
FDA Approves the First PROTAC in History, a $1B siRNA Wave Hits Cardiometabolic Disease, and Cytokinetics Cracks Non-Obstructive HCM –...
The FDA granted its first-ever approval for a PROTAC drug, Arvinas’ Veppanu, targeting ESR1‑mutated metastatic breast cancer, marking a regulatory milestone for targeted protein degradation. In parallel, precision‑medicine siRNA deals surged: Madrigal paid $25 million upfront (up to $975 million in milestones) for Arrowhead’s PNPLA3‑targeting asset, while GSK inked a $1 billion agreement for SiranBio’s ALK7 siRNA aimed at visceral‑fat reduction. Novo Nordisk’s oral Wegovy generated roughly $355 million in its inaugural full quarter, underscoring the profitability of GLP‑1 obesity therapies. Cytokinetics reported that aficamten achieved both primary and secondary endpoints in a Phase 3 trial for non‑obstructive hypertrophic cardiomyopathy, a disease lacking approved disease‑modifying treatments.

Endometriosis Imaging Study Highlights 99mTc-Maraciclatide as Diagnostic and Monitoring Tool
Serac Healthcare and Oxford’s Nuffield Department have published Phase 2 results of the DETECT study, showing that the gamma‑emitting radiotracer 99mTc‑maraciclatide can non‑invasively locate endometriotic lesions, including superficial peritoneal disease. Imaging findings matched laparoscopy in 16 of 19 participants and identified disease...

A Light at the End of the Tunnel for Huntington’s Disease Treatment
Researchers at Florida Atlantic University have uncovered a cellular pathway that enables mutant huntingtin protein (mHTT) to travel between neurons via tunneling nanotubes (TNTs). Using LC‑MS/MS, they identified the intracellular pH sensor Slc4a7 as a critical membrane partner of the...

Webinar Q&A Follow Up: Immunoassay Signal Amplification: Bold New Solutions for Existing ELISAs
Cavidi’s principal scientist Peter Stenlund explained how the BOLD signal‑amplification platform boosts ELISA sensitivity by lowering the lower limit of quantification while modestly reducing the upper limit. The technology relies on click‑chemistry conjugation of stable DBCO‑modified oligos, offering precise stoichiometry...

Cera and uMed Announce New Partnership
Cera, Europe’s largest HealthTech provider, has partnered with uMed’s automated living registry platform to bring clinical trial recruitment into patients’ homes. The collaboration leverages Cera’s network of over 2.5 million monthly home‑care visits and uMed’s regulated digital consent platform to reach...

FDA’s Expedited Drug Reviews Are Hailed in some Quarters but Other Approval Practices Are Problematic
In July 2025 the FDA launched the Commissioner’s National Priority Voucher (CNPV) pilot to slash drug review times from six‑to‑eight months down to one‑or‑two months, issuing 17 vouchers for therapies ranging from cancer to cholesterol drugs. Commissioner Marty Makary later...

Trump’s New Drug Advertising Proposals Fall Short on Public Health and the Constitution
The Trump administration’s FY 2027 budget proposes two contrasting measures: a sensible requirement that compounding pharmacies prominently disclose that their products have never been evaluated by the FDA, and a broader effort to give the FDA new authority to deem FDA‑approved...

Why Not Do Random Testing in Randomized Trials Designed to Measure Risk of Infection?
The post argues that randomized trials measuring infection risk, such as the recent Moderna flu study, rely on symptom‑driven testing rather than random testing of all participants. This selective approach inflates reported efficacy by omitting mild or asymptomatic cases and...

Supreme Court Keeps Abortion Pill Mail Access in Place for Now
The U.S. Supreme Court has left in place lower‑court rulings that allow the abortion pill mifepristone to be mailed while the case proceeds. By refusing emergency relief, the Court maintains the current federal framework for medication abortion. The decision highlights...

Pharmaceutical Executive Daily: Angelini Pharma Acquires Catalyst Pharmaceuticals in $4.1 Billion Agreement
Angelini Pharma announced a definitive agreement to acquire Catalyst Pharmaceuticals for $31.50 per share in cash, representing a 21% premium to the unaffected closing price and a total equity value of roughly $4.1 billion. The transaction gives Angelini its first direct...
Targeting Ischemic Disease with DiaMedica CEO Rick Pauls — Episode 254
DiaMedica Therapeutics, led by CEO Rick Pauls, is advancing a recombinant KLK1 protein to treat ischemia‑driven diseases such as preeclampsia, fetal growth restriction, and acute ischemic stroke. The podcast episode highlights the company’s focus on restoring vascular blood flow and...

Asembia AXS26: How Drug Innovation Could Strain Access Systems
In a follow‑up interview at the Asembia AXS26 summit, Aradigm Health CEO Will Shrank warned that emerging therapies priced above $1 million per patient could overwhelm existing market‑access and financing frameworks. He highlighted upcoming treatments for type 1 diabetes, wet macular degeneration...

Parallel 3D Bioprinting Builds Tissue Model Arrays in Minutes
Researchers have introduced a slippery‑liquid‑infused porous surface (SLIPS) droplet microarray that enables parallel digital light processing (DLP) bioprinting of hydrogel tissue models. By removing physical walls and using hydrophilic spots on a superhydrophobic background, the system prints dozens to hundreds...

The FDA's One-Day Inspection Pilot Is Already Running
The FDA launched a one‑day inspection pilot announced by Commissioner Marty Makary, with roughly 46 screening assessments completed since April across food, biologics, medical devices and clinical research sites. Most assessments resulted in a No Action Indicated outcome, though a...

Let's Do This: Two Concrete Steps You Can Take to Fight Back Against Recent Terrible Court Decisions
The 5th Circuit Court of Appeals temporarily blocked a nationwide ban on mailing the abortion pill mifepristone, while the Supreme Court weakened Section 2 of the Voting Rights Act, prompting redistricting efforts in Alabama, Louisiana and Tennessee. In Georgia, three state...

With Gilead’s Reasonableness Standard, Side Effects May Vary
The California Supreme Court heard oral arguments in the Gilead Tenofovir case, where plaintiffs seek to impose a novel “duty to commercialize a safer alternative” on drug makers. The proposed duty would require manufacturers to act reasonably in bringing a...

The Goalposts Shifted in Berlin, Your Trial Timeline Didn’t
The European Society for Medical Oncology (ESMO) Breast 2026 conference in Berlin showcased a wave of late‑stage breast cancer data that is reshaping therapeutic benchmarks. Several trials reported higher pathologic complete response rates and novel biomarker‑driven combinations, tightening the competitive...

Will Wegovy Tablets Spark a “Turnaround” At Novo Nordisk?
Novo Nordisk’s newly launched Wegovy oral tablet is delivering a rapid market surge, with over two million U.S. prescriptions and first‑quarter sales of about $350 million, outpacing expectations. Weekly new prescriptions reached roughly 200,000 by mid‑April, marking the fastest uptake for...

Pharma Pulse: GLP-1 Momentum Builds While Lilly Expands Genetic Medicine Manufacturing
Eli Lilly opened its first dedicated genetic‑medicine manufacturing plant in Lebanon, Indiana, expanding U.S. capacity for advanced gene‑editing and RNA therapies. GLP‑1 drugs accounted for eight percent of all prescriptions filled in March 2026, highlighting their growing role beyond diabetes. Bayer...

Strengthening the Generic Drugs Supply Chain
The API Innovation Center published a whitepaper titled “From Fragility to Resilience,” highlighting weaknesses in the U.S. generic‑drug supply chain and proposing data‑driven solutions. The report, authored by VP Natalie de Graaf, identifies the most vulnerable drug classes and quantifies supply‑chain...

What Elements Is the Conversation About Bringing Pharma Manufacturing Home Missing?
President Trump’s 100% tariff on branded drugs aims to force pharmaceutical reshoring, but industry leaders warn the focus on APIs overlooks a critical upstream layer—key starting materials (KSMs). Charlie Lyon of the API Innovation Center notes that 98% of U.S....

The Vaccine Safety Signal the Media Still Won’t Read
A peer‑reviewed study published in Vaccine (Sept 2022) re‑analyzed Pfizer and Moderna mRNA COVID‑19 trial data and found excess serious adverse events—10.1 per 10,000 for Pfizer and 15.1 per 10,000 for Moderna—yielding harm‑to‑benefit ratios of roughly 4.4 : 1 and 2.4 : 1 respectively. The...

The Latest News in Vaccine Obstruction
Large-scale safety studies of COVID‑19 and shingles vaccines, analyzing millions of records, found rare serious side effects, but the FDA blocked their publication citing unsupported conclusions. The agency also refused to file Moderna’s mRNA flu‑COVID combo vaccine, despite European approval...

Did the CDC Improperly Block a Study Showing the COVID Vaccines Were Effective?
The CDC’s acting director, Jay Bhattacharya, delayed and ultimately blocked a COVID‑19 vaccine‑effectiveness paper from being published in the agency’s flagship MMWR journal. The author of the Substack post argues the decision was justified, citing the study’s reliance on a test‑negative...

Metabolic Stability in Peptide Therapeutics
Peptide therapeutics are gaining traction but remain hampered by poor metabolic stability, limited permeability, and rapid clearance. The article outlines four primary metabolic pathways—hydrolysis, oxidation, reduction, and conjugation—and examines the hurdles of oral delivery, in‑vitro tools, and experimental workflows used...

AstraZeneca’s Camizestrant Hit by FDA Advisory Committee Vote While Truqap Moves Ahead
FDA’s Oncologic Drugs Advisory Committee issued a split verdict on AstraZeneca’s oncology pipeline this week. The committee voted 3‑to‑6 against the benefit‑risk profile of camizestrant combined with a CDK4/6 inhibitor for ESR1‑mutated advanced breast cancer, despite a 56% progression‑free survival...

Pharmaceutical Executive Daily: Zentalis Doses First Patient with Azenosertib in Phase III Trial
Zentalis Pharmaceuticals announced the first patient dosing in the Phase III Aspenova trial of Azenosertib, an oral WEE1 inhibitor for Cyclin E1‑positive, platinum‑resistant ovarian cancer. The study is run with the GOG Foundation, the European Network of Gynecological Oncology Trials (EN‑GOT), and...

How Specialty Drug Commercialization Differs in Canada From the US
Jessica Lovett, VP of Commercial Strategy at Innomar Strategies, outlined a 24‑month planning horizon for specialty drug launches in Canada, emphasizing early coordination with Health Canada. The roadmap moves from pre‑development regulatory and import considerations to a 12‑18‑month development and...

Eli Lilly Opens First Dedicated Genetic Medicine Facility
Eli Lilly inaugurated Lilly Lebanon Advanced Therapies, its first dedicated genetic‑medicine manufacturing site in Lebanon, Indiana, alongside a $4.5 billion capital infusion. The investment lifts Lilly’s Indiana spending to over $21 billion since 2020 and its U.S. commitments to more than $50 billion. The...
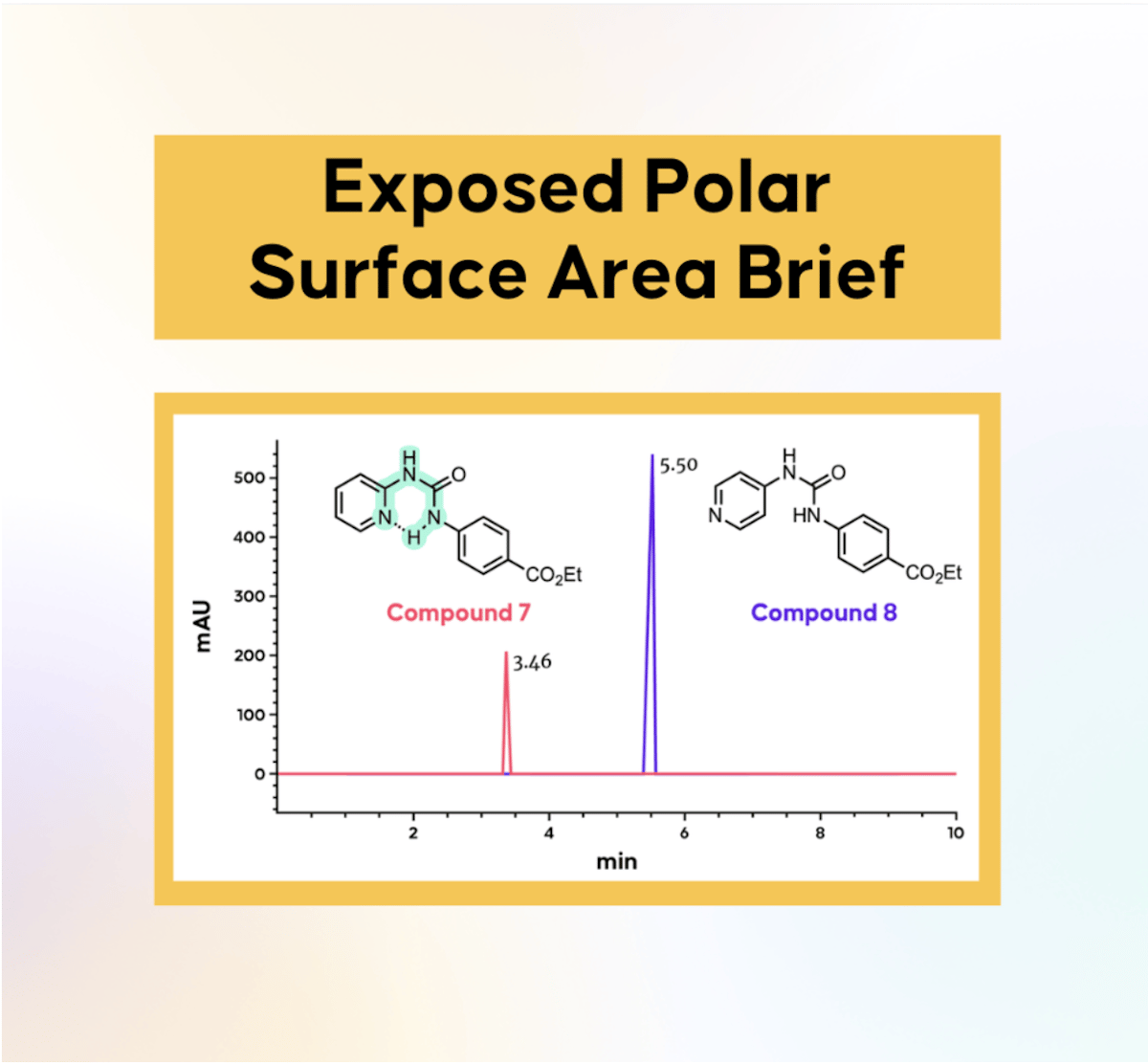
EPSA: A Useful Metric Across Chemical Space
The article highlights EPSA (Experimental Polarity Surface Area) as a robust metric for assessing molecular polarity across broad chemical space. Unlike traditional PSA, EPSA is derived from supercritical fluid chromatography, offering experimental insight into a compound’s three‑dimensional polarity profile. The...

Waiv Enters Collaboration with Daiichi Sankyo to Deliver AI-Derived Biomarkers for ADC Program
Waiv, the Paris‑based AI precision‑testing firm formerly known as Owkin Dx, has partnered with Daiichi Sankyo to lead digital pathology biomarker discovery for an antibody‑drug conjugate (ADC) program. The collaboration will apply Waiv’s end‑to‑end computational pathology platform to early‑phase trial data,...

Simulations Plus and NVIDIA Collaborate to Scale GPU-Accelerated, AI-Assisted Modeling Workflows
Simulations Plus announced a technical collaboration with NVIDIA to embed GPU‑accelerated and AI‑assisted modeling into drug‑development workflows. The partnership will re‑engineer Simulations Plus’ PBPK, PK/PD and QSP engines for NVIDIA GPUs, delivering up to a 75% reduction in simulation runtimes...

Most Oncology Brands Lose Before Launch Day — New ZoomRx Analysis of 40+ Drugs Quantifies the Awareness Gap That Predicts...
ZoomRx’s 2026 "Laggards and Leaders" analysis of 44 oncology brands shows that pre‑launch physician awareness drives commercial success. Brands entering the market with about 75% aided awareness outperform those starting at 55%, a 25‑point gap that persists through four years....

Understanding Supply Chain Constraints to Enable Growth for Cell & Gene Therapies
Cell and gene therapies are poised for growth, yet only 3 % of the 4,000+ candidates in development secure FDA or EMA approval. Prices ranging from $100,000 to $3 million per dose lack a unified reimbursement framework, creating payer uncertainty worldwide. Logistics...

Capturing True Single-Cell Resolution with Your Spatial Data
Spatial biology has transformed life‑science research, yet imaging and sequencing platforms still grapple with cell‑boundary segmentation and grid‑based spot limitations. Linda Orzolek of OMAPiX explains how Takara Bio’s Trekker technology delivers true single‑cell spatial resolution by isolating nuclei and pairing...

From Insight to Impact: Making Real-World Evidence Actionable in Urology
Real‑world evidence (RWE) is reshaping urology, but fragmented, unstructured data limits its impact on everyday clinical decisions. In non‑muscle invasive bladder cancer, real‑world datasets reveal wide variation in BCG maintenance and treatment sequencing, while prostate cancer diagnostics suffer from inconsistent...

Can Mammals Regrow Lost Limbs? This New Treatment Could Be the First Step
Researchers at Texas A&M have demonstrated that a two‑step treatment using growth factors FGF2 and BMP2 can trigger partial digit regeneration in mice. The protocol first applies FGF2 to create a blastema‑like cell mass, then adds BMP2 to drive bone...

“Sounds Great. Do You Want FDA Approval?” A Regulatory Analysis of Psychedelics
On April 18, 2026 President Trump signed an executive order compelling the FDA to prioritize review of psychedelic drugs, issuing priority‑review vouchers for three compounds already holding Breakthrough Therapy designation. The order also creates a Right‑to‑Try pathway for ibogaine, directs...

Pharmaceutical Executive Daily: BioNTech Reports 2026 First Quarter Results
Cellenkos secured FDA clearance to start a Phase 1b/2a trial of its off‑the‑shelf allogeneic T‑reg therapy CK0802 for steroid‑refractory graft‑versus‑host disease, with enrollment slated for late 2026. BioNTech reported first‑quarter 2026 revenue of $138 million, a 35% drop from the prior year,...

FDA Clears Investigational New Drug Application for Phase Ib/IIa Trial of CK0802 in Steroid-Refractory Graft-Versus-Host Disease
The U.S. Food and Drug Administration has cleared Cellenkos' investigational new drug application for CK0802, paving the way for a Phase Ib/IIa trial in patients with steroid‑refractory graft‑versus‑host disease (GVHD). The mid‑stage study will evaluate safety, tolerability and early efficacy, using...

Austin Russian: How Fragmentation Delays Rare Disease Therapy Access
Austin Russian, SVP of Program Excellence at PANTHERx Rare, warned that fragmentation across prescribers, insurers, pharmacies and manufacturers slows patient access to orphan drugs. As more rare‑disease therapies reach the market, the lack of a single coordinating entity creates miscommunication...

Partial Reprogramming Concern Altos Labs Is Becoming Less Stealthy
Altos Labs, launched in 2022 with roughly $3 billion in private funding, is intensifying its public profile as it pursues partial cellular reprogramming to reverse organ aging. The company is racing alongside rivals such as Life Biosciences, which has just begun...

PharmaLogic Expands Radiopharmaceutical Footprint With New Atlanta Facility
PharmaLogic announced the opening of a new PET radiopharmaceutical manufacturing facility in Atlanta, Georgia, as part of its $250 million US expansion. The state‑of‑the‑art plant will increase production capacity, accelerate delivery to Southeast hospitals, and serve as an innovation hub for...

Bracco Launches BubbleGen™ Early Access Program for Microbubble-Based Cell Selection and Activation at ISCT
Bracco Imaging announced an Early Access Program for its new BubbleGen™ technology, which uses buoyant microbubbles to isolate and activate specific cell subtypes. The platform offers a one‑step, magnetic‑residue‑free alternative to traditional bead‑based cell separation, initially demonstrated with CD3⁺ T‑cell selection...
New Pharmaceutical Reform Alliance Poll: Americans Agree on One Thing – Rein in Big Pharma
The Pharmaceutical Reform Alliance released a new national poll of 1,524 registered voters showing overwhelming bipartisan support for prescription‑drug pricing reform. Eighty‑nine percent of respondents favor reform, while 68% say drug prices have risen and 94% blame pharmaceutical companies for...

NImmune Biopharma Announces Presentations at Digestive Disease Week 2026 Supporting a Differentiated Profile and Superior Efficacy of Oral, Once-Daily NIM-1324...
NImmune Biopharma presented Phase 1 data for its oral LANCL2 drug NIM‑1324 at Digestive Disease Week, showing safety, tolerability, target engagement and superior efficacy versus existing IBD therapies. The study met all primary and secondary endpoints with no dose‑limiting toxicities and...
Hepta Reveals Blood-Based Epigenetic Signatures of GLP-1 Response, Enabling Precision Medicine in Obesity and MASH
Hepta unveiled a blood‑based cfDNA methylation assay at Digestive Disease Week 2026 that can identify patients who will lose at least 10% of body weight on semaglutide before the first dose. The SAMARA trial showed baseline epigenetic signatures distinguished responders...
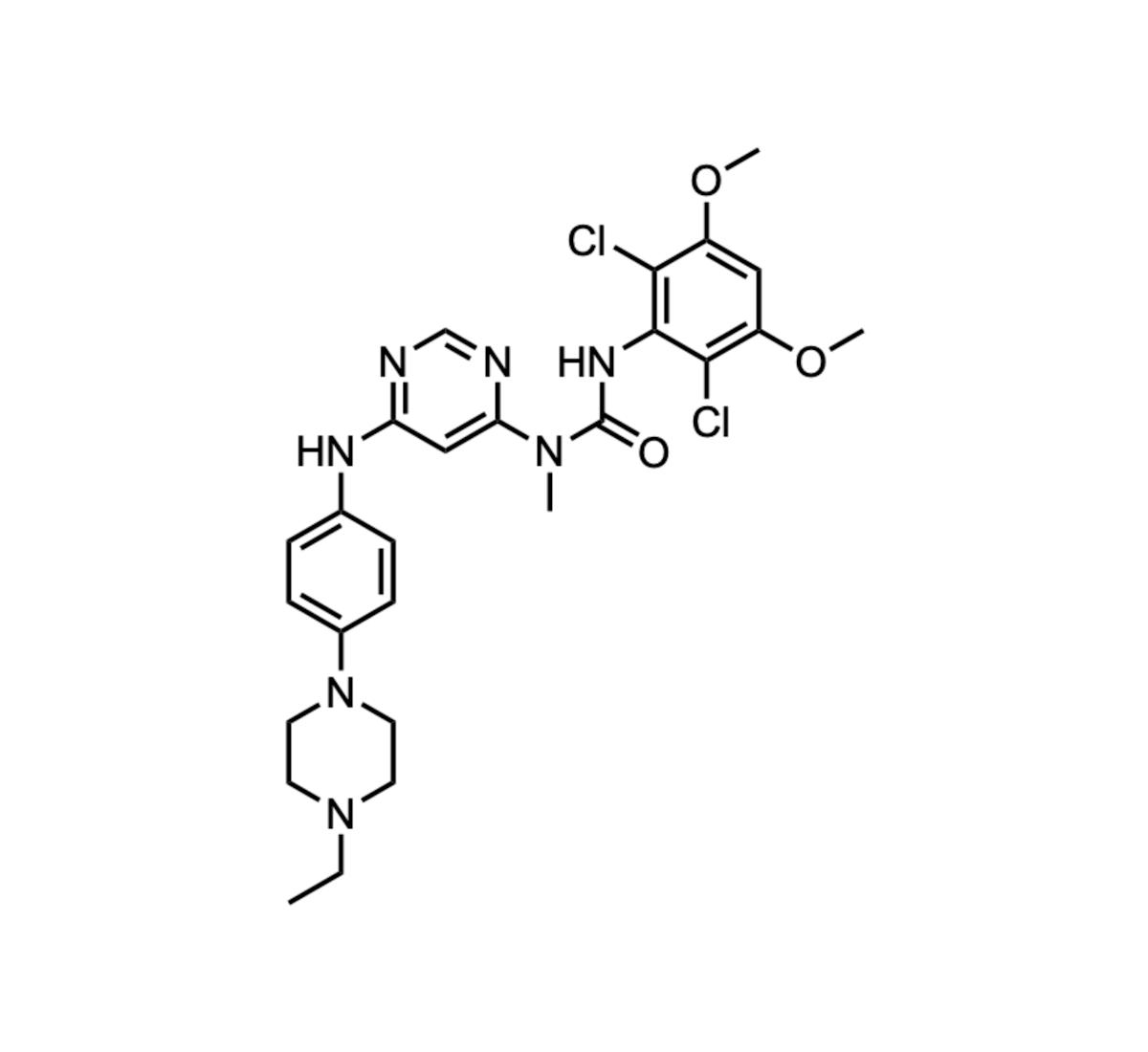
Infigratinib
Infigratinib, a pan‑FGFR inhibitor previously approved for cholangiocarcinoma, is being repurposed to treat achondroplasia. After its FDA accelerated approval was rescinded in 2024 due to enrollment challenges, BridgeBio reported that the Phase 3 PROPEL 3 trial met its primary endpoint in February 2026....

Connected by Design: How AI and Automation Are Transforming Drug Discovery at BMS
Bristol Myers Squibb (BMS) is shifting from isolated AI tools to an integrated, learning‑driven ecosystem that connects data, models, and automation across discovery and development. The company highlights its shared data backbone, AI co‑scientists, and lab‑in‑the‑loop automation as foundational layers,...

Odyssey Therapeutics (ODTX) IPO Deck
Odyssey Therapeutics, a clinical‑stage biopharma specializing in precision medicines for autoimmune and inflammatory disorders, unveiled its initial public offering deck in May 2026. The company aims to raise capital to advance its Phase 2‑tested pipeline, which targets conditions such as rheumatoid...