Avalyn Pharma Takes a Breath to Raise $300M in IPO Cash for Lung Drug Trials
Avalyn Pharma completed a $300 million IPO, pricing 16.6 million shares at $18 each and listing on Nasdaq under the ticker AVLN. The Boston‑based biotech will use the proceeds to advance inhalable versions of pirfenidone (AP01) and nintedanib (AP02), plus a combination product (AP03), targeting idiopathic and progressive pulmonary fibrosis. Inhalable delivery aims to lower systemic exposure, reduce side‑effects, and improve patient adherence compared with existing oral antifibrotics. The company projects its cash runway to extend through 2029, supporting mid‑stage trials and a planned move to Phase 3 studies.

Pharma Power Shifts: Controlling Diseases, Not Just Assets
Most pharma pipeline analysis is spreadsheet theater. I used AI to look at the 2020 → 2026 shift. The real question is not: Who has the most assets? It is: Who controls the disease category? GLP-1s are a metabolic land grab. Oncology is becoming a platform stack. Trials...

FDA Expedites Review of New HIV Treatment Combo
The FDA has granted priority review to Gilead Sciences’ new single‑tablet HIV regimen that combines bictegravir and lenacapavir. The application is backed by phase 3 ARTISTRY‑1 and ARTISTRY‑2 trials, which showed sustained viral suppression in adults already virologically controlled. If approved,...

FDA Moves to Speed Review Support for Psychedelic-Related Treatments
The FDA announced a new fast‑track mechanism for psychedelic‑based therapies, issuing Commissioner’s National Priority Vouchers to three programs targeting depression and PTSD with psilocybin or methylone. It also granted an IND for DemeRx NB to study noribogaine hydrochloride as a...

Varenicline
Varenicline (Chantix) received FDA approval in 2006 as a partial α4β2 nicotinic receptor agonist, offering a middle‑ground approach between nicotine replacement and bupropion. Its mechanism delivers enough receptor activation to ease cravings while antagonizing nicotine’s rewarding effects. The drug quickly...
IDEAYA Biosciences Files NDA for Darovasertib/Crizotinib Under FDA’s RTOR Pathway
IDEAYA Biosciences announced that the FDA has accepted its New Drug Application for the darovasertib‑crizotinib combo under the Real‑Time Oncology Review (RTOR) program. The submission follows Phase 2/3 OptimUM‑02 data showing a 58% drop in disease‑progression risk and a median progression‑free...

Merck Still Sees ‘Compelling’ Outlook for Terns Leukemia Drug
Merck agreed to acquire Terns Pharmaceuticals for $6.7 billion, paying $53 per share, after updated trial data showed its TERN‑701 leukemia drug achieving a major molecular response (MMR) rate north of 50% at 24 weeks. The data suggested TERN‑701 could outperform...

Harvard Scientists Unveil First ‘Smell Map’ Using 5.5 Million Neurons
A team at Harvard Medical School has built the world’s first detailed map of olfactory receptors, analyzing more than 5.5 million neurons from over 300 mice. The map shows horizontal stripe patterns rather than random distribution, a discovery that could reshape...

South Korea Clears Curocell's Limcarto‑Joo, First Home‑Grown CAR‑T Therapy
Curocell's Limcarto‑Joo received marketing approval from South Korea's Ministry of Food and Drug Safety, becoming the country's first domestically developed CAR‑T therapy. The treatment targets adult patients with diffuse large B‑cell lymphoma and primary mediastinal B‑cell lymphoma, marking a shift...

Other News to Note for April 30, 2026
Chinese biotech companies have reached an inflection point, gaining leverage as co‑dealmakers in cross‑border partnerships, according to multinational executives. Roche announced the discovery of new TREM2 agonist compounds aimed at modulating microglial activity for neurodegenerative diseases. Plasticity launched an advanced...

The Bangui Operation: A Story of Blood, Science and Biomedical Exploitation
In the early 1990s the Pasteur Institute in Bangui ran a covert HIV‑vaccine trial that recruited roughly 3,000 Central African soldiers, extracting over 11,000 blood samples. The research was funded by French institutions and aimed to fast‑track vaccine development at...
STAT+: Five-Way Obesity Drug Is Super Effective — in Mice
Researchers have unveiled a preclinical obesity candidate that combines five distinct mechanisms, delivering dramatic weight loss in mouse models and outperforming current GLP‑1 therapies. The multi‑modal approach targets appetite, metabolism, gut hormones, energy expenditure, and adipose tissue remodeling, achieving up...

Swiss BioTech Startup ALP Bio Raises €1.9 Million to Advance Immune Organoid and AI Platform
Swiss startup ALP Bio announced a €1.9 million (≈$2.1 million) pre‑seed round led by 42CAP to accelerate its immune organoid‑AI platform for antibody development. The platform couples human tonsil organoid readouts with generative AI to predict and reduce immunogenicity risk earlier in...
Top 15 Biopharma R&D Players and Late‑Stage Prospects
My annual report on the top 15 R&D players in biopharma is out. It's a beast at some 9000 words, but if you're interested in the top prospects in the late-stage pipeline, I got a bunch for you. The stakes...

Zealand Pharma and Roche Advance Petrelintide to Phase 3 for Chronic Weight Management
Zealand Pharma and Roche are moving the amylin analog petrelintide into Phase 3 trials to treat chronic overweight and obesity. The drug achieved up to 10.7% mean weight loss in the Phase 2 ZUPREME‑1 study, with tolerability comparable to placebo. A March 2025...

Merck Announces First Dose in Phase 3 Study with Enpatoran for Lupus Patients with Active Skin Manifestations
Merck announced the first patient has been dosed in its global Phase 3 ELOWEN program, testing the oral TLR7/8 inhibitor enpatoran in lupus patients with active skin manifestations. The double‑blind, placebo‑controlled studies—ELOWEN‑1 and ELOWEN‑2—will enroll roughly 200 participants each across 266...

FDA Proposes Excluding Novo, Lilly Weight Loss Drugs From Bulk Compounding List in Win for the Companies
The FDA has proposed removing the active ingredients of Novo Nordisk’s semaglutide and liraglutide and Eli Lilly’s tirzepatide from the bulk‑compounding list used by 503B outsourcing facilities. If the rule is finalized, these high‑demand obesity and diabetes drugs could only be...

Irisin Hormone Reverses Obesity and Insulin Resistance
Irisin, a hormone released by muscle during exercise, reverses obesity and insulin resistance in mice -- without cutting food intake or causing muscle loss. As a medical school professor, I find this striking. We have spent a decade asking how to...

Water Molecules Found to Actively Drive Gene Transcription Process
Researchers used sub‑2 Å cryo‑electron microscopy to map individual water molecules inside RNA polymerase II, revealing over a thousand waters positioned at catalytic sites. The study shows these waters actively mediate proton transfer, substrate recognition, and structural stabilization during transcription. This challenges...

CGT Global Appoints Charlotte Ivancic to Board of Advisors, Strengthening Strategic Leadership in Cell and Gene Therapy Policy and Market...
CGT Global announced the appointment of Charlotte Ivancic to its Board of Advisors, bringing over 25 years of federal health policy and legislative experience. Ivancic, a partner at FGS Global and former senior aide to Senate Majority Leader Bill Frist,...

What We Know (and Don’t Know) About Peptide Safety
Peptide safety is neither automatically assured by mimicking endogenous hormones nor inherently hazardous because research is incomplete. The risk profile depends on the specific peptide, the depth of clinical evidence, product purity, and real‑world usage conditions. Early‑phase trials provide maximum...

Canada’s Life Sciences Sector Welcomes Federal Economic Update’s Focus on SR&ED Implementation
BIOTECanada welcomed the 2026 Spring Economic Update, which confirms the rollout of SR&ED administrative reforms first announced in Budget 2025. The changes raise the annual expenditure limit and taxable‑capital phase‑out thresholds for the enhanced 35% credit, extend the credit to...

GLP-1s May Prevent Incident AF, Series of Studies Shows
Observational analyses presented at Heart Rhythm 2026 suggest GLP‑1 receptor agonists, including tirzepatide, lower the risk of incident atrial fibrillation by 33%‑47% across diverse patient groups. The benefit appears consistent in individuals with or without diabetes, obesity, and chronic kidney...

Gene Therapy Soon as Routine as Everyday Surgery
Eric Kelsic envisions the future of gene therapy will be viewed in the “the same way that many of us have had some form of surgery at certain points in our life.” As therapies become safer and more effective, the decision...

Why Protocol Complexity Keeps Increasing – and How to Fix It
Protocol complexity in late‑stage trials is spiraling, with Phase III studies now averaging about 6 million data points—a rise of roughly 11% each year. The growth is fueled by scientific advances, global regulatory demands, and the ease of capturing extra data via...
DNA Nano‑Ring Precisely Captures and Orients Membrane Proteins
A new DNA nano-ring platform enables precise capture and orientation of individual membrane proteins, advancing the study of these vital cellular gatekeepers and opening new possibilities in medicine, imaging, and synthetic biology. nanotechnology

Researchers Create DNA 'Nano-Rings' To Control Viral Cell Proteins
Scientists at Durham University and Jagiellonian University have engineered DNA‑origami nano‑rings that trap individual membrane proteins within nanodisks, creating a highly controllable platform for structural studies. The DNA‑Origami‑Constrained Nanodisks (DOC‑NDs) reliably capture single proteins and can dictate their orientation, a...
Plasmid Editor Adds API for AI-Driven Real-Time Editing
A feature I'm working on adding to my plasmid editor is an API for agent work. This lets your favorite Ai do work on the files in your plasmid editor directly, and the results update in your open GUI in...

New GLP‑5 Study Is Only Mouse
A friendly reminder, just because that new Nature study on a quintuple agonist (LOL GET READY FOR "GLP-5" discourse) is out today: It's a study on MICE. MICE TESTING IS NOT HUMAN TESTING. IT'S JUST MICE.

The FDA Real Time Clinical Trial Announcement Quietly Dissolves Phase Gates, Breaks Biotech Capital Markets Plumbing, and Opens a Founder...
The FDA announced on April 28 that real‑time clinical trials (RTCTs) are now live, with AstraZeneca’s phase‑2 lymphoma study and Amgen’s phase‑1b small‑cell lung cancer trial streaming data through Paradigm Health. The agency claims a 20‑40 % reduction in development timelines, but...
UniQure Pursues UK Approval for Huntington’s Gene Therapy
UniQure, in ‘symbolic’ win, to seek UK approval of Huntington’s gene therapy https://t.co/plxdEw43FD by @Lilah_Alvarado $QURE + 22% #GeneTherapy

Pinetree Therapeutics: Promising Biotech Worth Watching
A biotech you need to keep an eye on... Pinetree Therapeutics $AZN https://t.co/wVt9XMcr0Z (disclosure: SAB member)
Wearable Sweat Patch Detects Six Vitamins in Real Time, Hits 0.33 nM Sensitivity
Researchers have unveiled a flexible skin patch that measures six essential vitamins in sweat, achieving detection limits as low as 0.33 nanomolar for folic acid. Published in Nature Communications, the study demonstrates real‑time, non‑invasive micronutrient monitoring that could reshape personalized nutrition.

Wine‑and‑Cheese Fundraising: A Fresh Biotech Investor Model
This was a cool event. Took a different tact, brought new and very seasoned investors together over wine and cheese, and raised funds @AnywhereDx …Maybe an interesting future model for raising funds in a biotech atmosphere that feels increasingly disconnected.

Trump's Psychedelic Order Preserves FDA Oversight Balance
My op ed in today's @WashPost - "Trump’s executive order on psychedelics strikes a healthy balance: The president preserved the FDA's role, even as some want the agency's gatekeeping dismantled." https://t.co/9hH7kz107J

Clinical Trial Finds Rapamycin Undermines Exercise Gains in Older Adults
Researchers led by Brad Stanfield reported that a 13‑week, randomized trial of 40 sedentary older adults in New Zealand found rapamycin blunted the physical benefits of a modest home‑exercise program. Participants taking the drug showed weaker strength, more fatigue and...
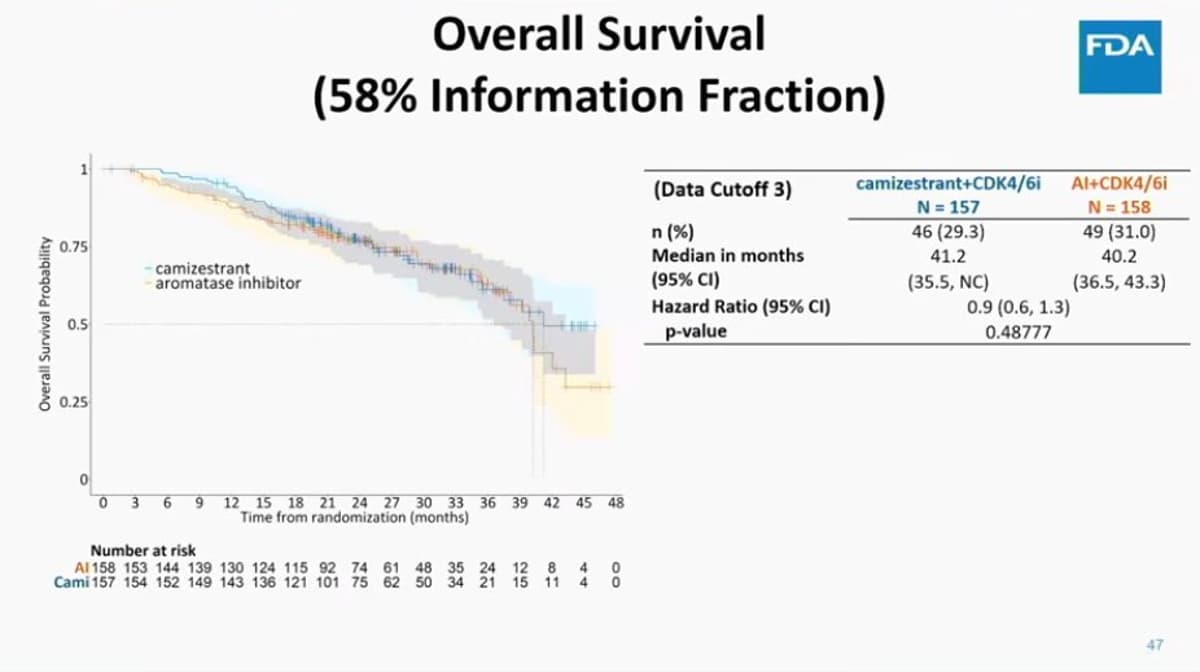
Serena-6 Shows Unexpected Immature OS Curves
$AZN Serena-6 immature OS curves as revealed at the adcom. Not sure we've seen these before. https://t.co/L9oAQxhrh9

Year Two Funding Crisis: Grants Dry, Competition Rises
With federal research funding cuts, the first year of Trump 2.0 was tough. Year 2 is in some ways grimmer, a @statnews survey shows. Bridge funding is drying up, existing grants are ending & competition for fewer new grants is...

FDA Grants Priority Review to BeOne’s TEVIMBRA Combo for HER2+ Gastric Cancer
The U.S. Food and Drug Administration has granted priority review to BeOne Medicines’ TEVIMBRA (tislelizumab) combined with ZIIHERA (zanidatamab) and chemotherapy for first‑line treatment of HER2‑positive gastroesophageal adenocarcinoma. The decision follows a Phase 3 trial that showed a median overall survival...

Better Prediction of Type 1 Diabetes Boosts Prevention Prospects
A jump in our ability to predict Type 1 autoimmune diabetes should help future preventive strategies @NatureGenet https://t.co/ako2Ic46QW https://t.co/XbW26qu5LE
Akeso’s ASCO Plenary Raises Questions on Lung Cancer Data
This week's Biotech Scorecard: An #ASCO26 mystery: What does Akeso’s primo plenary spot say about its ivonescimab lung cancer survival data? $SMMT If the news was good, we'd have heard by now, right? Or, is Akeso just being ultra conservative?...

Chiesi to Acquire KalVista for $1.9 B, Adding First Oral HAE Therapy
Chiesi Group announced a $1.9 billion cash deal to buy KalVista Pharmaceuticals, paying $27 per share—a 36% premium—to acquire the FDA‑approved oral hereditary angioedema therapy Ekterly. The transaction, slated to close in the third quarter of 2024, marks Chiesi’s largest acquisition...

High Accuracy in Omics May Be Illusory
The pitfalls of class prediction in omics 🧵 1/ You think you’ve built the perfect omics predictor. The accuracy is high. The p-value is low. But is it real—or just a story your data whispered back? https://t.co/LGNWQMKOfv

Pfizer Unveils Nanoparticle Platform Aimed at Precision Cancer Therapy
Pfizer disclosed a nanotechnology‑based drug‑delivery platform that uses engineered nanoparticles to target tumors with high precision. The initiative, led by Puja Sapra of the Targeted Therapeutics Unit, seeks to reduce collateral damage to healthy cells and could reshape oncology pipelines.

Remember Craig: Push Bold Biotech Innovation
RIP to one of our few ballers in biotech. I hope we use Craig's memory to not be afraid to go big in biotech. The world needs our tech and it's a loss to not have his voice...
GSK's EXDENSUR Secures FDA Approval with Groundbreaking Twice‑Yearly Dosing for Severe Asthma
GlaxoSmithKline's depemokimab, branded EXDENSUR, received FDA approval on Dec. 16, 2025 as an add‑on therapy for severe eosinophilic asthma. The biologic is administered subcutaneously at 100 mg once every six months, a dosing interval not seen before in this therapeutic class. Approval was...

Avalyn Pharma Prices $300 Million IPO at $18 per Share, Expanding Biotech B2B Financing
Avalyn Pharma announced the pricing of its upsized initial public offering, selling 16.67 million shares at $18 each to raise $300 million. The Nasdaq‑listed biotech will use the proceeds to advance its pulmonary‑fibrosis pipeline and deepen B2B collaborations across the pharmaceutical supply...

Five Companies Pushing Biotech in Scotland
Scotland’s life‑science sector now comprises roughly 750 companies and 46,000 employees, anchored by strong university spin‑outs in Edinburgh, Glasgow and Dundee. Within this ecosystem five biotech firms are drawing attention: Cumulus Oncology’s portfolio‑driven oncology asset creation, EnteroBiotix’s microbiome‑based IBS therapy...

BeOne Receives the US FDA Priority Review to Tevimbra Combination for 1L Treatment of HER2+ GEA Cancers
The U.S. FDA granted priority review to BeOne Medicines’ Tevimbra‑Ziihera‑chemotherapy regimen for first‑line treatment of unresectable HER2‑positive gastric, gastro‑esophageal junction and esophageal adenocarcinomas. The decision follows the Phase III HERIZON‑GEA‑01 trial, which enrolled 914 patients and compared the new combos against...

Canada First in G7 to Approve Generic Ozempic
Health Canada has granted approval for the first generic version of semaglutide, the active ingredient in Ozempic, making Canada the first G7 nation to do so. The generic injection, submitted by Dr. Reddy’s Laboratories, is indicated for once‑weekly treatment of adult...