
Junshi Biosciences Hits Primary Endpoints in Phase III NEOTORCH Study, Plans sNDA for Toripalimab
Junshi Biosciences announced that its NEOTORCH Phase III trial met its primary event‑free survival and major pathological response endpoints in 501 patients with resectable stage II‑III non‑small cell lung cancer. The company will file a supplemental new drug application to broaden toripalimab’s perioperative indication, a move that could reshape the Chinese lung‑cancer market.

FDA Expands TREMFYA Label to Cover Psoriatic Arthritis Joint Damage
Johnson & Johnson’s TREMFYA (guselkumab) received FDA approval for a label expansion that now includes inhibition of structural joint damage in adults with active psoriatic arthritis. The decision follows positive results from the Phase 3b APEX trial, positioning TREMFYA as the...

Can AI Crack Pharma’s Drug Repurposing Goldmine?
The FDA has opened a public comment period, ending June 11, to gather ideas on how to accelerate drug repurposing using existing therapies. It aims to identify candidates that already meet evidence standards or show early efficacy, reducing the need for...

Replimune to Resubmit Twice-Rejected Drug for Approval After FDA Shakeup
Replimune announced it will resubmit its melanoma drug to the FDA after two prior rejections under former Commissioner Marty Makary. The agency’s recent leadership turnover has led to an agreement on a path forward, with the FDA promising an urgent...
Targeted Therapy Shows Superior Results Over Chemotherapy in Treating Difficult Lung Cancer, ASCO Reports
At the American Society of Clinical Oncology (ASCO) meeting, researchers presented data showing a novel targeted therapy outperformed standard chemotherapy in patients with advanced non‑small cell lung cancer (NSCLC) that are difficult to treat. The trial reported a median progression‑free...

GSK Functionally Cures Hepatitis B, Lilly's $3.8B Vaccine Pivot, and Base Editing Holds for 18 Months – This Week in...
GSK and Ionis reported Phase 3 data showing bepirovirsen achieved roughly a 20% functional cure rate in chronic hepatitis B, a potential paradigm shift for a disease affecting over 250 million people. Eli Lilly announced up to $3.8 billion in vaccine acquisitions targeting shingles,...

Can WEE1 Inhibitors Finally Make Replication Stress Druggable?
Targeting replication stress through WEE1 inhibition is re‑emerging after early setbacks. First‑generation inhibitor adavosertib demonstrated tumor responses in ovarian and uterine cancers but was limited by dose‑limiting toxicity. Newer agents such as APR‑1051, azenosertib and zedoresertib are being tested in...
New Study Uncovers How Bacteria Seize a Rare Sugar Molecule
A multidisciplinary team has identified a previously unknown bacterial transporter that snatches a rare sugar molecule, N‑acetyl‑neuraminic‑acid analog, from host environments. Cryo‑EM imaging revealed the transporter’s architecture at 2.8 Å, pinpointing the binding pocket and key amino‑acid residues. Functional assays showed...

Merck Secures the US FDA Breakthrough Therapy Designation for Calderasib to Treat Newly Diagnosed Metastatic KRAS G12C-Mutant NSCLC
The U.S. FDA granted Merck’s KRAS‑G12C inhibitor calderasib (MK‑1084) breakthrough therapy designation for first‑line treatment of newly diagnosed metastatic non‑small cell lung cancer (NSCLC) that harbors a KRAS G12C mutation and expresses PD‑L1 (TPS ≥ 1%). The designation is backed by Phase I...

Tomato Gene Discovery Opens Door to Heat-Tolerant Varieties
Researchers at Japan's University of Tsukuba identified the SlIAA9 gene as a key brake on heat tolerance during tomato seed germination. Loss‑of‑function mutants lacking SlIAA9 germinated at near‑normal rates even under prolonged high‑temperature exposure and produced vigorous seedlings. Molecular analysis...

Cingulate Therapeutics Awaits FDA Verdict on Once‑Daily ADHD Drug CTx‑1301
Cingulate Inc. (CING) is set to receive an FDA ruling on May 31, 2026 for its ADHD candidate CTx‑1301, a once‑daily dexmethylphenidate product built on the company’s Precision Timed Release platform. The decision could reshape the stimulant market and test...
WHO Prioritizes Regeneron's Maftivimab for Bundibugyo Ebola Trials Amid Growing Outbreak
Regeneron Pharmaceuticals' maftivimab antibody has been recommended by the WHO Therapeutics Advisory Group for prioritized evaluation in clinical trials targeting the Bundibugyo Ebola outbreak in the Democratic Republic of the Congo and Uganda, where 906 suspected cases and 223 deaths...
GSK’s Nucala Secures NICE COPD Endorsement
NICE has added GSK’s Nucala (mepolizumab) to its list of approved biologic add‑on treatments for chronic obstructive pulmonary disease (COPD) in patients with elevated eosinophils. The endorsement is based on three Phase III trials that demonstrated up to a 21 % reduction...
MIT Nanotech Catheter Spots Bladder‑cancer Biomarker 50,000× More Sensitively
MIT engineers have created a catheter lined with carbon‑nanotube sensors that can detect the bladder‑cancer biomarker NMP‑22 up to 50,000 times more sensitively than conventional urinalysis. The breakthrough, detailed in a Nature Nanotechnology paper, could enable clinicians to locate tumors...
CVS Caremark Restores Zepbound and Adds Lilly's Foundayo, Expanding Coverage to 30 Million Americans
CVS Caremark announced it will reinstate coverage of Eli Lilly’s injection Zepbound on Oct. 1 and add the newly approved oral pill Foundayo on June 1, affecting roughly 25‑30 million commercial lives. The reversal follows a 2023 formulary drop that sparked patient backlash and...

Regulatory and Scientific Frontiers in Drug Repurposing: Accelerating Therapeutic Innovations for Unmet Medical Needs + Examples of Repurposed Drugs
On May 11, 2026 the FDA unveiled a formal drug‑repurposing program aimed at accelerating clinical trials for approved compounds targeting chronic, rare and underserved conditions. The initiative builds on prior legislation such as the Best Pharmaceuticals for Children Act and...
Pancreatic Cancer Halted by Virus Injection in Three Patients
In a U.S. Phase 1 safety trial, an engineered oncolytic virus halted tumor growth and prevented spread in three pancreatic cancer patients. Researchers administered only one‑tenth of the intended therapeutic dose, yet observed clear disease control. Lead developer Masato Yamamoto highlighted...

Kalohexis Doses First Patients in 710GO Phase I Trial for Obesity
Kalohexis has begun dosing the first participants in a Phase I, first‑in‑human trial of 710GO, an oral dual melanocortin‑3/4 receptor agonist aimed at treating general obesity. The randomized, double‑blind, placebo‑controlled study in Australia will enroll roughly 100 obese or overweight volunteers...

Samsung Bioepis Reports the EC Launch of Opuviz (Biosimilar, Eylea)
Samsung Bioepis announced the European commercial launch of Opuviz 40 mg/mL solution for injection, a biosimilar to aflibercept (Eylea). The product will be sold directly by the company across the EU for wet age‑related macular degeneration, diabetic macular oedema, retinal vein...

World MS Day Special: Immunic Reveals New Hope for Progressive MS
Immunic Therapeutics CEO Dr. Daniel Vitt used World MS Day to spotlight the company’s lead candidate, IMU‑838, an oral therapy for progressive multiple sclerosis. He detailed his personal biotech journey, explained how IMU‑838 differs from injectable disease‑modifying drugs, and presented...
Procept Secures FDA IDE for Aquablation and Completes WATER IV Study Enrolment
Procept BioRobotics secured a second FDA investigational device exemption (IDE) to launch a new randomized trial of its Aquablation water‑jet therapy, this time comparing it with active surveillance in men with low‑grade prostate cancer. The earlier WATER IV study has already...

AstraZeneca Receives the US FDA Approval for Imfinzi Regimen to Treat High-Risk Non-Muscle-Invasive Bladder Cancer (NMIBC)
AstraZeneca’s Imfinzi (durvalumab) combined with Bacillus Calmette‑Guérin (BCG) therapy received U.S. FDA approval for treating adults with BCG‑naïve, high‑risk non‑muscle‑invasive bladder cancer (NMIBC). The decision rests on the phase‑III POTOMAC trial, which enrolled 1,018 patients post‑TURBT and compared Imfinzi + BCG induction...

NASA Starts New Project Growing Stem Cells Aboard ISS
NASA’s InSPA‑StemCellEX‑H2 project on the International Space Station is testing large‑scale production of blood stem cells in microgravity. Researchers say the weightless environment preserves cell quality and accelerates expansion, potentially reducing immune rejection when transplanted. The initiative aims to generate...
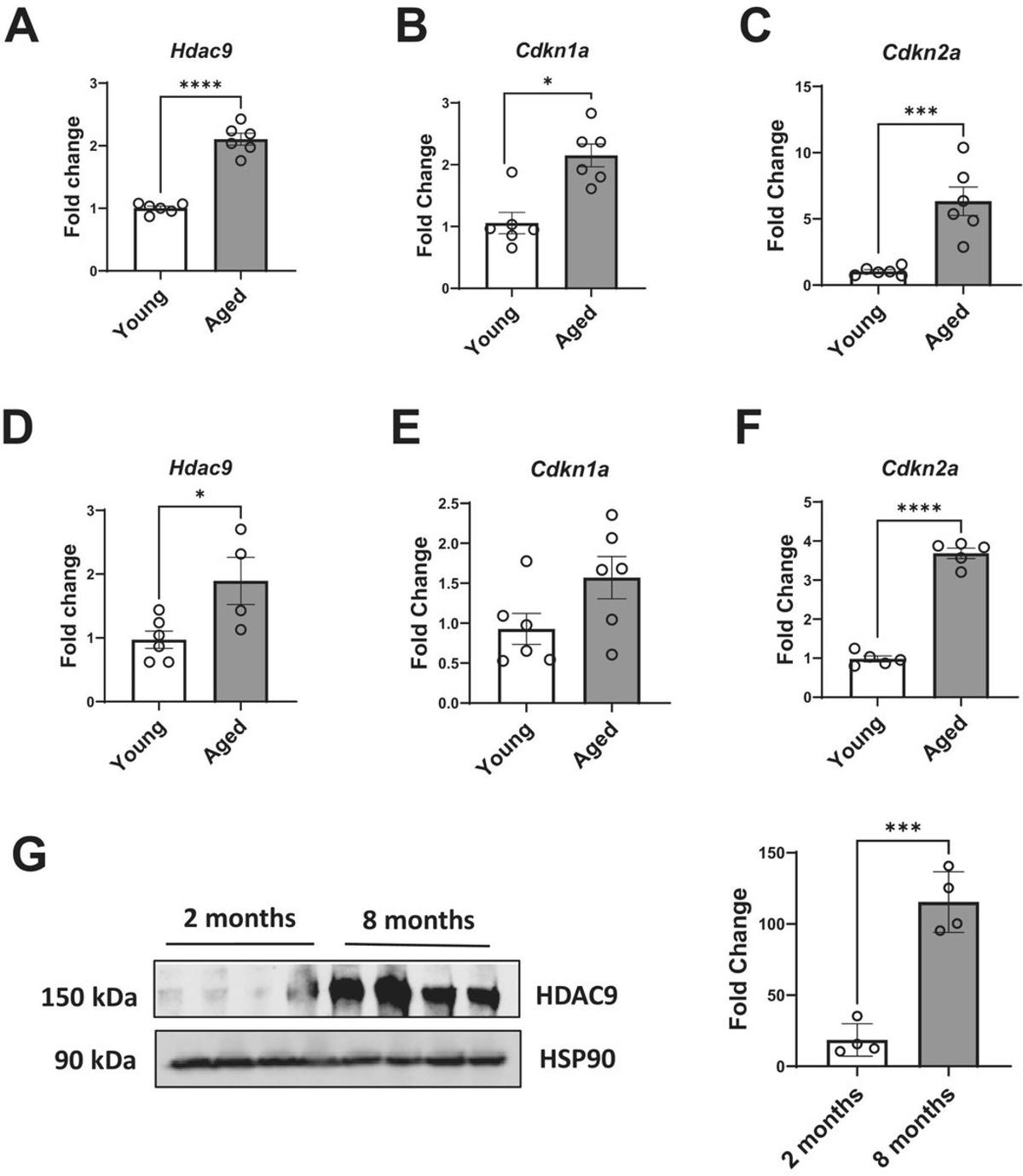
HDAC9 Deletion Reverses Age‑Related Fat Cell Decline
Histone Deacetylase 9 Gene Deletion Ameliorates Aging-Related Adipose Tissue Senescence and Mitochondrial Dysfunction in Mice ...suggesting that targeting HDAC9 may be a promising strategy to maintain healthy adipose tissue during aging." https://t.co/pi1aXAEoeB https://t.co/IB1yUwCaGC
Human Organoids Reveal How to Reverse “Irreversible” Nerve Damage
Scientists at the University of Cambridge have engineered miniature human brain‑spinal cord organoids that form functional neural circuits capable of triggering muscle contractions. By culturing these linked organoids for over a year, they identified a developmental cutoff—around day 150, equivalent to...

Swedish Trial Shows Single Psilocybin Dose Cuts Depression in 48 Hours
Researchers at Karolinska Institutet and the Northern Stockholm Psychiatric Clinic reported that a single 25 mg oral dose of psilocybin reduced depressive symptoms within 48 hours in a randomized, double‑blind trial of 35 patients. The rapid improvement, measured by a 7.27‑point...

Nitrogen-Fixing Genes Moved Into New Bacterial Strains, Opening Path Beyond Fertilizer
Researchers at Washington State University have successfully moved a large cluster of nitrogen‑fixation genes, known as a symbiosis island, from rhizobia into previously non‑fixing bacterial strains. Using a novel genetic tool, they achieved high‑efficiency mating and created bacteria that can...

A Better Algorithm for Predicting How Cells Behave
Altos Labs researchers unveiled PRiMeFlow, a flow‑based machine‑learning model that predicts how a cell’s gene‑expression profile will change after genetic or chemical perturbations. Unlike prior methods, PRiMeFlow operates directly in the high‑dimensional expression space instead of compressing data, and it...
UT Southwestern Identifies HELZ2 Liver Switch That Halts Cholesterol Production
UT Southwestern scientists have pinpointed HELZ2, a liver protein that destroys the messenger RNA for apoB, effectively stopping cholesterol synthesis before particles form. The discovery, validated in mouse models, could spawn a new class of cardiovascular drugs and reshape biohacking...

Nanotube-Coated Catheter Could Detect Bladder Cancer Biomarker 50,000 Times More Sensitively
MIT researchers have created a catheter coated with carbon‑nanotube nanosensors that can detect the bladder‑cancer biomarker NMP‑22 up to 50,000 times more sensitively than standard urinalysis. In animal models the sensor produced fluorescent chemical images that pinpointed tumors as small...

Target for Aggressive Prostate Cancer Prevention Identified in Mice
Columbia University researchers identified SIRT1 as a driver of neuroendocrine prostate cancer (NEPC) in mice. Using a Sleeping Beauty forward genetic screen, they pinpointed SIRT1 among 75 candidate genes and showed that silencing or pharmacologically inhibiting it dramatically reduced tumor...

STAT+: Biotech Veteran Jeremy Levin on Why the Industry’s Future Is Secure, but American Leadership Is at Risk
Jeremy Levin, founder of Ovid Therapeutics and former Teva CEO, argues that while biotech science is advancing rapidly, the sector’s future is jeopardized by eroding regulatory, investment, and public‑trust institutions. In his new book he highlights political upheaval and short‑term...

Electrical Pulses Reverse Aging in Sea Squirts, Offering Clues for Extending Human Longevity
Scientists at Stanford have shown that brief electrical pulses can reverse aging markers in sea squirts, extending their laboratory lifespan from months to several years. The 15‑minute treatment triggers a rapid shutdown and rebound of gene expression, effectively rebooting stem‑cell...

AAHI’s SLA-SE Adjuvant Technology Powers Lilly’s Acquisition of Curevo’s Next-Generation Shingles Vaccine
Eli Lilly has agreed to acquire Curevo Vaccine, securing its Phase‑3‑ready shingles shot CRV‑101, which incorporates Access to Advanced Health Institute’s (AAHI) proprietary SLA‑SE adjuvant. The adjuvant is designed to boost T‑cell immunity while maintaining a favorable safety profile, positioning the...
Long-Term Real-World Economic and Clinical Outcomes of TRIKAFTA® (Elexacaftor/Tezacaftor/Ivacaftor and Ivacaftor) in Cystic Fibrosis
Vertex Pharmaceuticals released a real‑world analysis of TRIKAFTA® (elexacaftor/tezacaftor/ivacaftor and ivacaftor) showing sustained clinical benefits for cystic fibrosis patients over five years. The study documented a 45% drop in annual hospitalizations, an average 8.2‑point rise in FEV1, and a net...

Floral-Scented Fungus (Engineered by Scientists) Lures Mosquitoes to Their Doom
Scientists at the University of Maryland have engineered a strain of Metarhizium fungus that releases the floral compound longifolene, luring mosquitoes and killing them within days. Laboratory tests showed 90‑100% mortality even in rooms with competing human and flower scents....

FDA Approves Durvalumab in Combination with Bacillus Calmette-Guerin for High-Risk Non-Muscle Invasive Bladder Cancer
The FDA approved durvalumab (Imfinzi) in combination with Bacillus Calmette‑Guerin for BCG‑naïve, high‑risk non‑muscle invasive bladder cancer. The decision follows the POTOMAC trial, which enrolled 1,018 patients and demonstrated a 32% reduction in disease recurrence risk (hazard ratio 0.68, p=0.015)....

Eli Lilly’s Triple‑Play Acquisitions Accelerate Vaccine Portfolio
28May: How expansive is @EliLillyandCo's expansion into infectious disease vaccine development, reflected in its 'triple play' acquisitions of @CurevoVaccine, Vaccine Company, and LimmaTech Biologics? My latest for @GENbio: https://t.co/vMUPpoCgCo

Bring on the Breakthroughs: ASCO Takes Translation to More Patients
The 2026 American Society of Clinical Oncology (ASCO) meeting in Chicago spotlights the theme “The Science and Practice of Translation,” showcasing data that move laboratory breakthroughs into everyday cancer care. Highlighted trials include PROTEUS, a phase‑3 study adding apalutimide to...

Blackstone Commits up to $1.3 Billion to Fund Apogee Therapeutics' Zumilokibart Program
Blackstone Life Sciences announced a financing package of up to $1.3 billion for Apogee Therapeutics, combining $800 million in synthetic royalty financing and $500 million in senior debt. The capital will accelerate development and potential commercialization of Apogee’s lead monoclonal antibody, Zumilokibart, targeting...
MediWound Q1 Revenue Drops 62% but Reaffirms $24‑$26M 2026 Outlook, Shares Rise
MediWound Ltd. posted first‑quarter 2026 revenue of $1.5 million, a 62% drop from a year ago, and a net loss of $3 million. Despite the short‑term hit, the company reaffirmed its full‑year revenue guidance of $24‑$26 million, prompting its shares to climb.

Beren Therapeutics Announces FDA Extension of Review Period for New Drug Application (NDA) for Adrabetadex for the Treatment of Infantile-Onset...
Beren Therapeutics announced that the FDA has extended the review period for its New Drug Application for adrabetadex, a cyclodextrin‑based therapy for infantile‑onset Niemann‑Pick disease type C, by three months. The new PDUFA target action date is November 17, 2026, following a major...
FDA Oncology Roundup: AbbVie’s Rare Cancer Approval, AstraZeneca in Breast Cancer Limbo
The FDA approved AbbVie’s CD123‑targeting antibody‑drug conjugate Decnupaz for blastic plasmacytoid dendritic cell neoplasm (BPDCN), an ultra‑rare aggressive blood cancer, after the CADENZA trial reported a 69.7% composite complete response in newly diagnosed patients and 15.7% in relapsed/refractory cases. The...
Cat Amongst the Pigeons
Merck secured FDA approval for its antibody‑drug conjugate sacituzumab tirutecan (sac‑TMT) in advanced endometrial cancer, marking a rare ADC win in a solid‑tumor indication. The drug targets the Trop‑2 protein and delivers a potent cytotoxic payload, offering a new therapeutic...
FDA’s Advisory Panel Votes in Favor of Updating Covid-19 Vaccine Shots to Target XFG Variant
The FDA’s Vaccines and Related Biological Products Advisory Committee voted to update COVID‑19 vaccine compositions for the 2026‑2027 season to target the XFG variant, which now accounts for more than half of U.S. cases. The recommendation follows a WHO call...

Biohub Open-Source AI Model Targets Protein Design for Drug Discovery
Biohub, part of the Chan Zuckerberg Initiative, launched an open‑source AI system that models protein biology at evolutionary scale to aid early‑stage drug discovery. The platform, described as a “world model,” was used to design protein binders targeting cancer and...

Is Immune Priming the Missing Piece in Oncology? Oncolytics Biotech Thinks So
Oncolytics Biotech is positioning its oncolytic virus pelareorep as an "immune‑priming backbone" to boost the efficacy of existing cancer therapies. Recent combination trials have shown striking survival gains—median overall survival of 27 months in metastatic colorectal cancer and a two‑year...
Biogen-Ionis Tau Antisense Drug Shows Cognitive Slow‑down in Early Alzheimer’s Phase 2
Biogen and Ionis announced that diranersen, an antisense therapy targeting tau protein, slowed cognitive decline and cut brain tau biomarkers in a 416‑patient Phase 2 study of early Alzheimer’s disease. The dual signal of biological impact and clinical benefit could...

Vanda’s Imsidolimab Wins Orphan‑Drug Designation in Japan for Rare Psoriasis
Vanda Pharmaceuticals announced that Japan’s Ministry of Health, Labour and Welfare has granted orphan‑drug designation to its IL‑36 inhibitor imsidolimab for generalized pustular psoriasis (GPP). The designation unlocks development subsidies and up to ten years of market exclusivity, a pivotal...

Q32 Bio Raises $55M as Guardant, Soligenix, Novavax Lead Biotech Gains
Q32 Bio Inc. closed a $55 million private placement, lifting its shares more than 85% in a single session. Guardant Health earned an American Cancer Society recommendation, Soligenix announced Ebola‑focused platform work, and Novavax posted solid gains, making them the day’s...