
India’s Praj Industries Opens AI-Enabled Precision Fermentation Lab, Signs Government MoU
Indian biotech firm Praj Industries has opened an AI‑enabled Advanced Precision Fermentation Lab at its Pune R&D centre, Praj Matrix. The facility focuses on large‑scale precision fermentation to boost efficiency, cut carbon emissions and reduce scale‑up risk for pharma, food, cosmetics and biofuel markets. Praj signed an MoU with the Biotechnology Research and Innovation Council’s National Centre for Cell Science (BRIC‑NCCS), a government‑funded body, to accelerate strain discovery, process development and workforce training. The move dovetails with India’s 2024 BioE3 strategy, which is driving a network of biomanufacturing hubs nationwide.

Opinion: Prasad’s FDA Exit Good for Rare Diseases but New CBER Head Must Repair Eroded Trust
Vinay Prasad’s exit from the FDA’s Center for Biologics Evaluation and Research (CBER) follows a Senate hearing that highlighted inconsistent approval pathways for rare‑disease therapies. Industry sponsors and patient advocates complained that the agency reversed previously negotiated trial designs, leaving patients...

'Sterility Failures' Prompt FDA to Threaten Radiopharmaceutical Producer with Disciplinary Action
The FDA issued a warning letter to the University of California San Francisco Radiopharmaceutical Facility after sterility testing uncovered Bacillus contamination in a PET‑imaging agent batch. The agency found the facility’s explanation—that the bacteria entered the test tube during analysis—insufficient...

Weizmann Study Finds Genes Explain Up to 50% of Lifespan Variation
Researchers at the Weizmann Institute of Science, led by Ben Shenhar, published a study showing that genetics may account for roughly 50% of human lifespan variation, double previous estimates. The finding challenges the long‑standing view that lifestyle dominates longevity and...

FGF21 Shows Dual Action: Reduces Drinking, Shields Liver
The study validated FGF21-a liver-derived hormone currently in clinical trials for fatty liver disease “as a dual-action therapeutic that both curbs harmful drinking behaviors and protects against alcohol-related liver injury https://t.co/VdPZvQQ9qp

Results May Vary, But Reporting Shouldn’t: FDA Sends a Not-So-Gentle Reminder on ClinicalTrials.gov Compliance
On March 30, 2026, the FDA emailed more than 2,200 sponsors and investigators linked to over 3,000 clinical trials that have not posted required results on ClinicalTrials.gov or failed quality‑control review. The agency reminded that results must be posted within...
Belite Bio Files NDA for Tinlarebant After 35.7% Lesion Reduction in Phase 3 Trial
Belite Bio began a rolling New Drug Application for tinlarebant on April 21, 2026, after its Phase 3 DRAGON trial demonstrated a 35.7% reduction in retinal lesion growth for Stargardt disease patients. The filing could accelerate approval for the first...

OZMOSI Teams with Planview to Deploy AI‑Driven Portfolio Planning for Pharma R&D
OZMOSI announced a strategic partnership with Planview, integrating its machine‑readable clinical intelligence into Planview’s AI‑driven portfolio management platform. The deal gives pharmaceutical teams access to data from more than 800,000 trials, 35,000 drugs and 4,000 diseases, promising faster, data‑backed R&D...

Cumberland Pharma’s $100 Million Sale to Apotex Affiliate Sends Stock 44% Higher
Cumberland Pharmaceuticals Inc. agreed to sell its branded U.S. drug portfolio to an affiliate of Apotex for $100 million, prompting a 44% surge in its Nasdaq‑listed shares. The deal highlights ongoing consolidation in the specialty pharma market and raises questions about...

Houston Friday Night: Comedy, Immunization Partnership Celebrate Vaccines
Friday night in HoustonTX I will be there along with The Immunization Partnership and COMEDY INJECTION, VACCINES: 9th Wonder of the World https://t.co/GPSrrB3poV https://t.co/THhFN31ebn

Study Finds Rapamycin May Undermine Exercise Benefits
Researchers at the Lifespan Research Institute reported that rapamycin, a leading longevity drug, may blunt the physiological gains from regular exercise. The finding challenges the common biohacker practice of pairing rapamycin with fitness regimens and highlights a potential trade‑off between...

Hims & Hers Gains 7% on FDA Peptide Review, Faces Amazon Competition
Hims & Hers Health surged more than 7% when the FDA scheduled a July 23‑24 meeting to review several peptide drugs, giving investors a new growth narrative. The rally was later tempered as Amazon launched a competing GLP‑1 weight‑loss service,...

Spinogenix Starts CLARITY Phase 2b/3 Trial of SPG601 for Fragile X Syndrome
Spinogenix, a clinical‑stage biopharma, has launched the CLARITY trial – a two‑part Phase 2b/3 study of its oral BK‑channel modulator SPG601 in male patients with Fragile X Syndrome. The initial arm will enroll up to 48 adult males, followed by...

Tempus Teams with USC to Scale AI‑Driven Precision Oncology for 1.5 Million Patients
Tempus AI, Inc. and the Keck School of Medicine of USC announced a multi‑year collaboration that will embed Tempus’ AI platform into more than 1.5 million yearly patient visits across USC’s cancer centers. The deal covers molecular testing, trial matching, care‑gap...
RFK Jr.’s FDA Shake‑up Sparks Biotech Uncertainty
Health Secretary Robert F. Kennedy Jr.’s overhaul of the FDA, marked by rapid leadership turnover, layoffs and recent rejections of experimental cancer therapies, has left biotech firms uneasy. The turmoil coincides with a plunge in venture capital to its lowest level in two...

Blood-Derived microRNA Signatures Associated with Hippocampal Structure and Atrophy Rate: Findings From the Rhineland Study
Researchers analyzed blood‑derived microRNA profiles from over 2,000 participants in the Rhineland Study to uncover associations with hippocampal structure and its longitudinal atrophy. Cross‑sectional analysis identified a set of miRNAs—including miR‑199a‑3p/199b‑3p, miR‑155‑5p, miR‑146a‑5p and miR‑505‑5p—linked to larger left hippocampal volume,...

New Bioreactor Turns Stem Cells Into an Immune-Cell Factory, Producing 40 Million Human Macrophages per Week
Researchers at Hannover Medical School have unveiled a medium‑scale bioreactor that converts induced pluripotent stem cells into human macrophages at commercial‑grade volumes. The system can harvest up to 40 million immune cells per bioreactor each week for up to ten weeks,...
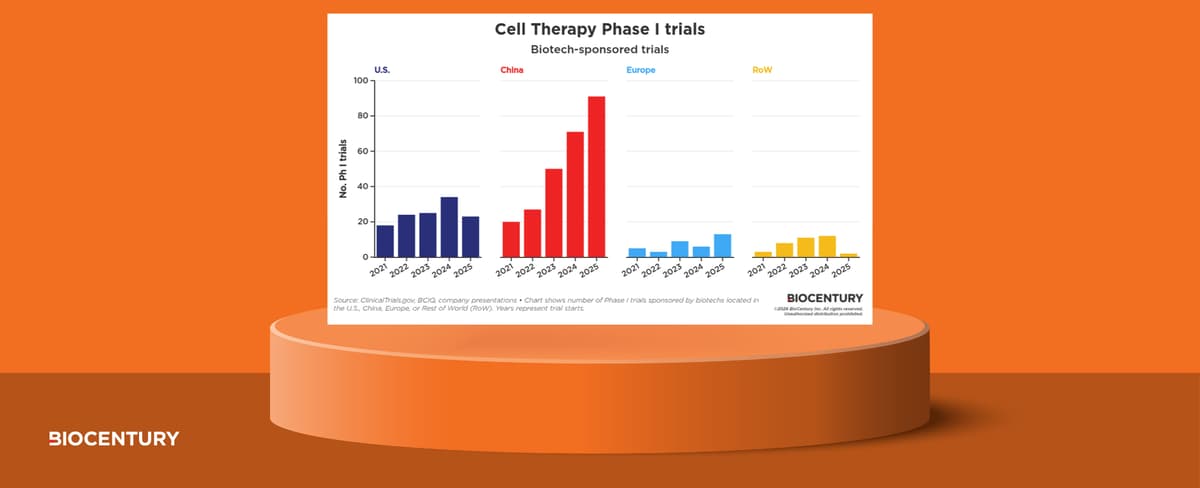
Cell Therapy Phase I Activity Accelerates on China Surge
BioCentury’s website now features a detailed Privacy Preference Center that categorizes cookies into strictly necessary, functional, marketing, advertising, and analytics groups. Users can toggle consent for each category, though disabling essential cookies may impair login and navigation. The policy clarifies...
Meta-Analysis Flawed by Mixing Disparate Anti‑amyloid Agents
A harsh critique of the recent @cochranecollab meta-analysis of the anti-amyloid drugs for Alzheimer's disease https://t.co/EU4JMOVWkV 'These results and their overarching conclusion that “successful removal of amyloid from the brain does not seem to be associated with clinically meaningful effects in...

These 'Good' Viruses Hold up a Booming Industry—AI Just Found a Faster Way to Track Them
Researchers at North Carolina State University combined electrochemical impedance spectroscopy with machine‑learning models to quantify viral vectors, eliminating the need for costly ELISA tagging. Six AI models accurately measured virus titers across five orders of magnitude, even with pH‑induced noise....
![[Comment] Alzheimer's Disease Immunotherapy and the Amyloid Hypothesis: When Aggregation Obscures Interpretation](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/614bb5987569a37ee2c14e1338f7df10.webp)
[Comment] Alzheimer's Disease Immunotherapy and the Amyloid Hypothesis: When Aggregation Obscures Interpretation
A Cochrane review released on April 16, 2026 pooled data from 17 randomized trials of amyloid‑beta‑targeting monoclonal antibodies, encompassing more than 20,000 participants with mild cognitive impairment or mild dementia due to Alzheimer’s disease. The analysis found little to no...

This Nasal Spray Rewinds the Aging Brain, Restoring Memory and Reversing Inflammation in Preclinical Models
Researchers at Texas A&M have created an intranasal spray containing extracellular vesicles derived from human induced pluripotent stem cell‑derived neural stem cells. In 18‑month‑old mice, equivalent to 60‑year‑old humans, two doses dramatically reduced hippocampal inflammation, restored mitochondrial function in microglia,...

Brad Stanfield Rapamycin Trials
Brad Stanfield’s recent clinical study found that participants receiving a placebo performed better than those given rapamycin, a drug touted for its anti‑aging potential. The unexpected outcome was reported within hours of the trial’s completion, prompting immediate scrutiny from the...
Designing Light-Controlled Chemistry with Custom Protein Pairs
Researchers at the University of California, Berkeley have engineered custom protein pairs that change conformation when illuminated with distinct wavelengths of light. By linking these light‑responsive proteins to catalytic domains, the team demonstrated precise, on‑demand activation of chemical reactions inside...

Metformin Fails to Boost Walking Distance in PAD Patients
Metformin to Improve Walking Performance in Lower Extremity Peripheral Artery Disease: The PERMET Randomized Clinical Trial "Among people with PAD without diabetes, metformin did not improve 6-minute walk distance at 6-month follow-up compared with placebo. These results do not support metformin...

Scientists Convert pMMR Tumors to Immunotherapy‑Sensitive dMMR
For anyone else fried with #AACR26 abstracts, we can now look at them from a different lens...

Sweet Protein: Pentasweet Breaks Ground on $76m Precision Fermentation Facility for Brazzein
Lithuanian biotech startup Pentasweet has broken ground on a €65 million ($76 million) precision‑fermentation facility in Vilnius that will produce commercial quantities of brazzein, a natural sweet protein up to 2,000 times sweeter than sugar. Phase I will establish core production capacity by early 2027,...
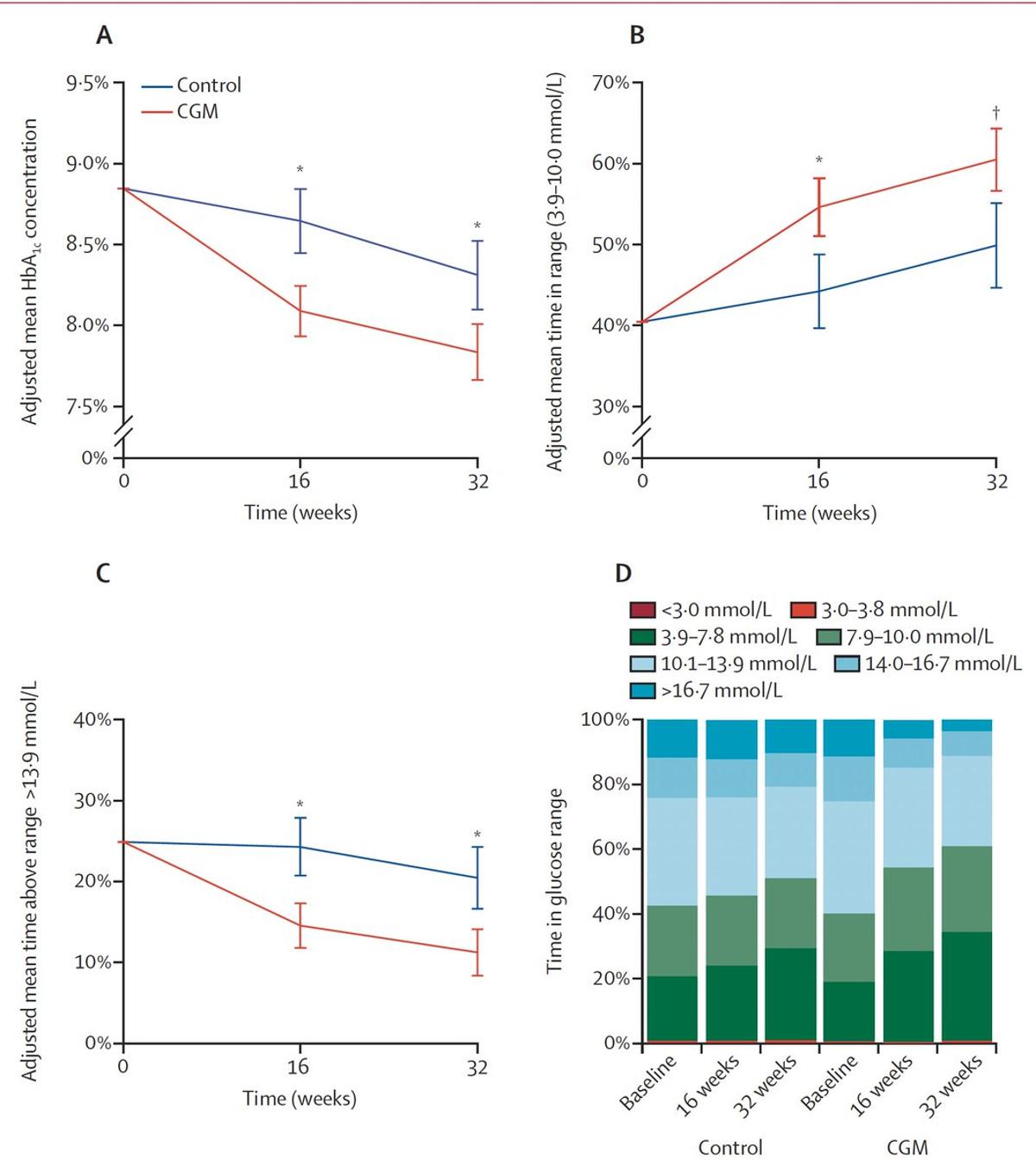
Continuous Glucose Monitoring Outperforms Self‑Monitoring in Type 2 Diabetes
A randomized trial of continuous glucose monitoring (CGM) vs self-monitoring for Type 2 diabetes on basal insulin and drug therapies shows superiority of CGM for gluocse regulation https://t.co/XUymGiov1c @TheLancetEndo https://t.co/lcjFDnEG6x

Alleged FDA Pressure Removes Study Confirming Long‑term Vaccine Efficacy
Vinay Prasad forced FDA scientists to withdraw a paper from Vaccine, the journal I am co-EIC of, because it showed that COVID vaccines work for everyone 6 months+. This is Gold Standard Science at Marty Makary’s FDA. Setting the Gold...
Increased Tumor Stiffness Accelerates Cancer Progression
Recent studies from Lund University demonstrate that increased stiffness of the tumor extracellular matrix directly drives cancer cell invasion through a β1‑integrin‑FAK‑Piezo1 mechanotransduction cascade. Using tunable 3D hydrogels, researchers showed that softening the matrix can reverse the invasive phenotype, but...

CAR‑T Turnaround: Kelonia’s LLY Deal Signals New Pharma Play
A VERY SPIRITED interview w/ Venrock VC Bryan Roberts re: Kelonia Therapeutics and its $LLY deal on this week's Readout LOUD podcast. A CAR-T biotech’s dramatic turnaround, and drugmakers’ tactics to drive more scripts https://t.co/600DsE1cBm via @statnews

Scalable Hypothesis‑testing Datasets Will Drive Biology’s AI ROI
Which AI-enabling datasets will bring the biggest ROI in biology? One type will be "ways to confirm hypotheses at scale". @GordianBio is building atlases of how every expressed gene target affects organs suffering diseases of aging.

FLAG-Based Regimen Yields Robust Results in Acute Myeloid Leukemia Subtype
A Phase 2 trial at MD Anderson evaluated FLAG‑based chemotherapy combined with either gemtuzumab ozogamicin (FLAG‑GO) or idarubicin (FLAG‑IDA) in 219 newly diagnosed core‑binding factor AML patients. The FLAG‑GO arm delivered an 80% five‑year overall survival rate and a 67% relapse‑free survival...

NIH Director Censors Routine Vaccine Study Over Personal Dislike
To be clear: @NIHDirector_Jay reportedly took issue with the study design, which is routine for evaluating vaccines post-market. Podcast Jay, who has no expertise in vaccine trials, decided unilaterally he didn’t like it & thus censored results of studies our...

Bioethicist Vs. Germline Editing Entrepreneur Sparks Uneven Debate
Starting right now. Debate between I. Glenn Cohen (Harvard/reprogenetic bioethics) vs. Cathy Tie, 'serial' human germline editing startup entrepreneur. This should not be an fair fight, but Glenn Cohen is a nice guy...so https://t.co/qfqivoLvhV

ESCMID Global 2026: Zelicapavir Demonstrates Benefits for High-Risk Adults with RSV
At ESCMID Global 2026, Enanta Pharmaceuticals presented Phase IIb data for zelicapavir, an oral once‑daily N‑protein inhibitor, in high‑risk adults with RSV. The double‑blind study of 186 participants missed the primary endpoint but demonstrated faster symptom resolution—up to 7 days quicker in...

New Scoring Tool Reveals How Radiation Reprograms the Pancreatic Tumor Microenvironment
A novel scoring system developed by researchers quantifies how ionizing radiation reshapes the pancreatic tumor microenvironment. By integrating spatial transcriptomics, collagen imaging, and immune cell profiling, the tool identifies three distinct radiation‑induced phenotypes that correlate with patient survival and response...

AI in Oncology Takes Off, Tackling HIV and Liver Disease, Pharma’s Recent Gains
The episode recaps highlights from the AACR conference, emphasizing that AI is moving from pre‑clinical research into clinical oncology, but adoption is hampered by low digital pathology uptake (under 10% of U.S. labs), trust issues, and infrastructure costs. It then...

Regeneron Will Offer Rare Disease Therapy Free As Part Of TrumpRx Deal
Regeneron announced it will provide its newly approved gene therapy for a rare pediatric hearing loss at no cost to U.S. patients under the TrumpRx program. The therapy, cleared by the FDA earlier this year, treats an X‑linked condition affecting...

Specialty Pharmacy's Critical Role as Advanced Therapies Move Into the Community: Fran Gregory, PharmD, MBA
Advanced therapies are set to expand dramatically, rising from 61 approved products today to roughly 180 by 2030. The growth includes cell and gene therapies, bispecific antibodies, and next‑generation monoclonal antibodies, many targeting high‑volume diseases such as cardiovascular and metabolic...
First Bispecific-ADC Combo Shows Strong Results in R/R Mantle Cell Lymphoma
A phase‑2 study published in Blood shows that combining the bispecific antibody mosunetuzumab with the antibody‑drug conjugate polatuzumab vedotin yields an 88.1% overall response rate in relapsed or refractory mantle‑cell lymphoma (MCL). The regimen produced a 78.6% complete response rate,...
AI Model Designs New Antibiotic for Staph Infections After Exploring 46 Billion Compounds
Researchers at McMaster University unveiled SyntheMol‑RL, a generative AI model that explored up to 46 billion virtual compounds and designed a novel, water‑soluble antibiotic named synthecin. The AI‑crafted drug demonstrated strong efficacy against drug‑resistant Staphylococcus aureus in mouse wound models when...

Pan-RAS Inhibitor ADCs Emerge at AACR 2026
At the 2026 American Association for Cancer Research meeting, four biotech companies unveiled antibody‑drug conjugates that deliver pan‑RAS inhibitors directly to tumor cells. Pan‑RAS inhibition has recently delivered unprecedented survival improvements in pancreatic and other RAS‑mutant cancers, but its clinical...

Digital Health Technologies (DHTs) for Drug Development
The FDA has issued a Request for Information and Comments on the use of Digital Health Technologies (DHTs) in drug and biologic development, with a comment deadline of June 1, 2026. Under PDUFA VII, the agency will publish a DHT framework, establish a...
The FDA Gives the Green Light to the First Gene Therapy for Deafness
The FDA has approved the first gene therapy designed to restore hearing in patients born with the rare OTOF‑related form of deafness. Developed by Regeneron, the treatment delivers a functional OTOF gene via adeno‑associated virus directly into the inner ear....

Pharma Meets AI Conference 2026: Key Barriers to Scaling AI in Drug Development
At the Pharma Meets AI conference in Barcelona, industry leaders warned that trust and governance are the biggest obstacles to scaling artificial intelligence in drug development. While AI is delivering gains in prediction, personalization and productivity, concerns over data quality,...

Grace Therapeutics Hits FDA Roadblock on GTx-104, Plans Resubmission
Grace Therapeutics announced that the FDA issued a Complete Response Letter for its GTx-104 NDA, flagging chemistry, manufacturing, and non‑clinical concerns. The company will address the items and seek a Type A meeting before resubmitting, keeping hopes alive for the first...

SELUTION DeNovo: Sirolimus DCB Safe and Effective in ACS Patients
A subanalysis of the SELUTION DeNovo trial presented at SCAI 2026 shows that the sirolimus‑eluting Selution drug‑coated balloon (DCB) is non‑inferior to drug‑eluting stents (DES) in acute coronary syndrome (ACS) patients, with 1‑year target vessel failure (TVF) rates of 5.3%...

Invest in Affordable At‑Home Diagnostics for Urgent Health Needs
@AnywhereDx is hosting an in person event near Boston on Monday to talk about investing in a future that helps consumers tackle some of their most urgent common health problems inexpensively and accessibly through at-home diagnostics See link https://t.co/7SJlx9vBvJ https://t.co/xVrBWUb8nW

E. Coli Editing Technique Expands Into a Universal Toolkit for Rewriting Bacterial DNA
Scientists at Gladstone Institutes have expanded their retron‑based genome editing platform, originally limited to E. coli, to work in 15 phylogenetically diverse bacterial species. The study, published in Nature Biotechnology, introduced ten engineered retron variants—dubbed recombitrons—that achieved editing efficiencies ranging from...