaglutide, a GLP‑1 receptor agonist originally developed for diabetes, is showing promise as a cartilage‑regenerating therapy. Preclinical pair‑fed mouse studies demonstrate cartilage protection independent of weight loss, while a 24‑week pilot in humans reported a 17% increase in cartilage thickness....

Alamar Biosciences IPO Raises $219.9M for Blood-Based Biomarker Tools
Alamar Biosciences closed an upsized IPO, raising $219.9 million by pricing 12.9 million shares at $17 each, and began trading on Nasdaq under the ticker ALMR. The California‑based firm offers the NULISA platform, which detects low‑level protein biomarkers in blood, and its...

Kailera Therapeutics IPO Raises $718.8M for Obesity Drug Pipeline
Kailera Therapeutics completed a $718.8 million IPO, pricing 44.9 million shares at $16 each and listing on Nasdaq under KLRA. The company’s lead candidate, ribupatide, is a once‑weekly injectable GLP‑1/GIP dual agonist currently in three global Phase III trials targeting obesity and type 2...
A Popular Senolytic Treatment Causes Brain Damage in Mice
A recent PNAS study shows that the widely used senolytic cocktail dasatinib plus quercetin (D+Q) impairs myelination in the mouse corpus callosum. The treatment altered oligodendrocyte morphology within minutes, reduced myelin thickness, and triggered endoplasmic reticulum stress, without killing the...

With Cell and Gene Therapies Proliferation, Specialty Pharmacy Faces New Pricing, Distribution Pressures
Cell and gene therapies are moving from ultra‑rare indications to broader patient populations, prompting a fundamental redesign of specialty‑pharmacy distribution and pricing models. At the 2026 Asembia meeting, experts warned that current flash‑title and buy‑and‑bill channels may not scale to...

Pharmaceutical Executive Daily: FDA Approves Breztri
Sun Pharmaceutical Industries agreed to buy Organon for $11.75 billion, propelling the combined entity into the top‑25 global pharma ranks. IMG Pharmaceutical announced the acquisition of Japan’s Matsumoto Pharmaceutical, adding a portfolio with more than 50 OTC approvals. The FDA granted...
Asembia AXS26 Summit: The Future of High-Cost Specialty Drugs
At the Asembia AXS26 Summit, Clarivate’s Dee Chaudhary warned that U.S. drug‑pricing policy is moving from theory to real‑time execution, reshaping payer behavior and market volatility. The Inflation Reduction Act’s Medicare price negotiations, the new One Big, Beautiful Bill Act, and renewed Most Favored...

Asembia AXS26: Jessica Lovett on the RWE That Matters Most
Jessica Lovett, VP of Commercial Strategy and Innovation at Innomar Strategies, discussed the most valuable real‑world evidence (RWE) for specialty medicines at Asembia AXS26. She highlighted two primary data sources: patient support programs and patient registries, both of which can...

Profluent and Lilly: The Next Gene Editor Will Be Designed by AI
Profluent, an Air Street Capital portfolio company, announced a multi‑program partnership with Eli Lilly to create AI‑designed recombinases for kilobase‑scale gene editing. The deal includes an upfront cash payment, committed R&D funding, and up to $2.25 billion in development and commercial milestones...

New York Proposes an Additional Layer of Legal Protection to Those Profiting From Injecting Vaccines
New York is introducing Senate Bill A9140, which would grant blanket legal immunity to doctors, pharmacists and other entities that prescribe, dispense, order, furnish, or administer vaccines to minors. The bill bars any civil lawsuit alleging injury, including residual effects,...
Ashvattha Therapeutics Announces Presentations Highlighting Mechanism of Action for Migaldendranib in Diabetic Macular Edema and Neovascular Age-Related Macular Degeneration
Ashvattha Therapeutics presented Phase 2 data on its subcutaneous nanomedicine migaldendranib (MGB) for diabetic macular edema and neovascular age‑related macular degeneration at ARVO. The two‑stage trial showed stable central subfield thickness for up to 12 weeks and maintained visual acuity without...

Cellares and Cabaletta Bio Sign 10-Year Commercial Supply Agreement to Scale Rese-Cel
Cellares has entered a 10‑year commercial supply agreement with Cabaletta Bio to manufacture rese‑cel, the company’s autologous CAR‑T therapy for autoimmune diseases, using its fully automated Cell Shuttle and Cell Q platforms. The deal secures long‑term capacity to produce thousands of...

Drug Pricing, PBM Reform, and the 2026 Midterms: What You Need to Know
Analysts at Avalere Health’s AXS26 summit warned that the 2026 midterm elections will reshape drug pricing and PBM practices. Recent FTC settlements require Express Scripts, CVS Caremark and Optum Rx to end spread pricing and unlink compensation from rebates, while...
FDA Approves Breztri for Patients 12 Years and Older with Asthma
The FDA has approved Breztri Aerosphere, a single‑inhaler triple‑combination of budesonide, glycopyrrolate, and formoterol, for maintenance treatment of asthma in patients aged 12 and older. This marks the first U.S. approval of a triple therapy that adds a long‑acting muscarinic...
Targeting Senescent Cells as a Treatment for Chronic Obstructive Pulmonary Disease
Researchers have introduced BCLXL-PROTAC, a proteolysis‑targeting chimera that degrades the anti‑apoptotic protein BCLXL in senescent lung cells. In primary small‑airway epithelial cells and fibroblasts from COPD patients, the compound induced caspase‑3‑mediated apoptosis and lowered classic senescence markers such as p21,...

Senescent Macrophages Are Important in Liver Aging and Liver Disease
Researchers identified a distinct p21‑positive, TREM2‑positive senescent macrophage population that accumulates in aging and fatty livers. These cells drive chronic inflammation through a senescence‑associated secretory phenotype linked to type I interferon signaling. In mouse models, senolytic agents that selectively eliminate these...