
Fat Cells Burn Energy to Make Heat – Making Them the Next Frontier of Weight Loss Therapies
New obesity drugs such as Ozempic, Wegovy and Mounjaro have proven that appetite suppression can drive significant weight loss, but they address only half of the energy balance equation. Researchers are now turning to adipose tissue, especially brown and beige fat, which burn calories by generating heat. While cold exposure can activate these thermogenic cells, the body often compensates with increased hunger, limiting weight‑loss potential. Combining appetite‑suppressing GLP‑1 agents with therapies that safely boost fat‑cell heat production could create a two‑pronged approach to obesity treatment.

TriSalus (TLSI) Initiates Patient Enrollment for PREDICTT Liver Tumor Clinical Trial
TriSalus Life Sciences announced on May 4 that patient enrollment has begun for the PREDICTT trial, a prospective study of its Pressure‑Enabled Drug Delivery (PEDD) platform in liver tumors. The investigator‑led trial at MD Anderson Cancer Center will enroll about 20...
Here Is Why Ovid Therapeutics (OVID) Is One of the Best Fast Growing Penny Stocks
Ovid Therapeutics saw its price target raised by H.C. Wainwright to $4 from $2, reflecting confidence in its advancing pipeline. The company reported clean safety data for a 7 mg dose of its GABA‑aminotransferase inhibitor OV329, with no serious adverse events....
AtaiBeckley (ATAI) Reports Positive Phase 2a Results for Social Anxiety Treatment EMP-01
AtaiBeckley announced expanded Phase 2a results for EMP‑01, an oral R‑MDMA formulation targeting Social Anxiety Disorder. The study showed a 38% reduction in patient‑reported symptoms and a 32% drop in real‑world avoidance behaviors by Day 43, with 49% of participants classified as...

Partner Therapeutics’ Bizengri Gains FDA Approval and Fast‑Track Voucher for Rare Bile‑Duct Cancer
Partner Therapeutics announced that its antibody Bizengri (zenocutuzumab‑zbco) has been approved by the U.S. FDA for adults with advanced NRG1‑fusion cholangiocarcinoma. The agency also granted a fast‑track voucher, signaling regulatory confidence in the drug’s potential to address an unmet oncology...

FDA Extends Review of Eisai/Biogen Subcutaneous Leqembi to Aug 2026
Eisai and Biogen announced that the FDA has added three months to its review of a supplemental biologics license application for weekly subcutaneous Leqembi IQLIK, moving the action date to Aug. 24, 2026. The agency cited a major amendment and a...

BridgeBio Q1 2026 Earnings Show Atruvio Revenue Surge to $180.6M
BridgeBio posted Q1 2026 revenue of $194.5 million, powered by Atruvio net product revenue of $180.6 million—a 24% sequential rise and 392% year‑over‑year increase. The company also announced a $500 million share‑repurchase program and a cash balance of $940.2 million, underscoring a stronger financial...

Real World Outcomes Support the Benefits of Psychedelic Therapy for Severe Depression
A Swiss compassionate‑use program evaluated psychedelic‑assisted psychotherapy using either 100 µg LSD or 25 mg psilocybin in adults with treatment‑resistant depression or anxiety. More than a third of participants reported at least a 50% reduction in depressive symptoms within three months, and...

Upcoming Meeting to Shape Gerotherapeutic Regulatory Frameworks
Affecting the Aging Trajectory: Regulatory Constructs for Gerotherapeutic Drug, Biologic & Device Development 👉 “On May 27, 2026, the Reagan-Udall Foundation for the FDA, in collaboration with @ARPA_H and the @xprize Foundation, will convene a hybrid public meeting on exploring...
Estrogen Deficiency Triggers Bone EVs Causing Cell Aging
Researchers have discovered that estrogen deficiency prompts bone cells to release extracellular vesicles (EVs) loaded with pro‑aging molecular cargo. These bone‑derived EVs travel through the bloodstream and induce cellular senescence in distant tissues, accelerating systemic aging. In mouse models, pharmacologic...
Glucose Nanoparticles Help CBD Cross the Blood-Brain Barrier
Researchers have engineered glucose‑coated polymer nanoparticles that dramatically improve cannabidiol (CBD) delivery across the blood‑brain barrier. The particles use a PEG‑PHB core to solubilize CBD and a surface glucose layer to hijack GLUT‑1 transport, while reactive‑oxygen‑species triggers release in inflamed...

Guardant Health Q1 Revenue Surges 48% YoY to $302M, Fastest Growth in Five Years
Guardant Health posted Q1 2026 revenue of $302 million, a 48% year‑over‑year increase and the strongest growth rate in five years. The biotech’s oncology and screening divisions drove the surge, prompting the company to lift its full‑year revenue outlook to $1.30‑$1.32 billion.

Magnesium Nanocoating Promises Safer, Self‑Absorbing Medical Implants
Scientists from the University of the Sunshine Coast, China’s First People’s Hospital and the University of Tokyo unveiled a magnesium‑based nanocoating that adds antibacterial activity and improves biocomibility of degradable implants. The breakthrough could eliminate the need for removal surgery,...

U.S. Evacuates 17 Americans From Hantavirus‑Stricken Cruise to Nebraska Biocontainment Unit
The United States dispatched an emergency aircraft to fly 17 American passengers from the MV Hondius, where a hantavirus outbreak has killed three, to the University of Nebraska Medical Center’s biocontainment unit. The operation, coordinated by the State Department, CDC...

Ginkgo Bioworks Q1 Shows 49% Revenue Drop but Strong B2B Platform Demand
Ginkgo Bioworks posted Q1 2026 revenue of $19 million, down 49% year‑over‑year, but emphasized growing demand for its synthetic‑biology platforms from industrial customers. The company’s Nebula autonomous labs, new cloud‑based sales channels, and a $47 million federal contract illustrate accelerating B2B adoption.

CRISPR Base Editing Provides Irreversible Microbial Biocontainment
A CRISPR-dCas9 base editing system now enables irreversible shutdown of essential genes in engineered microbes, offering a robust biocontainment strategy that minimizes escape and enhances biosafety for industrial and therapeutic applications. biotechnology
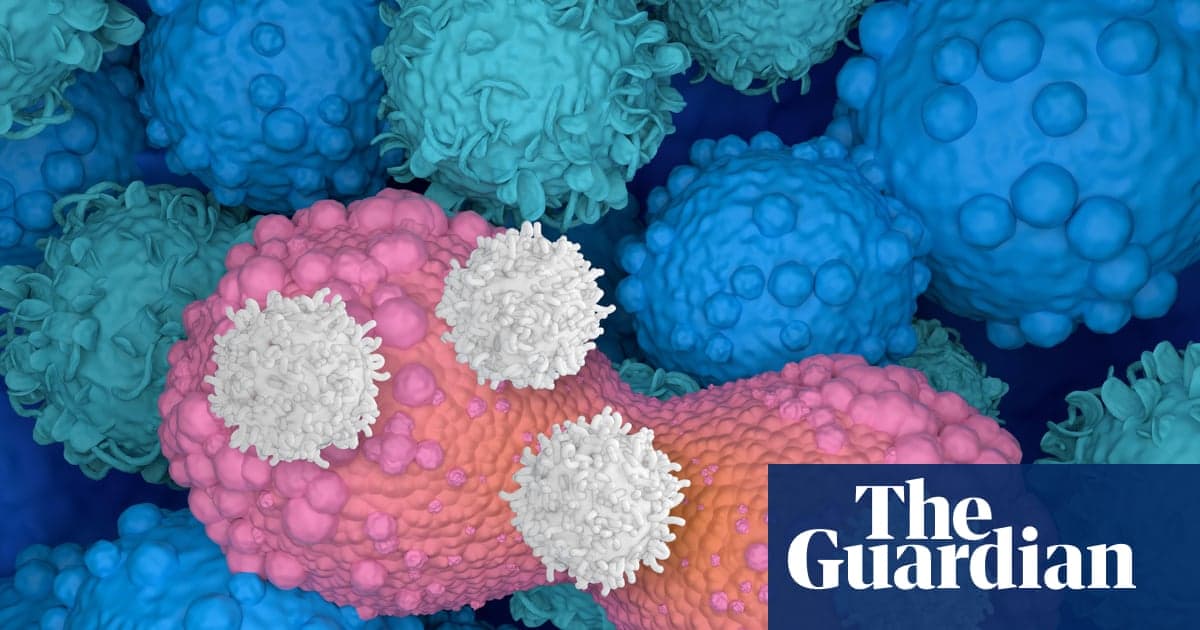
The Emerging Cancer Treatment That’s Exciting Scientists: ‘We’ve Just Scratched the Surface on What’s Possible’
CAR T-cell therapy, a genetically engineered immunotherapy, is gaining attention after Australian actor Sam Neill announced remission from stage‑three cancer following a clinical trial. Australia has approved four CAR T products since 2018, all targeting blood cancers, while researchers push the...

CRISPR Safeguard Changes How Engineered Microbes Can Be Controlled
Researchers have developed an irreversible CRISPR‑dCas9 base‑editing biocontainment system that disables essential genes in engineered microbes without causing DNA double‑strand breaks. By targeting the start codons of multiple essential genes, the platform permanently halts cell viability and dramatically lowers escape...
Esomeprazole Vs. Fexuprazan: Anti-Inflammatory Effects Compared
A 2026 study compared the anti‑inflammatory activity of the proton‑pump inhibitor esomeprazole and the potassium‑competitive acid blocker fexuprazan in LPS‑stimulated RAW 264.7 macrophages. Esomeprazole markedly reduced nitric oxide output by down‑regulating iNOS, while fexuprazan more strongly curtailed TNF‑α and IL‑6 release....

India Launches BHARAT Study, First Nationwide Search for Aging Biomarkers
The Indian Institute of Science has launched the BHARAT study, the country's first large‑scale, multi‑omics investigation of aging biomarkers. Led by Suramya Asthana and Deepak Kumar Saini, the project will enroll a demographically balanced cohort across urban and rural India...

Rznomics Secures FDA RMAT Designation for RZ-001 Liver Cancer Therapy
South Korean biotech Rznomics announced that the U.S. FDA granted Regenerative Medicine Advanced Therapy (RMAT) designation to its RNA‑editing candidate RZ‑001 for hepatocellular carcinoma. The designation, based on promising Phase 1b/2a data, speeds regulatory interaction and opens priority review pathways.

India’s Village Biotech Hub Unveils First Indigenous Bioreactor, Boosting Rural Innovation
Dyna Biotech, based in Phursungi village on Pune’s outskirts, launched India’s first indigenous bioreactor, marking a shift of high‑tech biotech into rural areas. The facility also rolled out a waste‑to‑energy system that claims five‑fold efficiency gains, positioning the hub as...

Interpol Seizes 6.42 Million Counterfeit Drug Doses Worth $15.5 M
Interpol coordinated a worldwide crackdown that confiscated 6.42 million doses of unapproved and counterfeit medicines, valued at $15.5 million. The operation, spanning 90 countries, resulted in 269 arrests and the dismantling of 66 criminal networks, underscoring persistent threats to pharmaceutical safety.

Antiglycation Potential of Launaea Taraxacifolia on Pentosidine- and Vesperlysine-Like Advanced Glycation End Products (AGEs)
Researchers evaluated a 70 % ethanol leaf extract of Launaea taraxacifolia for its ability to counteract advanced glycation end‑products (AGEs) linked to diabetic complications. Phytochemical analysis revealed notable levels of phenolics (7.27 mg GAE/100 mg) and flavonoids (11.03 mg QE/100 mg), and the extract showed...

RTMS Cuts Smoking by 11 Cigarettes Daily by Boosting Willpower, Study Finds
Researchers at the MUSC Hollings Cancer Center demonstrated that high‑frequency repetitive transcranial magnetic stimulation (rTMS) of the dorsolateral prefrontal cortex lowered cigarette consumption by an average of 11 cigarettes per day, outperforming reward‑targeting and sham treatments. The findings suggest a...

Texas A&M Study Shows Coffee Compounds Bind NR4A1, Boost Anti‑Aging Effects
Researchers at Texas A&M University have demonstrated that specific coffee phytochemicals bind to the NR4A1 receptor, a key stress‑response protein, and curb cancer cell growth in vitro. The findings suggest coffee’s longevity benefits stem largely from these compounds rather than...
Argenx Secures FDA Nod to Expand VYVGART to All Adult gMG Patients
argenx announced that the U.S. FDA has approved VYVGART and VYVGART Hytrulo for use in every adult with generalized myasthenia gravis (gMG), regardless of antibody status. The decision follows the Phase 3 ADAPT SERON trial, which demonstrated rapid, sustained symptom improvement across...
Choosing Traits: Mercy vs Competitive Advantage in Embryo Selection
Listening to @hsu_steve and @AlexTISYoung discuss the ethics of embryo selection is disorienting. Seems disingenuous to laugh off the difference between screening out Huntington’s and optimizing for height or IQ, as if the distinction is some kind of philosophical naivety. A...
Mabwell Secures FDA IND Clearance for 9MW5211 Antibody, Paving Way for IBD Trials
Mabwell (688062.SH, 02493.HK) announced that the FDA has cleared its investigational new drug application for 9MW5211, a first‑in‑class antibody targeting pathogenic immune cells in inflammatory bowel disease. The clearance enables the company to begin human trials and adds momentum to...

Traws Pharma Shares Surge 30% on Accelerated Hantavirus Drug Push
Traws Pharma (TRAW) saw its Nasdaq price climb roughly 30% to $2.20 after the company disclosed plans to fast‑track clinical candidates for hantavirus treatment and prevention. The move lifted the stock from an opening of $2.04 to a session high...

VIDEO: ‘New Era of Oral Therapies’ on Horizon in Psoriasis
At the American Academy of Dermatology meeting, data from phase 3 trials showed that next‑generation TYK2 inhibitors zasocitinib (Takeda) and envudeucitinib (Alumis) achieved significant plaque‑psoriasis reduction by week 24. These oral agents build on the earlier success of deucravacitinib, offering greater specificity...

This Organoid Can Menstruate—And Shows How Tissue Can Repair Itself
Researchers at the Friedrich Miescher Institute have engineered uterine‑lining organoids that can undergo a full menstrual cycle, shedding and then regenerating tissue without scarring. By exposing epithelial‑only spheroids to estrogen and progesterone, then withdrawing the hormones and mechanically inducing breakdown,...

UCSF Neurologist Finds Psilocybin Can Reset Depressed Brain
UCSF neurologist Robin Carhart‑Harris said a single high‑dose psilocybin treatment produced lasting symptom relief for patients with treatment‑resistant depression, positioning the psychedelic as a structural brain reset rather than a daily chemical tweak.
UCLA Study Shows Slow Breathing Calms Anxiety in Mice Without Mindfulness
UCLA neuroscientist Jack Feldman presented data at the Embodied Minds Summit showing that mice trained to breathe slowly displayed markedly less anxiety‑related behavior. The findings, based on optogenetic manipulation of the brainstem breathing center, argue that the calming impact of...

FDA Approves Veppanu, First PROTAC Therapy for ESR1‑Mutant Breast Cancer
The U.S. Food and Drug Administration has approved Veppanu (vepdegestrant), the first PROTAC drug for estrogen‑receptor‑mutant breast cancer. The decision follows the VERITAC‑2 trial, where Veppanu extended median progression‑free survival to five months in the ESR1‑mutant subgroup, compared with 2.1...

Nanoparticle Platform Achieves 100% Survival in Drug‑Resistant Cancer Mice
Researchers led by Prof. Eijiro Miyako at Tohoku University unveiled a multifunctional nanoparticle that first blocks P‑glycoprotein pumps, then releases doxorubicin, and adds near‑infrared photothermal heating. In mouse models of multidrug‑resistant tumors the treatment caused full tumor regression and 100%...

Isomorphic Labs Nears $2 Billion Funding Round Led by Thrive Capital
Isomorphic Labs, the DeepMind‑spun AI drug‑discovery startup, is negotiating a financing round of more than $2 billion. Thrive Capital will lead the round, with Alphabet also participating, positioning the company for global expansion and a next‑generation drug‑design platform.

China's NMPA Grants Breakthrough Therapy Designation to NHWD-870 for Rare NUT Carcinoma
Zhejiang Wenda Pharmaceutical's oral BET inhibitor NHWD-870 has earned breakthrough therapy designation from China's NMPA for advanced thoracic NUT carcinoma, after Phase II data showed a 45% response rate and median survival of 9.33 months. The designation accelerates review and...

Big Apple Exclusive: Dr. McCullough Goes Live to Unveil Potential Cancer Breakthrough
In this episode, Dr. Peter McCullough, chief scientific officer of a wellness company, discusses an observational report on the off‑label use of two generic drugs—ivermectin and mebendazole—in cancer patients. The study, now under peer review, found that nearly half of participants...

AI Predicts Chemoresistance in Bladder Cancer
Researchers published a machine‑learning framework that fuses tumor transcriptome profiles with high‑resolution digital pathology to forecast chemoresistance in muscle‑invasive bladder cancer (MIBC). The multimodal model outperformed single‑data approaches, accurately distinguishing resistant from responsive tumors in extensive validation cohorts. By pinpointing...

SuperDNA: AI-Driven Biotech Hub Accelerates Drug Discovery
Welcome to SuperDNA Up City - this will be the true automation biotechnology + AI Disneyland with 10 thousand humans in the loop. Most people do not realize that even today, with 30 developmental candidates some of which often trigger...
20 Years of iPSC Research: From Discovery to Applications
Two decades of induced pluripotent stem cell research: From discovery to diverse applications https://t.co/L9cObPcVwk https://t.co/gcoiPh6yqJ

17-Alpha Estradiol - Another Top Anti-Aging Drug
The post details efforts to source 17α‑estradiol (alfatradiol) for anti‑aging and renal‑protective research, listing vendors and pricing—including Octagonchem’s $1,200 for 100 g and Shanghai Jizhi’s roughly $80 for 200 mg (≈¥568). It proposes a group‑buy workflow that starts with small‑scale sampling, purity...

AI‑Created Bio Designs Lack Expert Oversight
bioslop (noun) - biological molecules and/or designs made by Ai without the oversight or review of a subject matter expert or specialist LLM
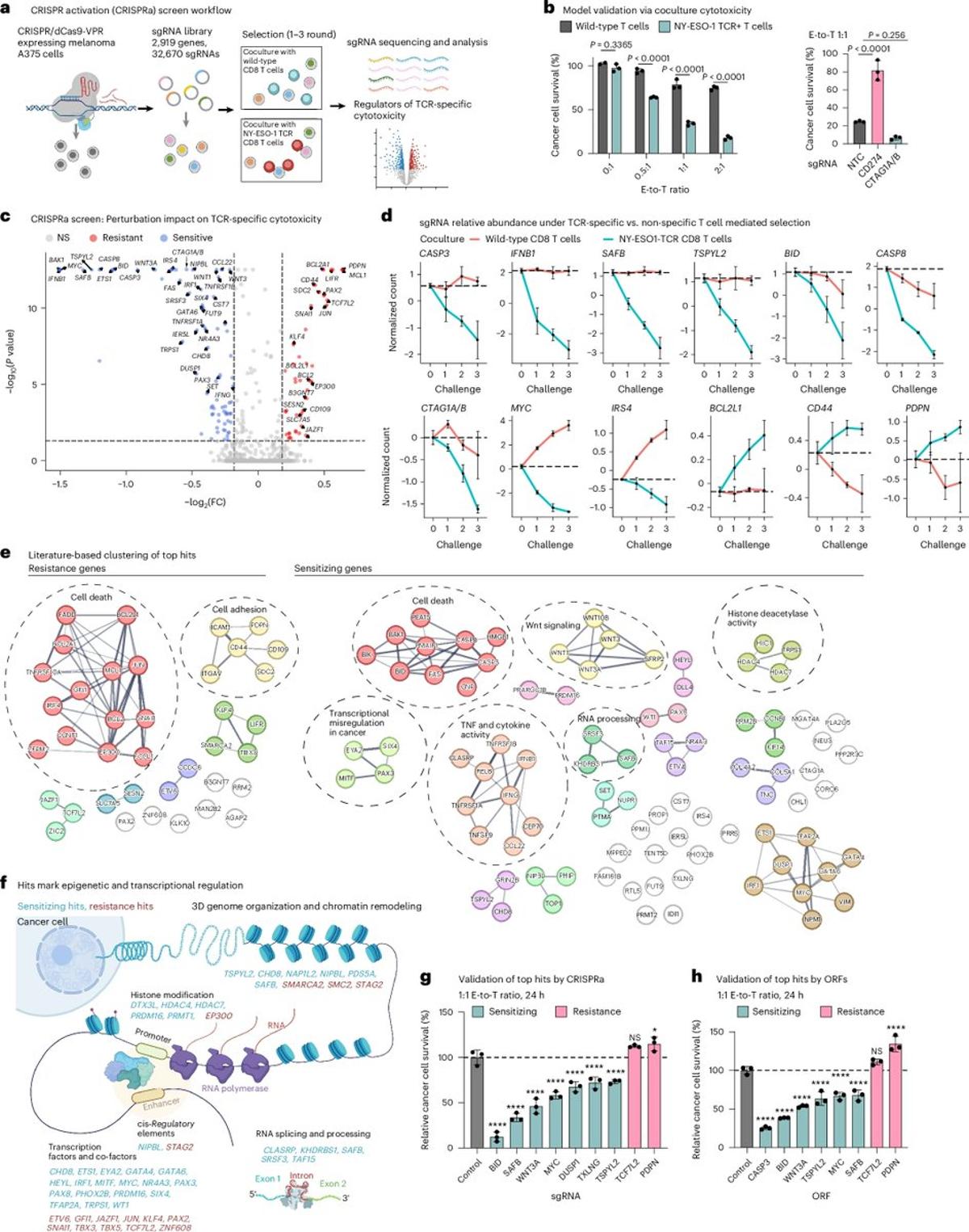
CRISPR Screens Reveal RNA Targets that Boost T‑cell Killing
High-content CRISPR activation screens identify synthetically lethal RNA-based mechanisms to sensitize cancer cells to targeted T cell cytotoxicity https://t.co/gi3hEvoi9V https://t.co/njFRebLRVo

Queensland Funds New Biomedical Manufacturing Projects to Strengthen Sovereign Capability
Queensland has earmarked about US$133 million from its Sovereign Industry Development Fund for its first biomedical manufacturing projects. More than US$12.9 million will support AdvanCell’s Thorium‑228 radiopharmaceutical plant and US$1.6 million will fund Southern RNA’s semi‑automated sterile fill‑finish platform, together creating roughly 100...

Isomorphic Labs Eyes $2B Raise, Biotech’s Second‑largest
Great scoop from @RebeccaTorrenc5 & @mhbergen on Isomorphic Labs in "advanced discussions to raise more than $2 billion in a new round of funding" led by Thrive Capital Would be second-largest VC raise ever in biotech, only behind $3B for...

Stop Wasting Hours Matching Sample IDs Across Assays
1/ How many hours do bioinformaticians lose matching sample IDs across assays? Too many. And it’s avoidable. Let’s talk about why this happens—and how to stop it. https://t.co/wxR6DIMzPv

AI-Driven Method Creates Robust Clinical Trial Surrogate Markers
Super interesting via @oziadias & @DeanKateBaicker on an approach to generating robust surrogate markers, urgently needed but elusive, for clin trials, essentially via channeling the AI-enabled empiric analysis that I associate w PRS (cc @pnatarajanmd) https://t.co/FDP4Ym8ycC

Low‑Cost Vaccines and Open Science Aren’t Evil
Hmmm, presumably making low-cost vaccines to help humanity, $2-4 per dose, no onerous patent restrictions, pioneering a way to bypass big pharma, helping the nation through a terrible pandemic, writing books with academic presses on history of science/medicine, constitute evil?