
Pharmaceutical Executive Daily: FDA Expands Approvals for Tzield and Dupixent
The FDA broadened Tzield’s approval to treat children as young as one year with stage 2 type 1 diabetes, aiming to delay progression to stage 3 disease. It also expanded Dupixent’s label to cover young patients with uncontrolled chronic spontaneous urticaria, adding another pediatric indication. Separately, AbbVie announced a $195 million investment to build a new manufacturing campus in North Carolina, reinforcing U.S. production capacity. These moves reflect a regulatory shift toward earlier intervention and a corporate push to reshore biologics manufacturing.
Department of Justice Officially Reschedules Marijuana: What Truckers Need to Know
The U.S. Department of Justice officially moved state‑licensed marijuana from Schedule I to Schedule III, allowing only FDA‑approved products and those tied to state medical licenses to benefit. The change acknowledges medical value but does not legalize cannabis federally and leaves commercial...

Zotatifin
Effector Therapeutics and Switzerland’s SJP Biotec have entered Phase 2 trials of zotatifin, an intravenous eIF4A inhibitor, in selected advanced solid tumors. The study targets cancers such as breast, lung and pancreatic that rely heavily on dysregulated protein translation. Early Phase 1...
Advancing Drug Discovery with Cell Line Development: Past, Present and Future
Cell line development underpins biologics manufacturing and drug discovery, from historic HeLa and CHO lines to modern CRISPR‑engineered clones. Recent advances in automation, artificial intelligence, and gene editing have accelerated clone selection, improved monoclonality verification, and increased protein yields. Tools...
New Genetically Engineered CHO Cell Line Boosts Protein Expression and Productivity
Sartorius has unveiled a genetically engineered Chinese hamster ovary (CHO) cell line that delivers up to twice the protein expression titers and three times the productivity of traditional wild‑type CHO hosts. The new line was validated across multiple therapeutic formats—including...

Medicare Steps Up on Obesity Care. Will Health Insurers?
The Centers for Medicare & Medicaid Services (CMS) announced an extension of its GLP‑1 Bridge program through 2027, offering seniors obesity drugs at a $50 monthly copay. The bridge operates outside Medicare Part D, removing financial risk for private insurers. CMS...
Cell Line Development: Pitfalls, Challenges and Solutions
Cell line development is critical for discovering targets and manufacturing biotherapeutics, yet achieving reliable monoclonality and early productivity assessment remains a bottleneck. Traditional approaches such as limiting dilution and FACS are labor‑intensive, often yield low‑viability clones, and delay project timelines....

Merck & Co., Inc. (MRK) Gains FDA Priority Review for KEYTRUDA Regimen in Muscle-Invasive Bladder Cancer
Merck announced that the U.S. FDA has granted Priority Review to two supplemental Biologics License Applications for KEYTRUDA and KEYTRUDA QLEX, each combined with Padcev, targeting muscle‑invasive bladder cancer patients eligible for cisplatin chemotherapy. The agency set an action date...

Viewpoint — ‘Completely Unethical’: RFK, Jr.’s Medical Ignorance Deprives Melanoma Cancer-Sufferers of a Life-Saving Therapy
Replimune’s RP1, a promising therapy for metastatic melanoma, received an initial FDA advisory panel recommendation for approval, but the agency’s biologics chief Vinay Prasad overruled the decision and halted the drug’s launch. At a House hearing, Health and Human Services...

GLP-1 Weight Loss Drugs Trigger a Life-Threatening Brain Condition by Depleting Vitamin B1
A recent analysis of FDA adverse‑event reports and case studies linked 15 instances of Wernicke encephalopathy—a severe, vitamin B1‑deficiency brain disorder—to the use of GLP‑1 weight‑loss drugs such as semaglutide and tirzepatide. Most patients experienced prolonged nausea, vomiting, and rapid weight...

What Does DOJ’s New Corporate Enforcement Policy Mean for the FDA- and DEA-Regulated Industry?
The Department of Justice released its first department‑wide Corporate Enforcement and Voluntary Self‑Disclosure Policy (CEP), establishing a uniform framework for prosecuting corporate crime. The policy lets companies obtain a declination of charges if they voluntarily disclose misconduct, fully cooperate, remediate...

Pharmaceutical Executive Daily: Idvynso Receives FDA Approval
The FDA granted approval to Idvynso, a new oral therapy for HIV‑1 infection in adults, citing trial data that showed sustained viral suppression and a safety profile on par with existing regimens. The drug adds to a shifting HIV market...
Neuromodulation and Nerve Ablation for Chronic Pain with NeuroOne CEO Dave Rosa — Episode 252
NeuroOne Medical Technologies, led by veteran CEO Dave Rosa, is advancing minimally invasive electrodes that combine nerve ablation, neuromodulation, and direct drug delivery for conditions like epilepsy and chronic pain. Rosa, who has spent three decades at firms such as...

Curve Biosciences Announces Key AI and Clinical Advancements of Whole-Body Intelligence for Chronic Diseases
Curve Biosciences announced two major milestones: its genomic AI foundation model will be presented at the International Conference on Learning Representations (ICLR) and its Whole‑Body Blood Test demonstrated strong performance in a real‑world liver cirrhosis monitoring study. The study enrolled...

New Review Casts Doubt On Alzheimers Drugs But Is Controversial
A new Cochrane review of 17 trials involving more than 20,000 Alzheimer’s patients concludes that amyloid‑targeting monoclonal antibodies deliver only trivial cognitive benefits and carry safety risks. The analysis groups together all anti‑amyloid antibodies—including older failures—thereby diluting the modest gains...

Progress Against Pancreatic Cancer, Part One
Revolution Medicines reported that its RAS‑targeting small molecule daraxonrasib more than doubled overall survival for patients with metastatic pancreatic ductal adenocarcinoma, extending median survival to 13.2 months versus 6.7 months on standard chemotherapy. The drug works by stabilizing a novel...

New Study Reveals CRISPR Enzyme that Responds to Human DNA Methylation
A collaborative team from Wageningen University & Research and the Van Andel Institute has identified a CRISPR-associated enzyme that senses DNA methylation, a key epigenetic mark distinguishing cancer cells from normal tissue. The enzyme selectively binds to methylated human DNA,...

Top Drugs at Risk of Supply Shortages: Report
The U.S. Pharmacopeia’s new vulnerable medicines report flags 100 drugs—half of which depend on a single‑country key starting material—as prone to supply shocks. While only 30 of these are in active FDA shortage, the list highlights injectables, ICU sedatives, IV...

CovAngelo Accurately Models Reaction Barriers for Covalent Drug Discovery
BEIT introduced CovAngelo, a layered QM/QM/MM platform that accurately predicts activation barriers for covalent inhibitors. By combining classical molecular mechanics, quantum‑mechanical embedding (ECC‑DMET), and high‑level quantum chemistry focused on the bond‑forming event, the method captures subtle electronic and environmental effects...

The Great Unbalding. Fallen Follicles, Rise! (NY Mag)
Scientists at Pelage Pharmaceuticals unveiled PP405, a novel drug that reprograms dormant hair‑follicle stem cells to regrow thick hair on balding scalp. Early Phase 2a data released in June 2024 showed rapid regrowth in areas previously considered irreversibly lost, sparking intense...

New Study Published Evaluating PharmaSens All-in-One Insulin Patch Pump
PharmaSens AG announced that the Journal of Diabetes Science and Technology published data from the first clinical feasibility study of its niia all‑in‑one insulin patch pump, which combines insulin delivery with continuous glucose monitoring. The single‑arm trial enrolled 18 adults...
Divalent siRNA Clinical Trial Is Now Recruiting
A first‑in‑human trial of a divalent PrP‑siRNA (2439‑s4) is now enrolling 15 symptomatic prion disease participants. The FDA‑cleared IND permits a single‑ascending‑dose study, testing 50 mg, 100 mg and 200 mg levels to assess safety and target engagement. The trial includes an optional...

Discovery of a Small Molecule HPK1 Inhibitor for Immuno-Oncology
A biotech firm has disclosed a novel small‑molecule inhibitor of hematopoietic progenitor kinase 1 (HPK1) that demonstrates potent immuno‑oncology activity in preclinical models. The compound achieves sub‑micromolar potency, oral bioavailability, and drives up to 70% tumor regression when combined with...

Scaling AI in Pharma Requires More Than Algorithms
Pharma companies are racing to embed AI, reporting productivity lifts such as 28% faster target identification and 22% quicker biomarker discovery. However, fewer than half of executives feel their firms are truly ready to scale these technologies. Capgemini’s Sheetal Chawla...

What Does It Take to Scale Cell and Gene Therapies From Discovery to Commercialization
MaxCyte CEO Maher Masoud says scaling cell and gene therapies requires developers to partner with manufacturers that can move seamlessly from R&D to commercial production. Integrated, best‑in‑class platforms eliminate the need for repeated process re‑optimization, enabling consistent, automated manufacturing. Advances...

The $500,000 Drug and the Cost of Modern Medicine
A 70‑year‑old man with no cardiac symptoms was diagnosed with wild‑type transthyretin amyloid cardiomyopathy after a routine coronary calcium scan revealed a score over 600. The diagnosis triggered a cascade of advanced imaging and a biopsy, leading to approval of...
CellCarta Eliminates 9-Hours-Per-Week Regulatory Bottleneck with RegASK’s AI-Driven Intelligence Platform
CellCarta, a global contract research organization, partnered with RegASK to overhaul its regulatory intelligence function. By deploying RegASK’s agentic AI platform, the CRO replaced a manual nine‑hour‑per‑week monitoring process with near‑real‑time updates. The new centralized hub automatically captures, validates and...

Agenus Names BAP Pharma as Exclusive Global Partner for BOT+BAL Access Programs
Agenus appointed BAP Pharma as its exclusive global partner to manage early‑access programs for the botensilimab‑balstilimab (BOT+BAL) immunotherapy combo. The collaboration will handle France’s government‑reimbursed Autorisation d’Accès Compassionnel (AAC) pathway and paid named‑patient programs in several other markets. Agenus has...

Charles River Highlights Effectiveness of VCGs in Toxicology
Charles River Laboratories published a retrospective analysis of 20 nonclinical toxicology studies that replaced traditional concurrent control groups with virtual control groups (VCGs). The review found 100% concordance in No Observed Adverse Effect Level (NOAEL) determinations and demonstrated up to...
Star Therapeutics Receives FDA Rare Pediatric Disease and Breakthrough Therapy Designations for VGA039 in Von Willebrand Disease Prophylaxis
Star Therapeutics announced that the FDA has granted both rare pediatric disease and Breakthrough Therapy designations to its lead candidate VGA039, a monoclonal antibody aimed at preventing bleeding in von Willebrand disease (VWD). The designations support the ongoing Phase 3 VIVID‑6 study,...

Simulations Plus Announces Collaboration with Lonza and U.S. FDA to Advance Predictive Frameworks for Complex Oral Drug Products
Simulations Plus announced a funded collaboration with CDMO Lonza and the U.S. FDA to create a mechanistic, predictive framework for amorphous solid dispersion (ASD) oral drugs. The partnership will combine Lonza's advanced in‑vitro dissolution testing with Simulations Plus' GastroPlus and...

The AI Drug Discovery Capital Stack in 2026: Who Has Raised the Most, Why Their Technical Approaches Actually Differ, and...
The essay maps AI‑driven drug‑discovery firms’ capital stacks as of April 2026, highlighting that Eikon, Xaira, Isomorphic Labs and Recursion sit at the top of disclosed funding. It separates the sector into four technical lanes—structure foundation models, generative chemistry, phenomics/perturbational biology,...
Patients Stay Cancer-Free Three Years After Clinical Trial
A phase II trial at UCL tested pembrolizumab as neoadjuvant therapy for 32 patients with stage II‑III MMR‑deficient/MSI‑high bowel cancer. After up to nine weeks of immunotherapy before surgery, 59% showed no detectable tumor and none experienced recurrence over a median...

“Cancer Isn’t Political, It’s Personal”: A Funding Update From the 2026 AACR Annual Meeting
At the 2026 American Association for Cancer Research (AACR) Annual Meeting in San Diego, scientists displayed “Thank you, Congress” signs after lawmakers blocked a proposed 40% cut to NIH funding. A policy town‑hall highlighted how the 2025 funding uncertainty delayed trials,...
A Pivotal Year for Regulatory Reform and Innovation
Britain’s Medicines & Healthcare products Regulatory Agency (MHRA) is advancing its MedTech regulatory reform, introducing a pre‑market statutory instrument that adds an international reliance scheme, updated classification, mandatory UDIs and stricter claim rules. The agency also plans an Early Access...
PEPITEM as a Potential Therapy for Autoimmune Arthritis
Researchers at the University of Birmingham have identified a decline in the anti‑inflammatory peptide PEPITEM as a key driver of worsening inflammatory arthritis with age. Laboratory tests showed that adding synthetic PEPITEM restores white‑blood‑cell responsiveness to adiponectin in early‑stage rheumatoid...

Dr. Kaeberlein's Optispan Podcast Series - Rapamycin and More
The Optispan podcast hosted by Dr. Kaeberlein outlines a translational protocol for 3‑hydroxyanthranilic acid (3HAA), a mouse‑tested longevity molecule. Using FDA BSA scaling, the human equivalent dose (HED) is calculated at roughly 1.1 g per day for a 70‑kg adult. Safety...

When a Text Message Tries to Short-Circuit the FDA
A text exchange between Joe Rogan and Donald Trump suggesting a quick FDA green‑light for ibogaine sparked concern about bypassing the rigorous drug‑approval process. The post explains that FDA approval is deliberately methodical, requiring phased clinical trials and safety data,...
AACR26 Innovative Early Stage Developments to Watch Out For
At the AACR annual meeting in San Diego, four cutting‑edge oncology programs were showcased in a single session. Each candidate is at or just beyond the threshold for first‑in‑human trials, spanning bispecific antibodies, RNA‑based therapeutics, CRISPR‑edited cell therapies, and novel...

Major Antineoplastic Mechanisms of Combination Ivermectin-Mebendazole
Recent preclinical analyses highlight the anticancer potential of combining ivermectin and mebendazole, two antiparasitic drugs repurposed for oncology. Ivermectin suppresses proliferative signaling pathways such as Wnt/β‑catenin, Akt/mTOR, and STAT3, while mebendazole disrupts microtubule polymerization, inducing G2/M arrest. Together they inhibit...

AACR San Diego 2026: New Drugs on the Horizon
The AACR 2026 Annual Meeting in San Diego unveiled 11 first‑time disclosed oncology candidates spanning small‑molecule degraders, bispecific antibodies, T‑cell engagers and ADCs. Highlights include NEO‑811, a CRBN‑mediated molecular glue targeting HIF‑1β for VHL‑deficient renal cancer, and AZD8359, a STEAP2‑directed T‑cell...

Pharmaceutical Executive Daily: Eli Lilly Acquires Kelonia Therapeutics
Eli Lilly announced a $7 billion deal to acquire Kelonia Therapeutics, adding genetic‑medicine and novel delivery technologies to its pipeline. In parallel, President Trump issued an executive order to speed FDA reviews of psychedelic therapies, signaling a policy shift toward emerging mental‑health...

Psychedelics Go Mainstream
President Donald Trump issued an executive order to speed up research and access to psychedelic therapies, allocating $50 million in federal funding and instructing regulators to dismantle long‑standing barriers. The move validates a growing investment thesis that the psychedelic sector will...

Mitrix Bio as an Example of the Trend Towards Alternative Paths to Initial Human Data
Mitrix Bio reported preliminary Phase 1 safety results for large‑dose mitochondrial infusions, showing no immediate adverse effects in two older participants. The company simultaneously opened Right‑to‑Try clinics in Dallas, Newport Beach and Palm Beach, offering the experimental therapy under a patient‑driven model. Its...

GLP-1 May Only Be the Beginning, Not the End of the Story
Researchers led by Richard DiMarchi and Matthias Tschöp published a paper in Molecular Metabolism showing that triple agonist retatrutide can drive weight loss even when GLP‑1 signaling is blocked. Their preclinical work demonstrates that co‑activating GIP and glucagon receptors produces...

Marty Makary: The FDA’s Quiet Blockade on Safer Nicotine
The article argues that FDA Commissioner Marty Makary is deliberately stalling approvals for low‑risk nicotine products such as vapes, heated tobacco and nicotine pouches, despite clear scientific consensus that they are far less harmful than smoking. Youth e‑cigarette use has...
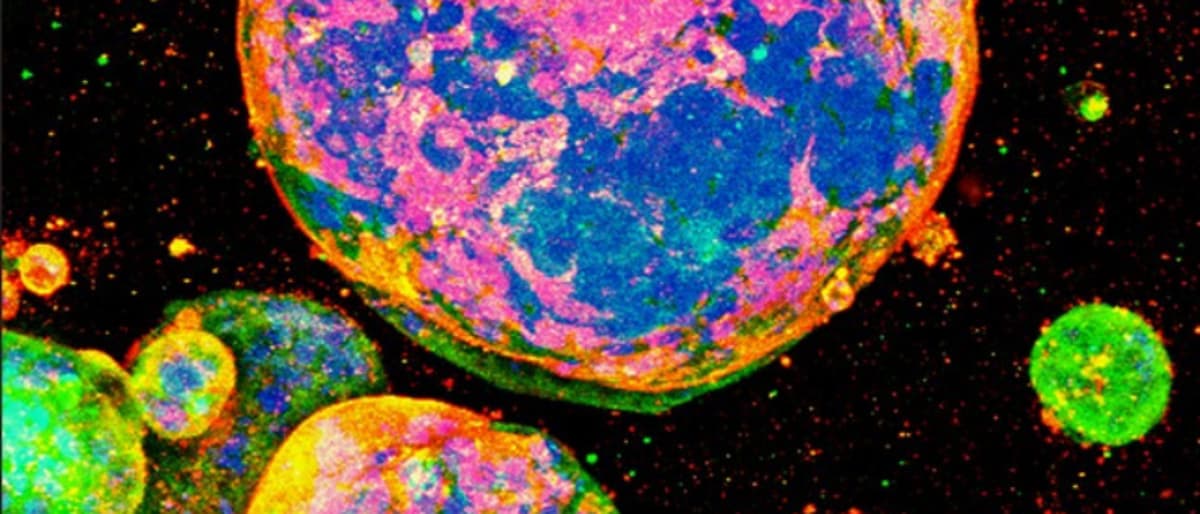
Fully Defined 3D Culture Substrate for Cancer Research
AMSBIO announced that its fully defined MatriMix 511 extracellular matrix enables patient‑derived colorectal cancer cells to form robust 3D organoids. In a Kyoto University study, the organoids preserved stage‑specific tumor biology and expressed metastatic markers, outperforming alternative matrices. MatriMix’s composition...

Using AI to Advance Validated Real-World Evidence
The HealthVerity event highlighted how AI can turn early‑stage life‑science questions into reproducible real‑world evidence (RWE) within minutes. Speakers from HealthVerity, Chiesi USA, Argenx and Medeloop shared practical methods for building transparent, iterative analyses that support feasibility, patient‑journey, regulatory and...

Rogan Tipped the Scales on Psychedelic Research in the US
Joe Rogan appeared at the White House as President Trump signed an executive order to fast‑track psychedelic research and clinical trials. The directive directs federal agencies to accelerate approval pathways for psychedelic therapeutics aimed at PTSD, veteran mental health, and...

What to Know Before Buying GLP-1 Drugs Online
The FDA warns that many online ads for GLP‑1 weight‑loss drugs promote compounded versions that lack the rigorous testing of brand‑name products like Ozempic, Wegovy, and Mounjaro. These compounded formulations can vary in concentration, contain unapproved ingredients, and have been...