Leishmanicidal Efficacy of Cold Atmospheric Multiple Plasma Jet Against Leishmania Major in a Murine Model: Effects on Parasite Burden, Cytokine...
The study evaluated a cold atmospheric multiple plasma jet (CAMPJ) as a novel therapy for cutaneous leishmaniasis in BALB/c mice. Topical applications of 5, 10 and 15 minutes, administered twice weekly for three weeks, markedly reduced lesion size and splenic parasite burden compared with untreated and air‑treated controls. The 15‑minute regimen produced the smallest lesions and the lowest Leishman‑Donovan Units, while also shifting cytokine profiles toward a Th1‑biased response. Its efficacy approached that of daily topical amphotericin B, suggesting CAMPJ could serve as an alternative or adjunct treatment.
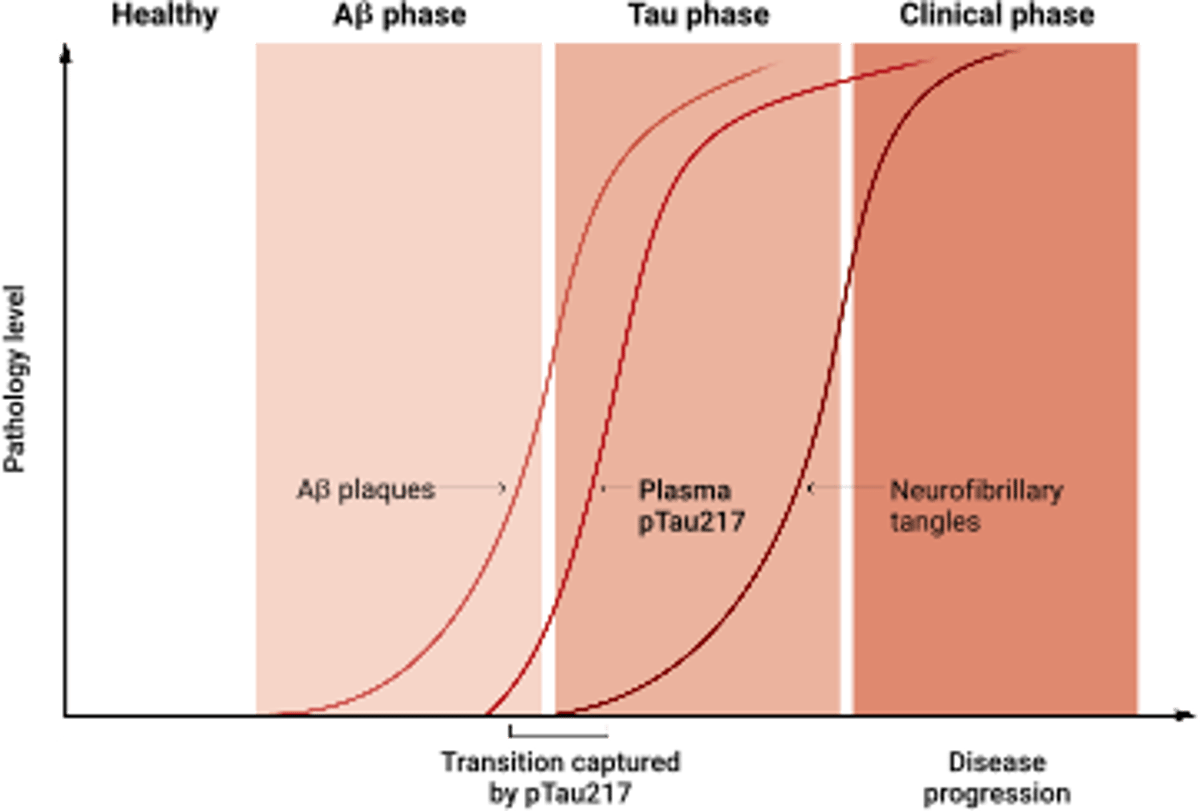
Blood Test Pinpoints Amyloid‑to‑tau Transition in Alzheimer’s
Tracking the turning point in Alzheimer’s disease A blood biomarker reveals the mechanistic shift from amyloid to tau pathology https://t.co/p2UfYwgBHW https://t.co/Kuu82UPrFv

Semaglutide Cuts Heavy Drinking and Cravings in AUD
GLP-1 Drug Semaglutide (Ozempic, Wegovy) Reduced Heavy Drinking & Craving in Adults with Alcohol Use Disorder https://t.co/FYRkjWYhJQ https://t.co/cGtEajk9lp

FDA, up Close and Personal: A Banker’s Recalibration
Former FDA senior advisor Peter Reikes argues that the agency’s perceived slowness stems from its core principles of independence, caution, and process discipline rather than dysfunction. He notes that despite recent leadership turnover and political pressure, a deep bench of...

Calorie Restriction Deactivates Complement C3a, Slowing Inflammaging
"complement C3a reduction is a metabolically regulated inflammatory checkpoint that can be harnessed to attenuate inflammaging" Exoproteome of calorie-restricted humans identifies complement deactivation as an immunometabolic checkpoint reducing inflammaging https://t.co/FxGHE6Obm0

The CRISPR Medicine That Cured a Child: How Yuri Milner’s Breakthrough Prize Brought Gene Therapy to Hollywood
At the 2026 Breakthrough Prize ceremony, Baby KJ was celebrated after receiving Casgevy, the first FDA‑approved CRISPR gene‑editing therapy for sickle cell disease and beta‑thalassemia. The $3 million prize honored researchers Stuart Orkin, Swee Lay Thein and others whose work on the BCL11A...

EnteroBiotix Raises $25 Million to Launch Largest Microbiome IBS Therapy Study
EnteroBiotix announced a $25 million financing round led by Thairm Bio and the Scottish National Investment Bank to fund the largest microbiome‑based Phase IIb trial in irritable bowel syndrome with constipation (IBS‑C). The capital will support a 300‑patient study in the...

Eli Lilly Inks $2.25 B AI‑driven Drug Discovery Pact with Profluent
Eli Lilly has signed a $2.25 billion agreement with AI‑specialist Profluent to co‑develop a platform for computational drug discovery. The deal, announced today, signals a major capital shift toward AI‑enabled R&D in pharma.

This Organoid Can Menstruate — and Shows How Tissue Can Repair Itself
Researchers at the Friedrich Miescher Institute have engineered 3‑D endometrium organoids that can undergo a full menstrual cycle, shedding and regenerating tissue without scarring. By exposing the organoids to estrogen and progesterone and then withdrawing the hormones, they induced hormonal...

We Tried to Get Trump to Back Psychedelic Therapy — Here’s What Happened
The authors drafted a bipartisan policy blueprint to accelerate psychedelic‑based treatments, targeting veterans with PTSD, addiction and depression. After Joe Rogan shared the proposal, it amassed over 1.5 million views on X, prompting meetings with health‑policy influencer Calley Means and a...
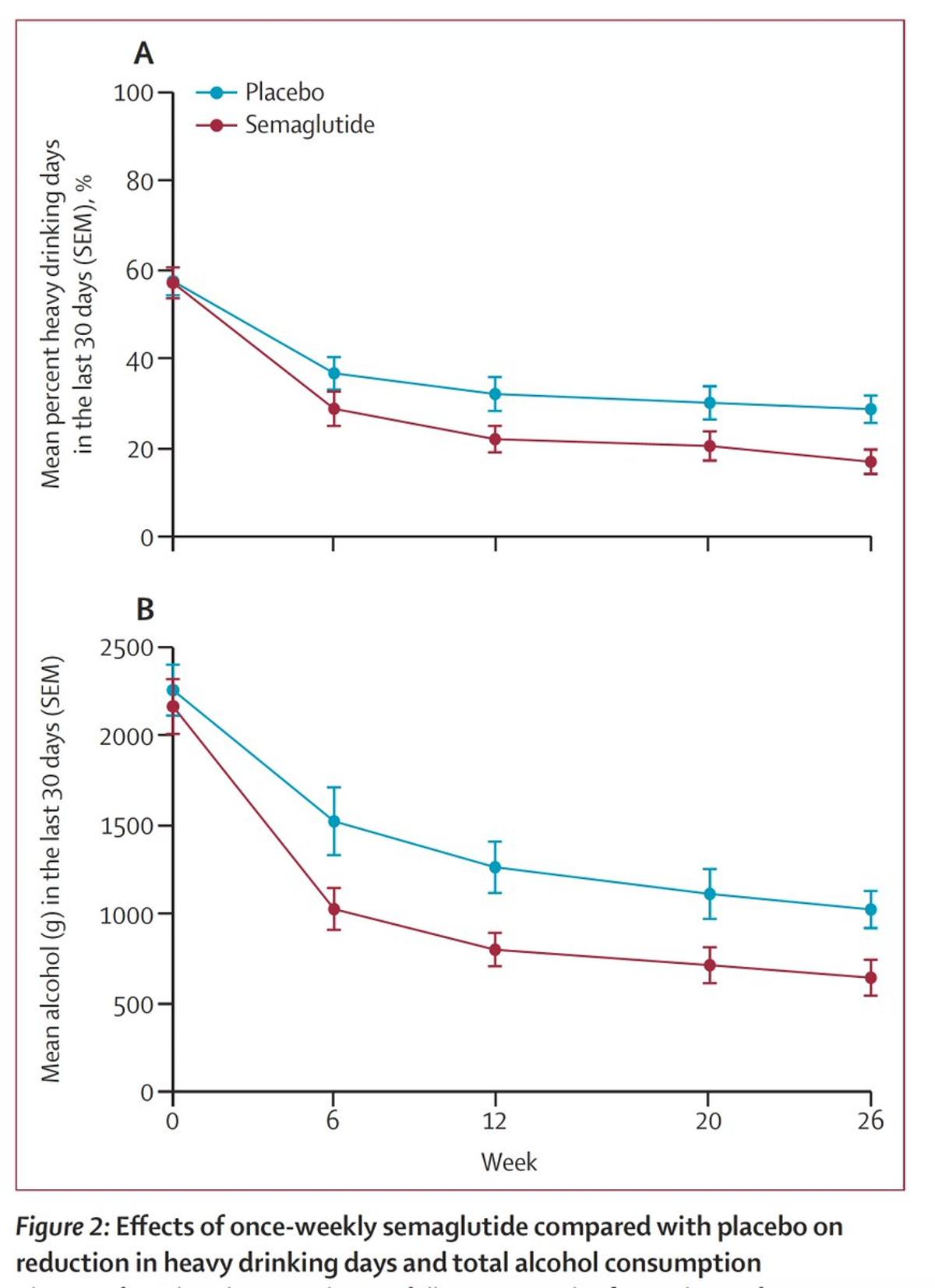
Ozempic Cuts Drinking in Obese AUD Patients
First randomized trial to show Ozempic reduces alcohol consumption in people seeking treatment for alcohol use disorder. Placebo-controlled, double-blind. Participants with BMI >30 kg/m2. https://t.co/M9Pk2gzKHD @TheLancet https://t.co/OT660QIn6N

GLP-1s Reduce Heavy Drinking Days in Patients With Obesity, Alcohol Use Disorder
A randomized, double‑blind trial in Copenhagen found that once‑weekly semaglutide 2.4 mg significantly reduced heavy‑drinking days in patients with alcohol use disorder and obesity. Over 26 weeks, the semaglutide group saw a 41.1‑percentage‑point drop in heavy‑drinking days versus 26.4 points for...

Remembering J. Craig Venter: A Relentless Scientist Who Changed Biotech — and Was All Too Easily Misunderstood
J. Craig Venter, the pioneering genomics entrepreneur who died at 79, reshaped biotechnology by accelerating the human genome sequence and building the first synthetic cell. His private‑sector efforts, including Celera and Synthetic Genomics, turned DNA sequencing into an industrial process. Venter’s...
GLP‑1 Drugs Cause Less Muscle Loss than Assumed
Do GLP-1 drugs really cause muscle loss? Turns out perhaps less than what we thought… here’s what you need to know, from: https://t.co/VwrRxnoblW https://t.co/M2RaE1f3EG
Single-Vesicle Profiling Could Push Liquid Biopsies Toward Everyday Clinical Use
Researchers from Incheon National University and the University of Pennsylvania reviewed cutting‑edge single‑extracellular vesicle (EV) profiling technologies that isolate and analyze vesicles one at a time. The review, published in TrAC Trends in Analytical Chemistry, highlights substrate‑based, droplet‑based and solution‑based...

The BioPharm Brief: Metabolic Phase III Progress, HER2 Oncology Momentum, and US Manufacturing Expansion Drive Industry Scale
Zealand Pharma and Roche are moving petrelintide, an amylin analog, into Phase 3 trials for chronic weight management, with enrollment slated for the second half of 2026 after earlier studies showed double‑digit weight loss and tolerability comparable to placebo. The FDA...
CRISPR Speed Patterns Can Identify Multiple Viruses and Variants Simultaneously
KAIST researchers and partners have unveiled a CRISPR‑Cas13 diagnostic that reads the enzyme's reaction speed to identify multiple viruses and variants in a single test. By encoding kinetic patterns as a barcode, the method distinguishes pathogens without needing separate gene...

Drug Trials Snapshots: BIMZELX
UCB’s BIMZELX, a dual IL‑17A/F monoclonal antibody, received FDA approval on Oct 17 2023 for adults with moderate‑to‑severe plaque psoriasis. The approval was based on two phase‑3 placebo‑controlled trials that enrolled 839 patients across 13 countries, with a demographic profile of 72%...

Drug Trials Snapshots: AGAMREE
Santhera Pharmaceuticals received FDA approval on October 26, 2023 for AGAMREE (vamorolone), a corticosteroid designed to treat Duchenne muscular dystrophy (DMD) in patients aged two years and older. The approval rests on a multinational, double‑blind, 24‑week trial of 121 male patients that...

Drug Trials Snapshot: TALVEY
TALVEY (talquetamab‑tgvs), a bispecific antibody from Janssen Biotech, received FDA approval on August 10 2023 for adult patients with relapsed or refractory multiple myeloma who have exhausted at least four prior therapies. The pivotal trial enrolled 339 participants across 47 sites, with...

Drug Trials Snapshots: OJJAARA
OJJAARA (momelotinib) received FDA approval on September 15 2023 for adults with myelofibrosis‑related anemia. The approval rests on two pivotal trials—MOMENTUM, which enrolled JAK‑inhibitor‑experienced patients, and SIMPLIFY‑1, which enrolled JAK‑naïve patients. In MOMENTUM, 25% of OJJAARA recipients achieved a ≥50% reduction in...
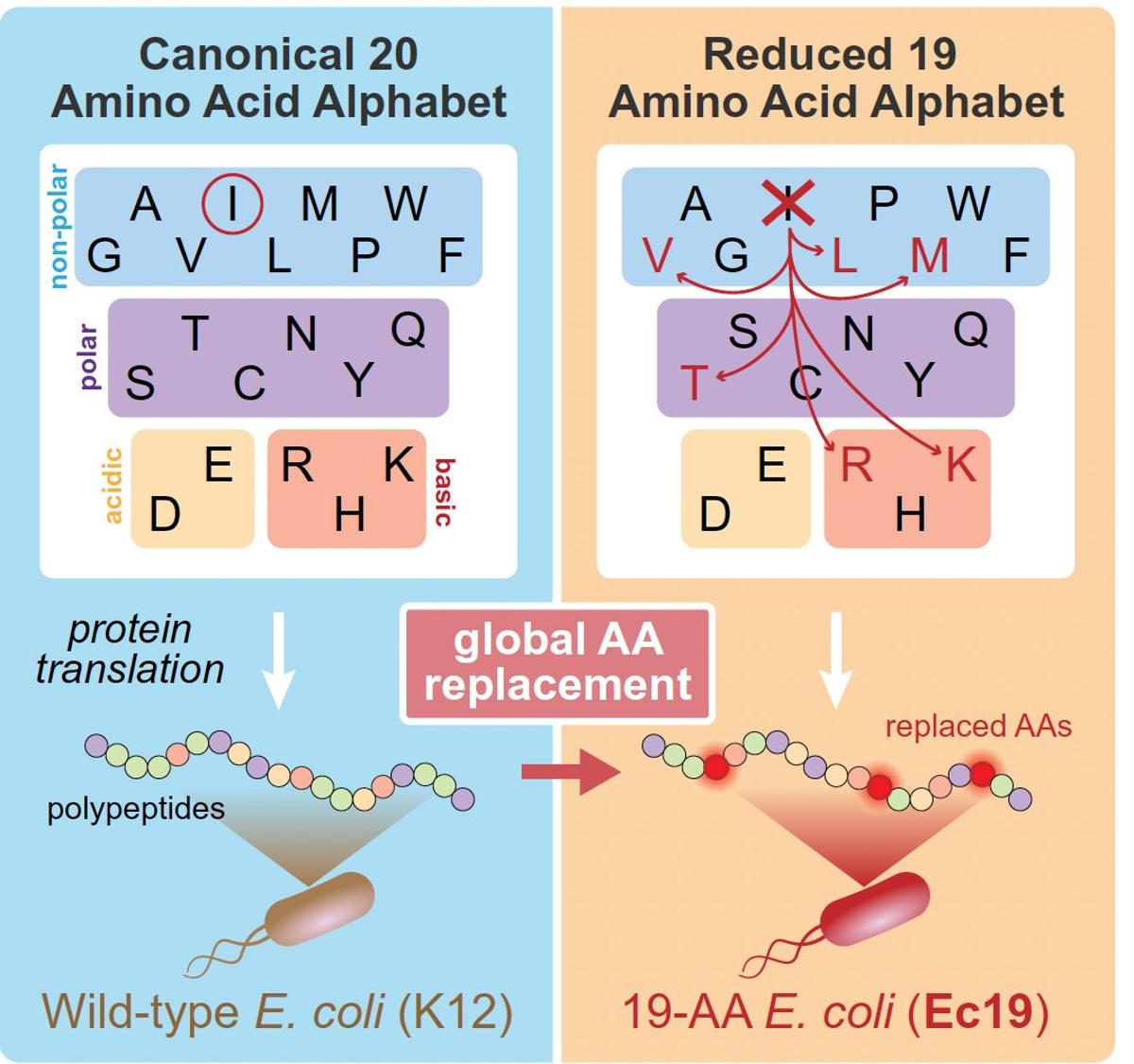
AI Designs Life Using only 19 Amino Acids
Not something you'd see everyday—changing the alphabet of life. All of life organisms are are built from 20 amino acids. Now genAI is enabling life to be built with 19 amino acids, making isoleucine dispensable. @ScienceMagazine https://t.co/7CBn0Xhuxs https://t.co/tkxtCrFx9Y

There Are Two GLP-1 Side Effects Your Doctor Doesn’t Know About, and They Can Affect Your Workouts
A new *Nature Health* study used AI to scan 400,000 Reddit posts, finding that roughly 70,000 users were taking GLP‑1 drugs such as Ozempic, Wegovy, or Zepbound. While nausea and fatigue remain the most common side effects, about 4% of...

RNA-Built Droplets Create Customizable Organelles Inside Living Cells
UCLA researchers have engineered programmable artificial organelles by assembling RNA nanostars into droplet‑like condensates inside living cells. The RNA sequences encode assembly instructions, allowing precise control over condensate size, composition, and subcellular location. Published in Nature Nanotechnology, the study demonstrates...

Interim PFS Review Confirms Study Continuation
$SMMT ivo HARMONi-3 update on squamous subgroup interim PFS analysis: "At this early interim PFS analysis reviewed exclusively by the Independent Data Monitoring Committee (iDMC), the iDMC recommended that the study continue as planned."

Gene Circuits Reshape DNA Folding and Affect How Genes Are Expressed, Study Finds
MIT researchers published in Science that the physical arrangement of genes—termed "gene syntax"—dramatically reshapes DNA supercoiling and alters transcription. Divergent gene pairs boost expression of both genes, while tandem pairs cause the upstream gene to suppress the downstream one, producing...

Drug Trials Snapshots: ELREXFIO
Pfizer’s ELREXFIO (elranatamab‑bcmm) received accelerated FDA approval on Aug 14 2023 for relapsed/refractory multiple myeloma after at least four prior therapies. The approval rests on a single‑arm trial of 187 patients, with 97 efficacy‑eligible participants showing a 57.7% objective response rate, including...

Pfizer Pushes Elrexfio Into Earlier Multiple Myeloma Line After Phase III Success
Pfizer will ask regulators to approve Elrexfio (elranatamab) as a second‑line treatment for relapsed/refractory multiple myeloma after the MagnetisMM-5 Phase III trial met its primary endpoint. The study, which enrolled 944 patients, demonstrated a statistically significant PFS advantage over J&J's...

Drug Trials Snapshots: VEOPOZ
VEOPOZ (pozelimab‑bbfg) received FDA approval on August 18, 2023 to treat CD55‑deficient protein‑losing enteropathy (CHAPLE disease) in patients of all ages. The decision was based on a single‑arm trial of ten patients enrolled in Turkey, Thailand and the United States, with a...
Abbott Secures FDA Clearance for Ultreon 3.0 AI‑Powered OCT Platform
Abbott announced FDA approval and CE Mark for its Ultreon 3.0 AI‑powered optical coherence tomography platform, enabling real‑time, high‑resolution coronary imaging during PCI procedures. The clearance removes a key regulatory barrier and positions Abbott at the forefront of AI‑driven cardiac...

Axsome Gets FDA Approval for First Non-Antipsychotic Drug to Treat Agitation Associated with Alzheimer’s Dementia
The FDA has expanded the indication for Axsome Therapeutics’ Auvelity, approving it to treat agitation associated with Alzheimer’s disease dementia. This marks the first non‑antipsychotic medication cleared for this distressing symptom. Auvelity, originally approved in 2022 for major depressive disorder,...

FDA Proposal Would Exclude Bulk Compounding of Weight-Loss Drugs Semaglutide, Tirzepatide, and Liraglutide
The FDA announced a proposal to remove semaglutide, tirzepatide and liraglutide from the 503B bulk drug list, concluding there is no clinical need for outsourcing facilities to compound these GLP‑1 weight‑loss agents from bulk substances. Because FDA‑approved versions are already...

Genomics Pioneer J. Craig Venter Dies, Leaving a Data‑Driven Legacy
J. Craig Venter, the founder of the J. Craig Venter Institute and a trailblazer in massive genomic sequencing, died on April 29, 2026. His work turned biology into a data‑rich discipline, influencing how big‑data tools are applied in life sciences. The loss...

Researchers Engineer Programmable RNA Condensates Inside Mammalian Cells
A team of biochemists has demonstrated the first programmable artificial RNA condensates in living mammalian cells using single‑stranded RNA nanostars. The condensates can be engineered to remain orthogonal, offering a modular platform for intracellular nanotechnology and synthetic organelle design.

When a Species’ Survival Hinges on Every Single Embryo
The northern white rhino is functionally extinct, with only two non‑reproductive females left. BioRescue has produced 39 embryos using frozen sperm and eggs harvested from the remaining female, Fatu, but surrogate pregnancies in southern white rhinos have failed. A new...

Intellia Raises $180 M, Files BLA After First In‑Vivo CRISPR Phase 3 Success
Intellia Therapeutics priced a $180 million public offering of 16.7 million shares and filed a rolling Biologics License Application for its hereditary angioedema candidate lonvo‑z after the drug met primary endpoints in the first global in‑vivo CRISPR Phase 3 trial. The dual milestones...

Drug Trials Snapshots: EXXUA
Fabre‑Kramer Pharmaceuticals received FDA approval for EXXUA (gepirone) on September 22, 2023 as a once‑daily oral antidepressant for adult major depressive disorder. The approval was based on two placebo‑controlled, flexible‑dose Phase III trials that enrolled 456 patients, with 442 forming the...

The Future of Biomanufacturing: Key Highlights From INTERPHEX 2026
INTERPHEX 2026 highlighted a rapid shift toward digital integration and smarter single‑use technologies in biomanufacturing. Panels emphasized automation, continuous processing, and real‑time monitoring as ways to boost scalability while tackling data‑driven control challenges. A second discussion focused on supply‑chain resilience,...

Drug Trials Snapshots: POMBILITI
The FDA approved POMBILITI (cipaglucosidase alfa‑atga) on September 28, 2023 for adult patients with late‑onset Pompe disease (LOPD) weighing ≥ 40 kg who are not responding to existing enzyme replacement therapy. Approval is based on a randomized, double‑blind trial of 123 participants across 61 sites...

ESOC 2026: What to Expect in Maastricht
The 12th European Stroke Organisation Conference (ESOC) convenes in Maastricht from May 6‑8, drawing roughly 4,200 delegates from 100 nations. Six scientific tracks will showcase the latest clinical trial data on stroke prevention, diagnosis, and treatment, including late‑breaking sessions on pre‑hospital...

AXSM Jumps 10.5% on Approval and Analyst Boost
$AXSM up over 10.5% on approval, calls hit nicely from recent write-up on this catalyst play https://t.co/H2E75g3iRB

Asembia ASX26: The Strain on the Development of Infrastructure for Sustainable C&G Therapy
Cell and gene therapies promise transformative treatments but remain hampered by steep costs across research, manufacturing, and delivery. At the Asembia ASX26 Summit, Aradigm Health CEO Will Shrank highlighted the fragmented U.S. payment landscape as a core barrier to sustainable...

FDA's Oncology Advisors Vote Against 'New Paradigm' In AstraZeneca Trial
The FDA’s oncology advisory committee voted 6‑3 that AstraZeneca’s late‑stage trial of camizestrant, an oral selective estrogen‑receptor degrader (SERD), did not demonstrate a clinically meaningful benefit. AstraZeneca had positioned camizestrant as a potential first‑in‑class oral therapy for hormone‑receptor‑positive metastatic breast...
Should Regulators Rely More on AI to Accelerate Drug Approvals?
Regulators, led by the FDA, are testing AI tools to speed drug approval processes. The agency’s pilot projects include AI‑driven real‑time monitoring of clinical trials and automated data review. Proponents argue AI can cut analysis time and uncover hidden patterns,...

Exclusive eBook: Inside the Stealthy Startup that Pitched Brainless Human Clones
MIT Technology Review released a subscriber‑only eBook exposing R3 Bio, a stealth biotech startup that pitches "brainless clones"—human bodies without brains—to serve as backup vessels for longevity seekers. The company envisions these clones as disposable shells that could host a...
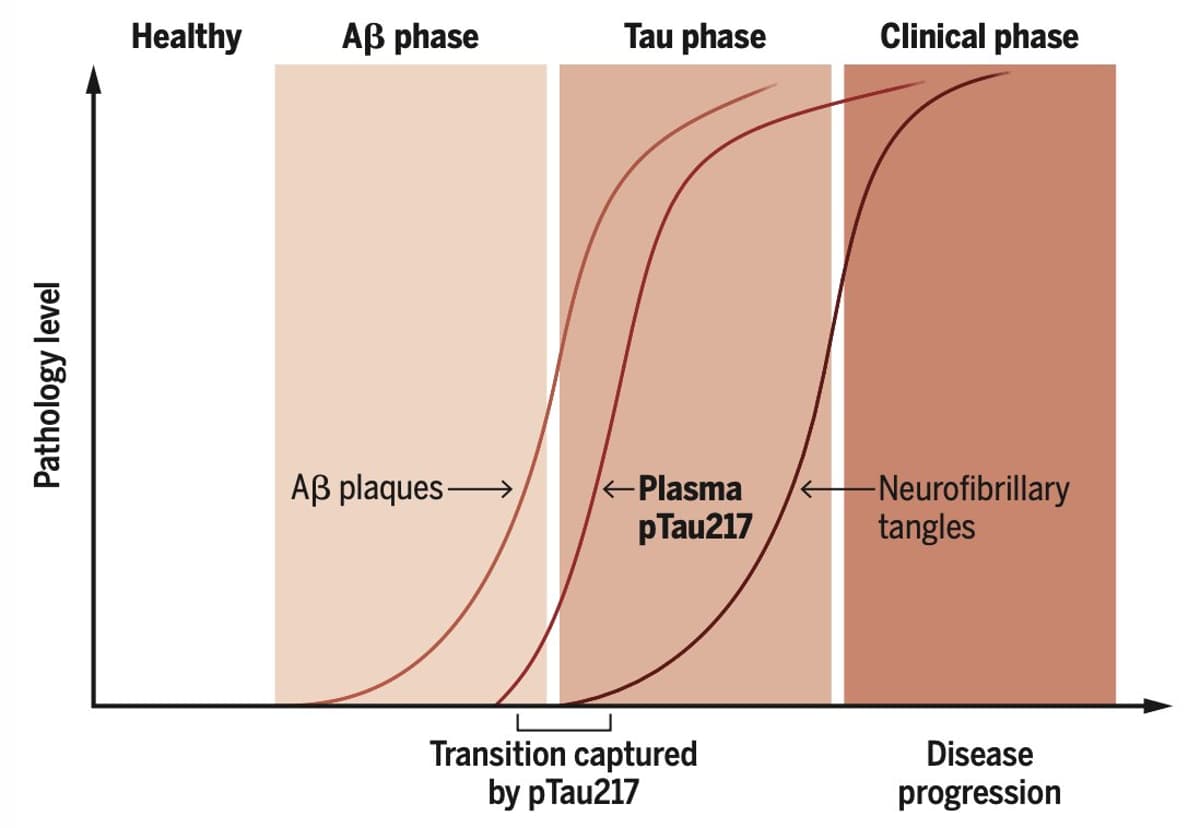
P‑tau217 Blood Test Predicts Alzheimer’s Risk Early
Good summary of p-tau217, the breakthrough blood test to predict risk of Alzheimer's in people well before onset of symptoms @ScienceMagazine https://t.co/qVJtYR4bnz https://t.co/OhjLExTroG

Genmab A/S (NASDAQ:GMAB) Upgraded at Truist Financial
Truist Financial upgraded Danish biotech Genmab A/S (NASDAQ:GMAB) to a strong‑buy, joining recent upgrades from Goldman Sachs and BNP Paribas. The stock currently trades around $26.11, well below its 52‑week high of $35.43, with a market cap of $16.78 billion and a...

Synthetic Biologist Reza Kalhor Receives $250,000 President's Innovation Award
Synthetic biologist Reza Kalhor received the $250,000 President’s Innovation Award at Johns Hopkins University, recognizing his work on genomic recording technologies that capture biological events in DNA. His approach enables scientists to trace how early‑life signals contribute to diseases such...
BIO Panel at HERS Explores Women’s Health Biotech Ecosystem
The Biotechnology Innovation Organization (BIO) convened a panel at the inaugural Health Executive and Research Summit in San Diego to spotlight the women’s health biotech ecosystem. Speakers noted that only six percent of private healthcare investment goes to women’s health,...

Novartis Finalizes US Expansion with Seventh Facility to Strengthen End-to-End Drug Manufacturing
Novartas has completed its US expansion plan by adding a seventh facility in Morrisville, North Carolina, dedicated to API production for oral solid‑dose and RNA‑based therapies. The site is part of a $23 billion multi‑year investment to build an end‑to‑end domestic...