
MediciNova Completes Phase 2 Trial of MN-001 for Diabetic NAFLD
MediciNova Inc. announced the last‑patient, last‑visit milestone for its Phase 2 MN-001‑NATG‑202 trial, evaluating tipelukast in hypertriglyceridemia and non‑alcoholic fatty liver disease (NAFLD) linked to type‑2 diabetes. The trial’s completion clears the path for data analysis and potential Phase 3 planning, underscoring a new therapeutic avenue for a high‑prevalence metabolic condition.

Are We Ready for the Next Wave of Proximity Degraders?
Molecular glue degraders are emerging as the most advanced induced‑proximity modality, allowing tiny molecules to tether disease‑causing proteins to E3 ubiquitin ligases for rapid proteasomal destruction. Industry giants have poured billions into the space, with deals ranging from AbbVie’s $1.64 billion...

10 Obesity Drug Companies to Watch in 2026
Obesity drug development is diversifying beyond injectable GLP‑1s, with companies pursuing oral pills, dual/triple agonists and amylin‑based therapies. Major players such as Eli Lilly and Novo Nordisk are expanding pipelines—Lilly’s retatrutide delivered up to 28% weight loss in Phase III, while Novo’s oral...

FDA Removes Boxed Warning About Risk of Leg and Foot Amputations for the Diabetes Medicine Canagliflozin (Invokana, Invokamet, Invokamet XR)
The FDA has eliminated the boxed warning on canagliflozin (Invokana, Invokamet, Invokamet XR) that highlighted a heightened risk of leg and foot amputations. The decision follows new data from three clinical trials showing the amputation risk is lower than originally...
Injectable Peptides – The New Snake Oil
Steven Novella warns that injectable peptides are the latest wave of snake‑oil products, exploiting loopholes created by the 1994 Dietary Supplement Health and Education Act. While legitimate peptide drugs like insulin and GLP‑1 agonists exist, many unapproved compounds are marketed...
Popular Anti-Aging Drug Combo Caused Severe Brain Damage in Mice
Researchers at the University of Connecticut discovered that the popular anti‑aging drug combo dasatinib plus quercetin (D+Q) causes severe myelin damage in mice, affecting both young and old subjects. The study, published in PNAS, showed dramatic loss of the protective...

First Universal Protein Model Promises New Therapies
Today Biohub is sharing a major scientific advance: our first world model for protein biology. This system can help scientists discover and design proteins that will hopefully lead to new medical therapies.

Makary’s Reforms Will Live on at FDA Even as Leadership Turns Over
Former FDA Commissioner Marty Makary’s reforms, especially the Commissioner’s National Priority Voucher (CNPV) program, will continue unchanged despite his resignation and a broader leadership vacuum at the agency. The CNPV, launched in July 2025, grants accelerated review to drugs that...

D&D Pharmatech Reports P-II Trial Results on Zabopegdutide for Metabolic Dysfunction-Associated Steatohepatitis (MASH)
D&D Pharmatech disclosed topline Phase II data for its GLP‑1‑like agent zabopegdutide in 67 overweight or obese patients with metabolic dysfunction‑associated steatohepatitis (MASH). After a 2‑week titration to a 40 mg weekly maintenance dose, 48‑week histology showed a 50% fibrosis‑stage improvement versus...

Junshi Biosciences Achieves Phase 3 Success with Toripalimab Combo in NSCLC
Junshi Biosciences announced that its phase 3 NEOTORCH trial met primary endpoints, showing toripalimab plus chemotherapy reduced recurrence risk by 60% in resectable stage II‑III non‑small cell lung cancer. The data pave the way for a supplemental new drug application...
Precision BioSciences Reports First cccDNA Elimination in Hepatitis B Patients
Precision BioSciences announced that its ELIMINATE-B study demonstrated the first clinical evidence of complete cccDNA elimination in hepatitis B patients after two doses of PBGENE-HBV, achieving a 10‑fold reduction in viral transcripts. The result could reshape curative strategies for chronic...

Cartesian Therapeutics Secures $150 Million Credit Facility to Push Autoimmune Cell Therapy
Cartesian Therapeutics announced a $150 million credit facility with K2 HealthVentures, including an initial $50 million tranche that will fund operations through 2028. The financing backs the upcoming Phase 3 AURORA trial readout for Descartes‑08, its lead cell‑based therapy for myasthenia gravis and...

Jade Biosciences Doses First Patient in JADE‑101 Phase 2 Trial for IgA Nephropathy
Jade Biosciences announced the first dose of JADE‑101 in its JUNIPER Phase 2 trial for immunoglobulin A nephropathy, enrolling about 30 patients. The monoclonal antibody targets APRIL, a driver of pathogenic IgA, and interim results are slated for 2027.

This Medication Was Linked To A 56% Lower Risk Of Breast Cancer Recurrence
A new observational study of 841,000 U.S. breast‑cancer patients found that GLP‑1 drugs such as semaglutide and tirzepatide were linked to markedly lower mortality and recurrence rates in women with obesity or type 2 diabetes. In obese survivors, GLP‑1 use cut...

First Participant Dosed in Jade’s Phase II JUNIPER Trial for IgAN
Jade Biosciences has dosed the first participant in its Phase II JUNIPER trial evaluating JADE101 for immunoglobulin A nephropathy (IgAN). The open‑label study will enroll 30 patients and assess safety, tolerability, and early efficacy signals such as protein‑to‑creatinine ratio, eGFR and haematuria...

TOBY Secures US FDA Breakthrough Device Designation for Urine-Based Alzheimer’s Disease Test
The U.S. Food and Drug Administration has granted Breakthrough Device Designation to TOBY’s urine‑based test for Alzheimer’s disease, targeting adults aged 50 and older with cognitive impairment. The test analyzes volatile organic compound patterns in urine using mass spectrometry and...

Mapping Cellular Stress Biology to Tackle Undruggable Targets
In this episode, Daniel Levine interviews Yeremiá Gizarian, co‑founder and CEO of Soleil Therapeutics, about the company’s AI‑driven platform that maps cellular stress responses to discover drugs for traditionally "undruggable" targets. The discussion explains how Soleil flips the conventional drug‑discovery...

Sobi Highlights P-III (CORE & CORE2) Trials Results of Olezarsen for Severe Hypertriglyceridemia (sHTG) at EAS 2026
Sobi presented pooled Phase III data from its CORE and CORE2 trials, evaluating the antisense drug olezarsen (Tryngolza) in 455 patients with severe hypertriglyceridemia (TG ≥ 500 mg/dL). At six months, the 80 mg dose cut triglycerides by 66% and the 50 mg dose by 59%...
Scientists Create Supercharged Vitamin K that Helps the Brain Heal Itself
Researchers at Japan's Shibaura Institute of Technology have engineered a novel vitamin K analogue that boosts neuronal differentiation threefold compared with natural MK‑4. By hybridizing vitamin K with retinoic‑acid motifs and a methyl‑ester side chain, the compound—dubbed Novel VK—demonstrates stronger...
Matt Kaeberlein's New Longevity Science Podcast / Youtube Channel (May, 2026)
Dr. Matt Kaeberlein’s Longevity Science podcast provides a biochemistry‑focused audit of the burgeoning peptide market, clarifying that true peptides are short amino‑acid chains and excluding compounds like NAD+ and rapamycin. He evaluates leading peptides—synthetic mitochondrial agent Elamipretide and the popular...

One-Time Gene Editing Cuts LDL Cholesterol in Early Hypercholesterolemia Trial
A single intravenous infusion of VERVE‑102, an in‑vivo base‑editing therapy targeting the PCSK9 gene, produced dose‑dependent reductions in PCSK9 protein and LDL cholesterol in a phase 1 trial of 35 adults with heterozygous familial hypercholesterolemia or premature coronary disease. At the...

Retatrutide Reshapes Metabolism in Obesity and Type 2 Diabetes, Study Finds
A post‑hoc analysis of two phase‑2 trials shows that the triple‑receptor agonist retatrutide reshapes fatty‑acid oxidation and insulin‑resistance biomarkers in participants with obesity, with or without type 2 diabetes. Higher doses raised 3‑hydroxybutyrate by up to 198% and altered acyl‑carnitine ratios,...

Recce Pharmaceuticals Signs Distribution Agreement for R327 Diabetic Foot Gel Across MENA
Recce Pharmaceuticals has inked a 10‑year licensing deal with a leading Middle Eastern partner to sell its R327 topical gel for diabetic foot infections across the MENA region, including Saudi Arabia, Egypt, Algeria, Morocco and the GCC. The agreement provides...
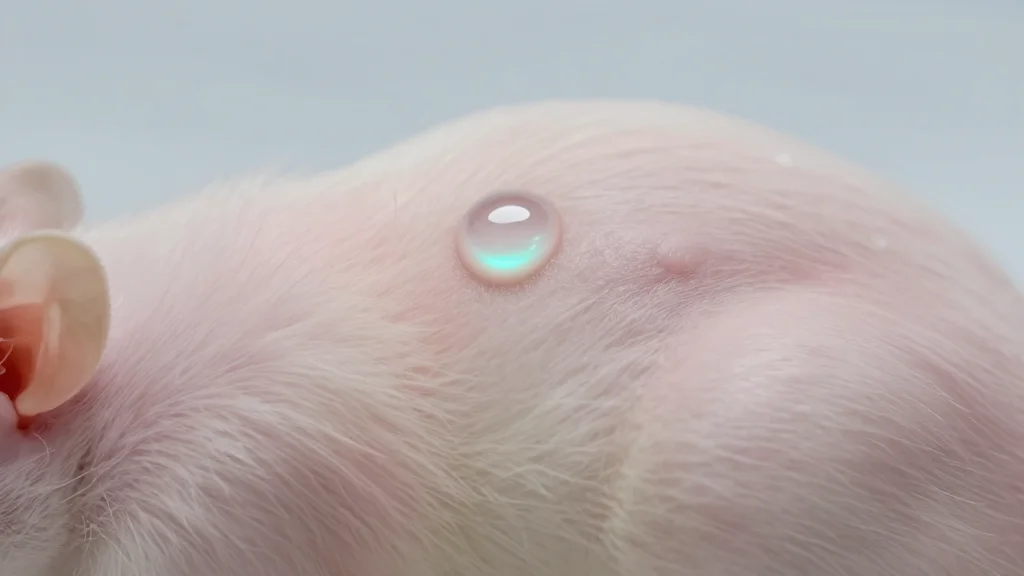
Topical Senolytic Navitoclax Clears Zombie Cells, Boosts Wound Healing in Aged Mice
Researchers led by Dr. Daniel S. Roh at Boston University applied a low‑dose topical formulation of the senolytic drug navitoclax to the skin of 24‑month‑old mice. The treatment eliminated up to 15% of senescent cells, triggered a brief inflammatory response,...

Mediwound Ltd (MDWD) Q1 2026 Earnings Call Transcript
MedWound reported Q1 2026 revenue of $1.9 million, a sharp decline from $5.8 million a year earlier, driven primarily by a U.S. government shutdown that delayed contract awards. R&D spending jumped to $4.5 million as the company advanced the Phase III VALUE trial for...

FDA’s Concerns with Unapproved GLP-1 Drugs Used for Weight Loss
The FDA warned that unapproved compounded versions of GLP‑1 receptor agonists such as semaglutide and tirzepatide are being marketed for weight loss, posing safety and quality risks. Compounded products are not reviewed for efficacy, may be stored improperly, and have...

Kailera Reports First Quarter 2026 Financial Results and Provides Clinical Data Updates
Kailera Therapeutics reported Q1 2026 results, highlighting major clinical progress across its obesity pipeline and a successful $718.8 million IPO. The company launched a global Phase 3 KaiNETIC program for ribupatide injection, reported strong Phase 2 oral ribupatide weight‑loss data, and disclosed positive...

Agios Secures EU Marketing Authorisation for PYRUKYND in Thalassaemia
Agios Pharmaceuticals and partner Avanzanite Bioscience announced that the European Commission granted marketing authorisation for PYRUKYND (mitapivat) to treat adult patients with transfusion‑dependent and non‑transfusion‑dependent alpha‑ or beta‑thalassaemia. The decision marks the drug’s second EU indication, following its 2022 approval...

Pharmaceutical Executive Daily: FDA Approves Hepcludex
Eli Lilly announced three acquisitions valued at roughly $3 billion to broaden its pipeline beyond diabetes and obesity, targeting oncology, immunology and other growth areas. The FDA approved Hepcludex, the first U.S. therapy for chronic hepatitis delta virus infection, offering a new...

An Update on Progress at Repair Biotechnologies, Developing Means to Regress Atherosclerotic Plaque
Repair Biotechnologies announced that its lead candidate, now designated REP-0004, is a liver‑targeted mRNA lipid‑nanoparticle that produces a fusion protein to break down intracellular free cholesterol. The approach triggers a rapid, systemic pull of cholesterol from plaques, delivering dramatic regression...

Niowave, Fresh From Novartis Deal, Starts Building $75M Radioisotope Plant
Niowave announced the construction of a new $75 million actinium‑225 production facility in Lansing, Michigan, slated to begin operations in 2028. The plant will house proprietary superconducting linear accelerators and create about 70 high‑skill jobs in research, engineering, and quality assurance....
Self-Assembling Peptide Helps Liver Cancer Drugs Escape Lysosome Traps
Researchers engineered a self‑assembling peptide, RS‑FS, that remains as nanospheres in blood but converts to nanofibers inside the acidic, reducing environment of hepatocellular carcinoma lysosomes, where it damages the organelle and frees trapped drugs. In mouse models, RS‑FS combined with...

Corbus Pharmaceuticals Holdings, Inc. (CRBP) Discusses Clinical Data Update From Phase 1/2 Study of CRB-701 Presented at ASCO Transcript
Corbus Pharmaceuticals announced that it will present Phase 1/2 data for its oncology candidate CRB‑701 at the upcoming American Society of Clinical Oncology (ASCO) meeting. The data snapshot reflects an April 1 cut, and the company briefed investors via a conference call...

Arthritis Drug Tocilizumab Shows Promise for Treatment‑Resistant Depression
Researchers at the University of Bristol reported that tocilizumab, an anti‑inflammatory arthritis medication, helped 54% of participants achieve remission in a four‑week trial for treatment‑resistant depression, versus 31% on placebo. The findings suggest targeting inflammation could expand options for patients...
Compass Therapeutics to Unveil Phase 1 CTX‑8371 Data at ASCO 2026
Compass Therapeutics announced that it will present promising Phase 1 data on its bispecific antibody CTX‑8371 at the American Society of Clinical Oncology’s 2026 annual meeting. The data, derived from patients with advanced malignancies who have progressed after checkpoint‑inhibitor therapy, could...

Adcendo Inks MSD Supply Deal to Run ADC-Keytruda Combination Study
Adcendo, a Copenhagen‑based biotech, has signed a supply agreement with MSD for Keytruda to launch a Phase Ib trial of its tissue‑factor‑directed ADC ADCE‑T02 in combination with the checkpoint inhibitor. The study, slated for the second half of the year, will...

FDA Approves First Interchangeable Biosimilars to Simponi and Simponi Aria; Janssen Seeks a Preliminary Injunction to Block Their Launch
The FDA approved Accord BioPharma’s IMMGOLIS™ and IMMGOLIS INTRI™ as the first interchangeable biosimilars to Janssen’s Simponi® and Simponi ARIA® on May 15, 2026. Both products treat moderately to severely active rheumatoid arthritis, and IMMGOLIS also covers ulcerative colitis. Accord will commercialize the drugs...
New Drug Works Against Diseases Like Measles and Croup
Researchers at Georgia State University have identified GHP-88310, a new oral antiviral candidate that targets orthoparamyxoviruses such as measles and human parainfluenza virus type 3. The drug demonstrated potent, once‑daily efficacy and high tolerability in both rodent and non‑rodent animal models,...
Banner Health Report Shows Arizona Research Driving National Medical Innovation
Banner Health’s $80.5 million research enterprise reported 1,300 active clinical and translational studies in 2025, delivering an FDA‑approved device for severe mitral annular calcification and launching a $74.5 million NIH‑funded Alzheimer prevention trial in Colombia. The organization enrolled more than 89,000 participants...
Lilly Gets Back Into the Vaccine Business With New Deals
Eli Lilly & Co. announced it will acquire three clinical‑stage vaccine developers for up to $3.8 billion, marking a major re‑entry into the infectious‑disease space. The deals are financed largely by cash generated from its blockbuster obesity drugs, such as Mounjaro and...

Brazil Clears First Generic Copy of Novo Nordisk’s Ozempic Shot
Brazil’s health regulator Anvisa approved EMS’s generic semaglutide injection, Ozivy, marking the country’s first GLP‑1 copy of Novo Nordisk’s Ozempic. EMS plans to price Ozivy about 30% lower than the branded shot and expects to launch within 30 days, offering...
Beta Bionics to Launch Its First Insulin Patch Pump to Compete with Insulet
Beta Bionics announced it will debut its first insulin patch pump, called Mint, by the end of Q2 2027 pending FDA clearance. The three‑day wearable features a 200‑unit reservoir and blends reusable and disposable components, linking to the company’s adaptive dosing...

New DNA Assembly Method Bridges Design and Synthesis
A new DNA assembly method closes the gap between generative genome design and lab-scale synthesis. https://spectrum.ieee.org/faster-dna-synthesis-sidewinder?share_id=9503681
Paricalcitol Shows Promise in Metastatic Pancreatic Cancer
A small randomized trial in patients with metastatic pancreatic cancer found benefit for paricalcitol, a Vitamin D analog https://t.co/CDnIwm0xeo Good @salkinstitute explainer https://t.co/QgEKHiquIa https://t.co/m32CFTInJA

STAT+: Pharmalittle: We’re Reading About a Lilly Gene Therapy for Cholesterol, Three New Lilly Deals, and More
Eli Lilly announced that its gene‑editing therapy, acquired from Verve Therapeutics for $1 billion, lowered LDL cholesterol by 62% in a Phase 1 trial. The study reported no treatment‑related serious adverse events, paving the way for a Phase 2 trial. In parallel, Lilly...

Nanobiotix Raises $100 Million in Global Offering, ABIVAX Posts €52 Million Q1 Loss
Nanobiotix (NBTX) completed a global offering that raised $100 million after underwriters exercised their option, while ABIVAX (ABVX) posted a €52 million (≈$56.6 million) loss for Q1 2026 as R&D spending surged. The contrasting results underscore divergent trajectories among Euro‑listed biotech firms.

Biotech Surge Powers Nasdaq: AKTX Jumps 255% on KRAS Data
Akari Therapeutics (AKTX) surged more than 255% for a second straight day after preclinical data showed synergistic activity of its AKTX-101 drug with KRAS inhibition in pancreatic cancer models. The rally lifted the broader biotech sector, with Govix (GOVX) and...

Can a Chaotic FDA Still Deliver on Faster Drug Development?
Recent leadership upheaval at the FDA, including the departure of Commissioner Martin Makary, has sparked concerns about the agency’s ability to maintain momentum on accelerating early‑stage clinical trials in the United States. Nonetheless, industry insiders like Parexel’s Tala Fakhouri argue...

AstraZeneca, Daiichi Sankyo Win EU CHMP Nod for Enhertu in HER2 Tumors
AstraZeneca and Daiichi Sankyo received a positive opinion from the European Medicines Agency's CHMP for Enhertu, expanding the antibody‑drug conjugate to HER2‑positive solid tumours after prior therapy. The endorsement is based on subgroup analyses from Phase II DESTINY trials that showed...
Strep A Vaccine Gets $140mn Backing
Global health philanthropists have launched a $140 million fund, led by Coefficient Giving and donors such as Dustin Moskovitz, to accelerate development of a Group A Streptococcus (Strep A) vaccine. The fund aims to double the pipeline of vaccine candidates and bring at...