
Trump Appeal on Vaccines Validates Connecticut's New Immunization Law
The Trump administration appealed a federal court order that blocked its effort to strip vaccine recommendations for flu, rotavirus, hepatitis, meningitis and RSV, a move championed by Robert F. Kennedy Jr. Connecticut responded by passing a law that anchors the state’s immunization standards to multiple reputable medical bodies, not just the CDC’s Advisory Committee on Immunization Practices. The legislation also safeguards pharmacists’ authority to vaccinate and guarantees insurance coverage for the listed shots. Additionally, it launches a voluntary adult flu‑vaccine pilot for the uninsured and underinsured.

Pointed Ironies: SERD Wars, ADC Hype, and What Really Works in Breast Cancer
The FDA rejected camizestrant at the ODAC meeting, while approving vepdegestrant a day later. AstraZeneca’s vepdegestrant leverages ctDNA to detect ESR1 mutations early, allowing a treatment switch while patients remain on a CDK4/6 inhibitor backbone. In contrast, Arvinas pursued a...

The Technology Behind the Story: 3D Skin Grafts and the World of Scarpetta
The article highlights how 3D‑printed skin grafts are transitioning from laboratory experiments to clinical tools for complex wounds. Researchers at Columbia University have demonstrated patient‑specific, three‑dimensional grafts that fit irregular body parts like a glove, reducing surgery time and improving...
NHS to Roll Out ‘1-Minute’ Immunotherapy Jab to Tens of Thousands with Cancer
The NHS is introducing a sub‑cutaneous form of pembrolizumab (Keytruda) that can be administered in just one minute, replacing the traditional two‑hour IV infusion. The rapid jab is approved for 14 cancer types, including lung, breast, head‑and‑neck, and cervical cancers,...

Inhibiting ID1 and ID3 Reduces Lung Fibrosis
Researchers identified that the transcription factors ID1 and ID3 are markedly elevated in fibroblasts from idiopathic pulmonary fibrosis (IPF) patients. Using both small‑molecule inhibitors and gene‑silencing techniques, they suppressed these proteins in mouse models, which halted fibroblast activation and triggered...

Canagliflozin - Another Top Longevity Drug
The class of sodium‑glucose cotransporter‑2 (SGLT2) inhibitors has moved from glucose‑lowering pills to a cornerstone of cardiometabolic care. Large‑scale trials and real‑world registries consistently show that agents such as canagliflozin, dapagliflozin and empagliflozin cut heart‑failure admissions, slow chronic kidney disease...

Loyal Raises $100 Million: Dog Longevity Drugs Targeting IGF-1 and PPAR Pathways
Loyal, a biotech startup focused on canine longevity, raised $100 million in Series A funding to advance its anti‑aging pill that targets the IGF‑1 and PPAR pathways. The drug received a favorable “likely effective” assessment from the FDA, positioning it for market...

The FDA Wants to Make Many Popular Prescription Drugs OTC—A Great Idea. Here’s Why It’s Unlikely to Happen
FDA Commissioner Marty Makary argues that most prescription drugs should be reclassified as over‑the‑counter unless safety concerns exist. The article highlights how prescription status inflates prices by routing purchases through insurance, citing lower cash prices for OTC versions of Claritin,...

Telomeres: History, Health and Hallmarks of Aging
Bill Andrews, a co‑discoverer of human telomerase, argues that telomere shortening is the primary limiter of human lifespan and that systemic activation of telomerase can reverse biological aging. He promotes small‑molecule activators such as TAM‑818 and botanical blends like Telo‑Vital,...

Tonight in Your Rights: A Shadow Docket Surprise
The Supreme Court’s shadow docket, led by Justice Samuel Alito, issued a one‑week administrative stay that temporarily restores nationwide mail‑order access to the abortion pill mifepristone. The stay halts a Fifth Circuit ruling that had blocked remote dispensing after Louisiana...

The Fight to Lower Prescription Drug Prices in America #CareTalk
In a May 4 2026 CareTalk episode, host Laura Packard sits with Vinny DeMarco of Maryland Health Care for All to dissect the latest state‑level tactics for lowering prescription‑drug prices. The conversation spotlights Maryland’s $35‑per‑month insulin cap and Colorado’s new drug‑price transparency...

More OMQ Letters After FDA Inspections? Don't Treat Them as Noise
Pharmaceutical manufacturers are receiving more post‑inspection letters from the FDA’s Office of Manufacturing Quality (OMQ), even after inspections with only one or two Form FDA 483 observations. The rise aligns with a 27% jump in FY2024 drug quality inspections—62% of...

Pharmaceutical Executive Daily: UCB to Acquire Candid Therapeutics
UCB signed a definitive agreement to acquire privately held Candid Therapeutics for $2 billion upfront with up to $200 million in milestones. The deal, slated to close by late Q2 or early Q3 2026 pending antitrust clearance, adds a suite of T‑cell engager...

Two Strategies for Specialty Drug Savings—And How to Choose the Right One
Specialty drug prices are climbing, prompting employers to seek sustainable savings through two main pathways: alternative funding programs (AFPs) and in‑benefit optimization. AFPs tap manufacturer assistance or non‑traditional sourcing to lower out‑of‑pocket costs for high‑price therapies, but they add coordination...
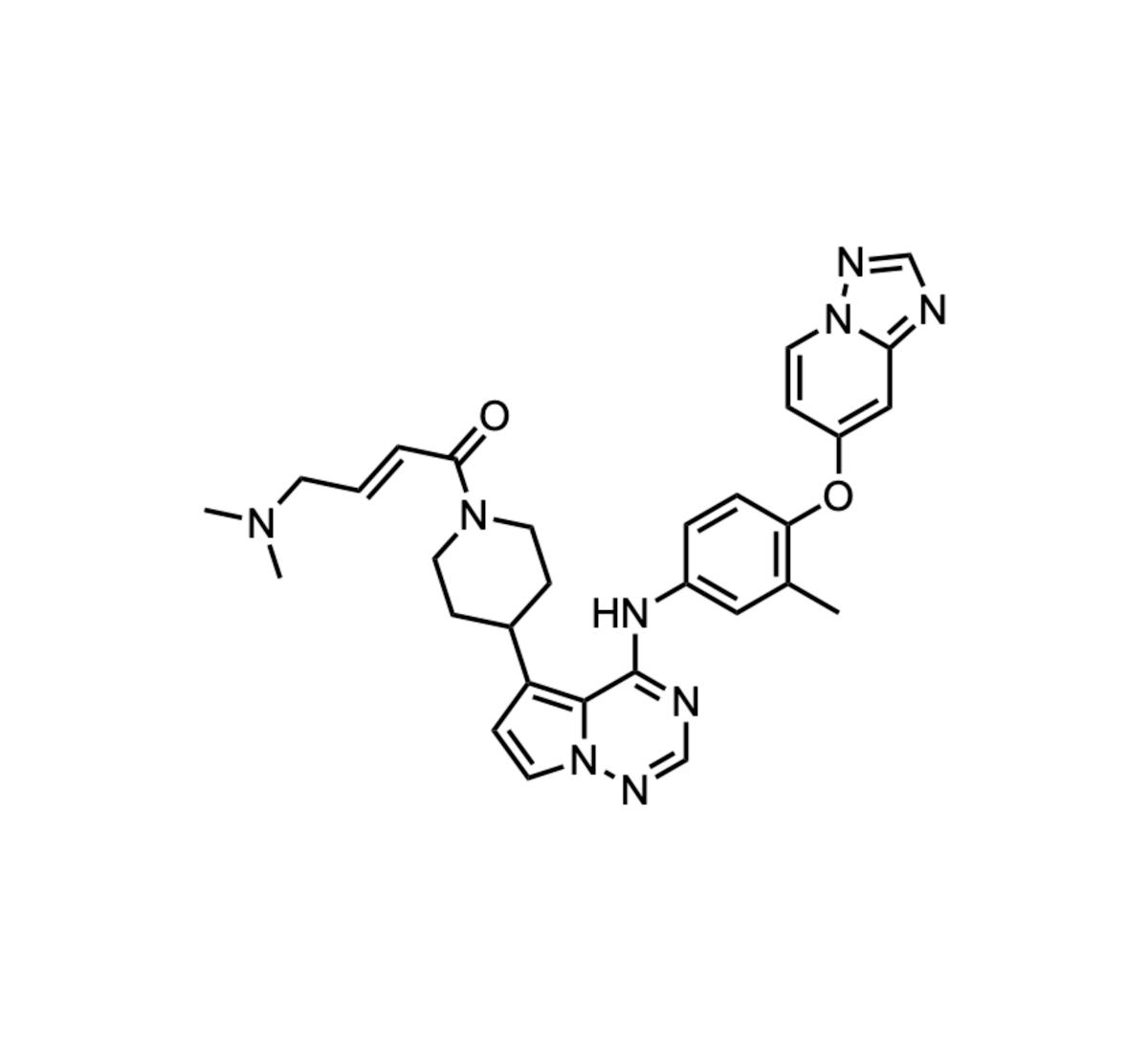
IAM1363
Iambic Therapeutics of San Diego announced the initiation of a Phase 1 clinical trial for an oral covalent inhibitor targeting HER2‑mutant cancers. The molecule, identified through an AI‑guided high‑throughput screening campaign, binds irreversibly to the mutant HER2 kinase domain. Preclinical data...

Asembia AXS26: Why Pharma Can’t Afford to “Go It Alone” Anymore
At the Asembia AXS26 Summit, Syneos Health’s SVP of Market Access Kim Plesnarski warned pharma that rising costs and complex access hurdles demand a shift from legacy patient‑support models to agile, outcome‑focused partnerships. She highlighted that many manufacturers still rely...

For Once, some Scientific Fraudsters Have to Pay Their Money Back to the Government.
Dana‑Farber Cancer Institute agreed to pay $15 million to resolve False Claims Act allegations that it submitted false certifications on six NIH research grants between 2014 and 2024. The institute admitted that researchers reused and altered images in 14 grant‑linked publications,...

Module 4, Section 2: All About Assays
The Module 4, Section 2 briefing provides a concise overview of modern assay platforms used in early‑stage drug discovery. It references key literature on PRMT5 fragment‑based screening that produced the MRTX1719 candidate, as well as thermal‑shift, surface plasmon resonance (SPR), and polymerase...

MAHA Vs. The FDA: Dredging up Old Anti-Regulation Revisionist History
A new essay in Science-Based Medicine denounces the “health‑freedom” narrative that seeks to dismantle the FDA. It traces the agency’s authority back to the 1962 Kefauver‑Harris amendment, which linked drug approval to rigorous safety and efficacy trials. The author dismantles...

Viewpoint: How ‘Health Care Guru’ Joe Rogan Circumvented the FDA’s Skepticism on Psychedelics
Joe Rogan directly messaged President Donald Trump about the therapeutic potential of psychedelics, prompting the president to sign an executive order that fast‑tracks FDA review of these drugs. The order creates a priority‑voucher system that accelerates approvals for psychedelic manufacturers,...

Hidden Ingredient in GLP-1 Tablets Raises New Gut Health Questions
Oral semaglutide tablets rely on the absorption enhancer SNAC (salcaprozate sodium) to cross the stomach lining, but only 0.4%‑1% of the drug reaches the bloodstream. A 21‑day rat study published in the Journal of Controlled Release found that the majority...

RESEARCH: NICLOSAMIDE in CANCER and Other Diseases - 2025 Review Paper From Henan, China
A 2025 review paper from Henan, China, evaluates niclosamide—a decades‑old anti‑parasitic—as a repurposed oncology agent. The analysis compiles pre‑clinical data across breast, lung, pancreatic and colorectal cancers, and highlights early‑phase clinical trials showing modest tumor responses. Researchers also discuss formulation...

FDA Closes the 503B Bulks Door on Semaglutide, Tirzepatide, and Liraglutide
On April 30, 2026 the FDA issued a proposal to exclude semaglutide, tirzepatide and liraglutide from the 503B Bulks List, arguing there is no clinical need for outsourcing facilities to compound these GLP‑1 drugs. The move follows the resolution of...

Rethinking Blood Thinners for Atrial Fibrillation Patients
At the American College of Cardiology meeting, a three‑year trial demonstrated that the Watchman left‑atrial‑appendage closure device provides stroke protection comparable to lifelong anticoagulation while causing far fewer bleeding events. The findings challenge the entrenched belief that atrial fibrillation patients...

Mifepristone Ruling and a Dive Into Trump's Dementia Riddled Brain
The Fifth Circuit Court of Appeals has halted telehealth prescriptions for mifepristone in Texas, Louisiana and Mississippi, leaving the ruling’s geographic reach and the prospect of a nationwide injunction uncertain. At the same time, former President Donald Trump’s Truth Social...

Promising New Technique Uses Nanoparticles to Detect Pancreatic Cancer
Scientists at Oregon Health & Science University have unveiled a blood‑based assay that uses an electronic jolt to harvest tumor‑derived nanoparticles, achieving 97% accuracy in detecting pancreatic cancer. The technique, validated in a blinded study of 36 participants, outperforms the...

The Commodification of Sensitive Open Data
The European Union’s European Health Data Space (EHDS) regulation, adopted in March 2025, will make the electronic health records of roughly 450 million residents available for secondary use by March 2029. The framework defaults to inclusion, requiring citizens to opt out and offering...

Incyte Announces FDA Approval of Jakafi XR™ (Ruxolitinib) Extended-Release Tablets for the Treatment of Myelofibrosis, Polycythemia Vera and Graft-Versus-Host Disease
Incyte announced FDA approval of Jakafi XR, an extended‑release ruxolitinib tablet, for adult intermediate‑ or high‑risk myelofibrosis, hydroxyurea‑intolerant polycythemia vera, and steroid‑refractory acute or chronic graft‑versus‑host disease in patients 12 years and older. The 55 mg once‑daily tablet is bioequivalent to the 25 mg...

224. The Return of the Mifepristone Mess
The Fifth Circuit issued a stay that reinstates the in‑person dispensing requirement for Mifepristone, undoing the FDA’s 2021 and 2023 rules that allowed mail‑order and telehealth prescriptions. The order, grounded in the Administrative Procedure Act, takes immediate nationwide effect, echoing...

Katherine Szarama, PhD, Named Acting CBER Director: Who’s Next?
Vinay Prasad stepped down as director of the FDA's Center for Biologics Evaluation and Research (CBER), and Katherine Szarama, PhD, was named acting director. Szarama, a biologist with prior experience as CBER deputy director and stints at CMS, Emerson Collective...

Pharmaceutical Executive Daily: FDA Approves Auvelity
The FDA approved Axsome Therapeutics’ Auvelity for agitation associated with Alzheimer’s disease, marking the first non‑antipsychotic and only the second drug cleared for this indication. Earlier, the FDA’s Oncologic Drugs Advisory Committee gave AstraZeneca’s Truqap a 7‑1 favorable benefit‑risk vote,...

How to Write Better IQ, OQ, PQ Protocols
Pharma, biotech, and medical‑device firms routinely draft IQ, OQ and PQ protocols, but many fail regulatory scrutiny because they treat the documents as paperwork rather than engineering verification. The most common flaws are vague acceptance criteria, copy‑forward templates that ignore...

GoodRx Expands Cash-Pay Access to Oral Ozempic for Patients With Type 2 Diabetes
GoodRx announced a new cash‑pay option for oral Ozempic, the tablet form of semaglutide, making it available through its nationwide pharmacy network. The program lists transparent monthly prices of $149 for the 1.5 mg dose, $199 for 4 mg, and $299 for...

Update on Brad Stanfield's Rapamycin Clinical Study in NZ
Brad Stanfield’s New Zealand rapamycin trial enrolled older adults on a 12‑week protocol, with participants typically taking 6 mg every other week. The study measured functional outcomes such as the chair‑stand test, sparking debate over whether short‑term dosing can reveal longevity benefits. Commentators...

Asembia AXS26: How CGTs Are Reshaping Specialty Pharmacy
Cell and gene therapies (CGTs) are fundamentally changing the specialty pharmacy landscape, moving beyond traditional buy‑and‑bill models toward highly coordinated, patient‑specific workflows. The therapies demand specialized treatment‑center activities such as apheresis, conditioning, and strict cold‑chain logistics. High upfront costs create...

RA/QA News Roll: Late April 2026
The FDA saw a leadership shuffle as Principal Deputy Commissioner Sara Brenner moved to a senior HHS counseling role, while Commissioner Robert Makary pushed a bold agenda to fast‑track psychedelic therapies, issuing three new National Priority Vouchers and signing an...
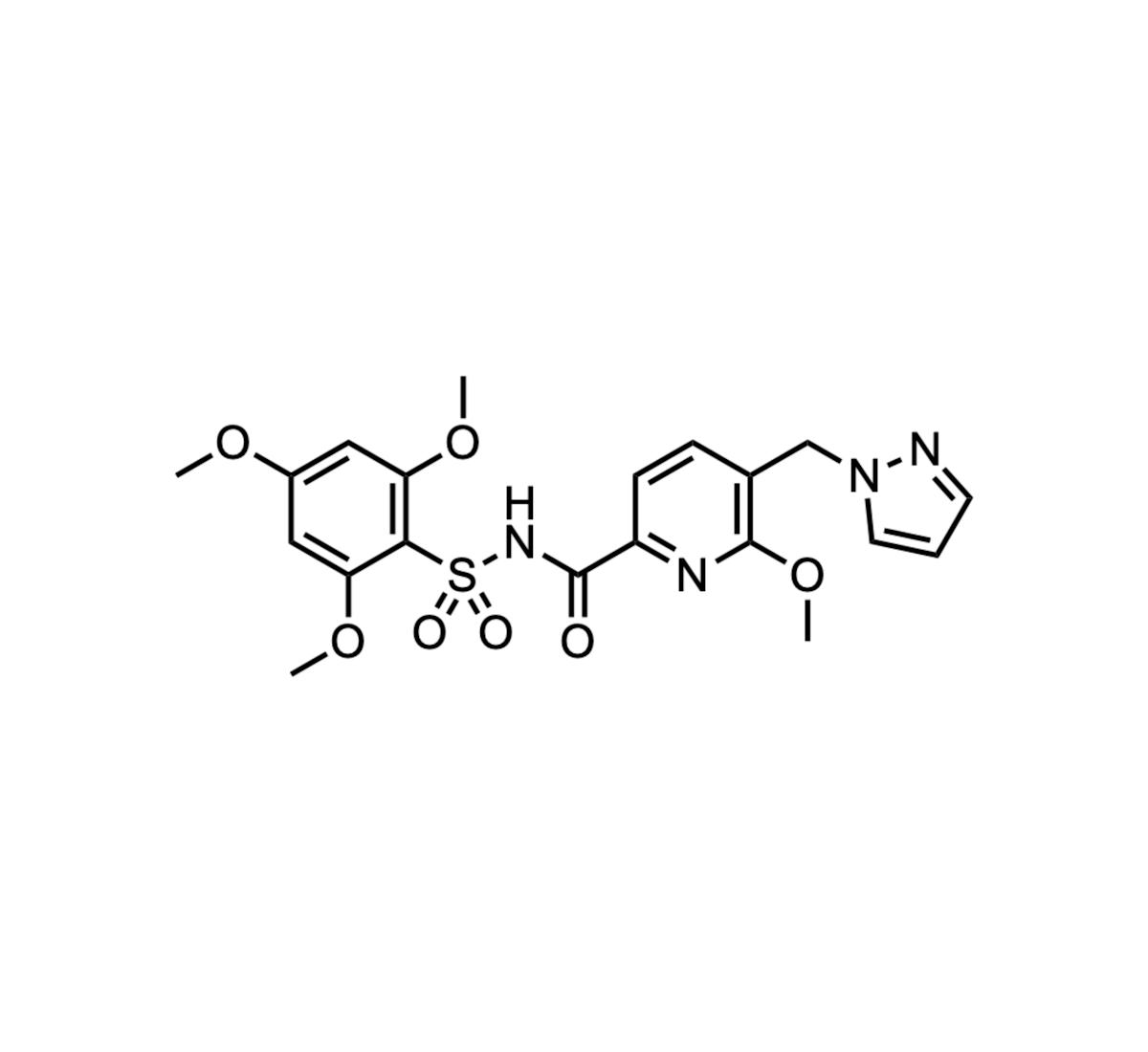
OP-3136
OP‑3136, a KAT6A‑selective inhibitor, entered Phase 1/2 trials for advanced hormone‑receptor‑positive breast cancer. The drug mimics the pyrophosphate of acetyl‑CoA using an acyl‑sulfonamide scaffold, delivering high specificity for the epigenetic writer KAT6A. Olema Pharmaceuticals is testing OP‑3136 in combination with SERDs...

Sydnexis to Present New Data From Phase 3 STAR Trial of SYD-101 at ARVO 2026 Annual Meeting
Sydnexis announced it will unveil new subgroup analysis data from the Phase 3 STAR trial of its low‑dose atropine eye drop SYD‑101 at the ARVO 2026 meeting in Denver. The analysis focuses on children with fast‑progressing myopia, a cohort that typically...

World Asthma Day 2026: New Anti-Inflammatory Drugs and More for Asthma Control
World Asthma Day on May 5 highlighted the persistent gap in access to anti‑inflammatory inhalers, a core theme of the Global Initiative for Asthma. The FDA approved AstraZeneca’s Breztri Aerosphere, the first single‑inhaler triple therapy that combines an inhaled corticosteroid, a...

New Semaglutide for Alcohol Use Disorder Trial Shows Big Drops in Drinking
A Lancet‑published, double‑blind, 26‑week trial found once‑weekly semaglutide markedly reduced alcohol consumption in participants with alcohol use disorder and obesity. Across primary drinking endpoints, the semaglutide arm showed statistically significant declines compared with placebo, despite both groups receiving identical cognitive‑behavioral...

Arrowhead Pharmaceuticals Receives TGA Approval of REDEMPLO® (Plozasiran) in Australia, Expanding Global Access for Patients with Familial Chylomicronemia Syndrome (FCS)
Arrowhead Pharmaceuticals announced that Australia’s Therapeutic Goods Administration approved REDEMPLO® (plozasiran), the first siRNA therapy for familial chylomicronemia syndrome (FCS) in the country. The drug, administered subcutaneously every three months, targets apoC‑III to dramatically lower triglycerides. In the Phase 3 PALISADE...

Global Disruptions to the Pharma Supply Chain: Q&A with Jeff Golfman
Jeff Golfman, founder of Send 123, warned that rising geopolitical tensions—particularly around Iran and the Strait of Hormuz—are tightening global pharmaceutical and medical supply chains. Shipping bottlenecks and shrinking capacity are driving up prices and threatening access to critical therapies such...

Space-Efficient Cryogenic Sample Storage
Azenta Life Sciences introduced the CryoArc™ Pico Automated Storage System, a compact LN‑2 cryogenic platform that maintains samples at –190 °C. Designed for biobanking, clinical research, and cell‑gene therapy labs, the system integrates barcode‑driven software for sample tracking, chain‑of‑custody, and CFR 21...

Zepbound Soars and Lilly Advises Patience on Foundayo
Eli Lilly reported a blockbuster first‑quarter, with revenue jumping 56% to $19.8 billion driven by soaring sales of its obesity drugs Zepbound and Mounjaro. Zepbound alone generated over $4 billion, up about 80% year‑over‑year, while Mounjaro nearly doubled to close to $9 billion. The...

Drug Amount Reporting: FDA Publicly Identifies over 7,700 Noncompliant Companies
On March 31 2026 the FDA released a public list showing that more than 7,700 drug manufacturers failed to submit the required 2024 drug‑amount reports. The list separates 1,254 firms with active drug listings from 6,480 firms with inactive listings, highlighting a...
New Genetic Discovery Could Spell This Aggressive Cancer’s Downfall
UCLA researchers uncovered a genetic weakness in small cell neuroendocrine carcinoma (SCNC) by creating prostate‑derived organoid models and running genome‑wide CRISPR screens. The screens identified the transcription factor E2F3 as a synthetic‑lethal partner of RB loss, and inhibiting E2F3 halted...

We Tried to Get Trump to Back Psychedelic Therapy — Here’s What Happened
The authors drafted a bipartisan policy blueprint to accelerate psychedelic‑based treatments, targeting veterans with PTSD, addiction and depression. After Joe Rogan shared the proposal, it amassed over 1.5 million views on X, prompting meetings with health‑policy influencer Calley Means and a...

Justices Poised to Protect Generics Manufacturers From Liability for Decisions of Pharmacists About Prescribing Their Products
The Supreme Court heard arguments in Hikma Pharmaceuticals USA v. Amarin Pharma, where Amarin seeks to hold generic maker Hikma liable for pharmacists dispensing its generic product in ways that infringe Amarin’s Vascepa patents. The Court appeared skeptical that Hikma’s...

Asembia ASX26: The Strain on the Development of Infrastructure for Sustainable C&G Therapy
Cell and gene therapies promise transformative treatments but remain hampered by steep costs across research, manufacturing, and delivery. At the Asembia ASX26 Summit, Aradigm Health CEO Will Shrank highlighted the fragmented U.S. payment landscape as a core barrier to sustainable...
Should Regulators Rely More on AI to Accelerate Drug Approvals?
Regulators, led by the FDA, are testing AI tools to speed drug approval processes. The agency’s pilot projects include AI‑driven real‑time monitoring of clinical trials and automated data review. Proponents argue AI can cut analysis time and uncover hidden patterns,...