
A Single Dose of Psilocybin Outperforms Nicotine Patches for Quitting Smoking
A Johns Hopkins pilot trial found that a single, weight‑adjusted dose of psilocybin combined with cognitive‑behavioral counseling helped 40% of smokers remain abstinent for six months, far surpassing the 10% quit rate achieved with standard nicotine patches. The psychedelic group also showed higher point‑prevalence abstinence and smoked roughly half as many cigarettes post‑treatment. Researchers reported no serious safety issues, though the study was unblinded and involved a relatively homogenous sample. The findings suggest a potentially transformative, low‑frequency pharmacotherapy for tobacco addiction.
Multiple GLP-1 Drugs Linked to Lower AFib Risk
A retrospective analysis of 13,034 patients who started GLP‑1 receptor agonist therapy between 2020 and 2024 found a significant reduction in atrial fibrillation (AFib) incidence compared with a propensity‑matched cohort of over 385,000 untreated individuals. The benefit persisted regardless of...

Asembia AXS26: What the Rise in CGTs Means for the Supply Chain
Cardinal Health’s VP Joel Wayment told Pharmaceutical Commerce that the surge in cell and gene therapies is forcing a redesign of the supply chain toward reusable, multi‑use shipping systems and geographically closed packaging models. These changes aim to cut waste,...

Cellular Rejuvenation Has the Potential to Reverse Aging
Researchers have identified a natural cellular rejuvenation process that resets embryonic cells to a youthful state within two weeks, effectively erasing parental age markers. Over the past two decades, labs have revived skin cells from 90‑year‑olds and rejuvenated diseased mice,...

FDA Clears Subcutaneous Anifrolumab Autoinjector for Moderate to Severe SLE
The FDA has cleared AstraZeneca’s subcutaneous anifrolumab autoinjector (Saphnelo) for adults with moderate to severe systemic lupus erythematosus (SLE) on standard therapy. The new formulation mirrors the approved intravenous product but allows weekly self‑administration at home. Approval rests on the...
FDA Priority Review Advances Nipocalimab for Adults With Warm Autoimmune Hemolytic Anemia
The FDA has granted priority review to Johnson & Johnson’s nipocalimab‑aahu (IMAAVY) for adults with warm autoimmune hemolytic anemia (wAIHA), a rare disease lacking any approved U.S. therapy. The designation compresses the review clock to roughly six months, marking the...

UT Southwestern Study Links Refeeding Phase to 41% Lifespan Boost in Worms
UT Southwestern scientists discovered that the metabolic response to refeeding after a 24‑hour fast extends Caenorhabditis elegans lifespan by about 41%, highlighting the refeeding phase—not the fast itself—as the key driver of longevity. The finding could reshape human dietary recommendations...

STAT+: Astellas Retries XLMTM Gene Therapy After Deaths
Astellas Pharma announced it will restart its next‑generation gene therapy trial for X‑linked myotubular myopathy (XLMTM) after pausing the program following two patient deaths. Meanwhile, Intellia Therapeutics reported its one‑time CRISPR treatment lonvo‑z reduced hereditary angioedema attacks by 87% in...

Pilot Study Finds 300 Mg NMN Cuts Post‑Exercise Inflammation in Young Men
Researchers in Taiwan reported that a six‑day regimen of 300 mg NMN lowered key inflammatory cytokines in young men after intense blood‑flow‑restriction resistance training. The crossover trial suggests NMN could become a targeted supplement for athletes and biohackers seeking faster recovery.

STAT+: Pharmalittle: We’re Reading About a Lilly Deal, an Intellia CRISPR Rare-Disease Treatment, and More
Intellia Therapeutics reported that a single dose of its CRISPR‑based therapy lonvo‑z dramatically reduced swelling attacks in hereditary angioedema patients during a Phase 3 trial, positioning it for a rolling FDA submission as the potential second approved CRISPR drug. The treatment...
Fragments vs DsbA: Towards a Chemical Probe
Researchers targeting the bacterial oxidoreductase DsbA—a key virulence factor—have advanced fragment‑based efforts toward a chemical probe. Initial screens identified fragments binding a shallow groove and a hidden cryptic pocket, but affinities were modest (~150 µM). By designing molecules that extend beyond...
FDA Review Sought for Subcutaneous Risankizumab Induction in Crohn Disease
AbbVie has filed a supplemental NDA with the FDA to add subcutaneous (SC) risankizumab induction for adults with moderate‑to‑severe Crohn disease. The request relies on the phase 3 AFFIRM trial, which enrolled 289 patients—65% of whom had failed prior advanced therapies—and...

FDA Clears Tandem Control-IQ+ for Type 1 Diabetes During Pregnancy: First AID System to Earn Indication
The FDA has cleared Tandem Diabetes Care’s Control‑IQ+ system for use in pregnancies complicated by type 1 diabetes, making it the first automated insulin‑delivery (AID) platform with a pregnancy indication in the United States. The clearance covers both the t:slim X2 pump...

Dartmouth Researchers Launch Smartphone Study to Predict Alzheimer’s Risk in Williamstown Seniors
Dartmouth Medical School researchers began a pilot study with 23 Williamstown seniors, part of a nationwide 200‑person trial, to test the RealVision smartphone app that analyzes walking, speech, eye‑tracking and smiling to flag early Alzheimer’s risk. The effort showcases big‑data...

WuXi AppTec Q1 Profit Jumps 31% on Booming CRO and Manufacturing Demand
WuXi AppTec posted a first‑quarter profit of RMB4.651 billion ($651 million), up 31% year‑over‑year, and revenue of RMB12.435 billion ($1.74 billion), a 28.8% increase, driven by expanding contract research and manufacturing work for biotech firms worldwide.
Sun Pharma to Acquire Organon for $11.75 Billion, Expanding B2B Drug Portfolio
Sun Pharmaceutical Industries has signed a definitive agreement to acquire Organon & Co. for $11.75 billion in cash, paying $14 per share. The deal will lift combined revenue to $12.4 billion, push Sun into the top three global women’s‑health players and make...

Gilead’s Pipeline Strategy in 2026: A Broadening Focus Beyond HIV
Gilead Sciences is accelerating a diversification push in 2026, adding $12.6 billion of oncology and immunology assets to a portfolio still dominated by HIV drugs. The company completed three major acquisitions—Arcellx for $7.8 billion, Tubulis for $3.15 billion, and Ouro Medicines for $1.67 billion—bolstering...
CureVac Takes Moderna to Court over mRNA Vaccine Patents
CureVac has filed a lawsuit against Moderna alleging infringement of its mRNA‑stabilisation and delivery patents that underpin Spikevax. The case, backed by BioNTech after its 2025 acquisition of CureVac, seeks a share of the multibillion‑dollar revenues from COVID‑19 mRNA vaccines....

FDA’s Need for Speed Could Strain Small Biotechs. Here’s How They Can Keep Up.
The FDA is accelerating drug approvals through the National Priority Voucher pilot, which can shrink review timelines from ten months to as little as two, and a new plausible mechanism pathway for niche therapies lacking large trial data. While the...
Unverified Tips Can Mislead: False Compass Data
Interesting side note on today's disappointing $CMPX results: Yesterday, I received a message on Signal from someone claiming to be a Compass employee. He gave me the purported tovecimig PFS and OS data. I didn't do anything with the supposedly material...
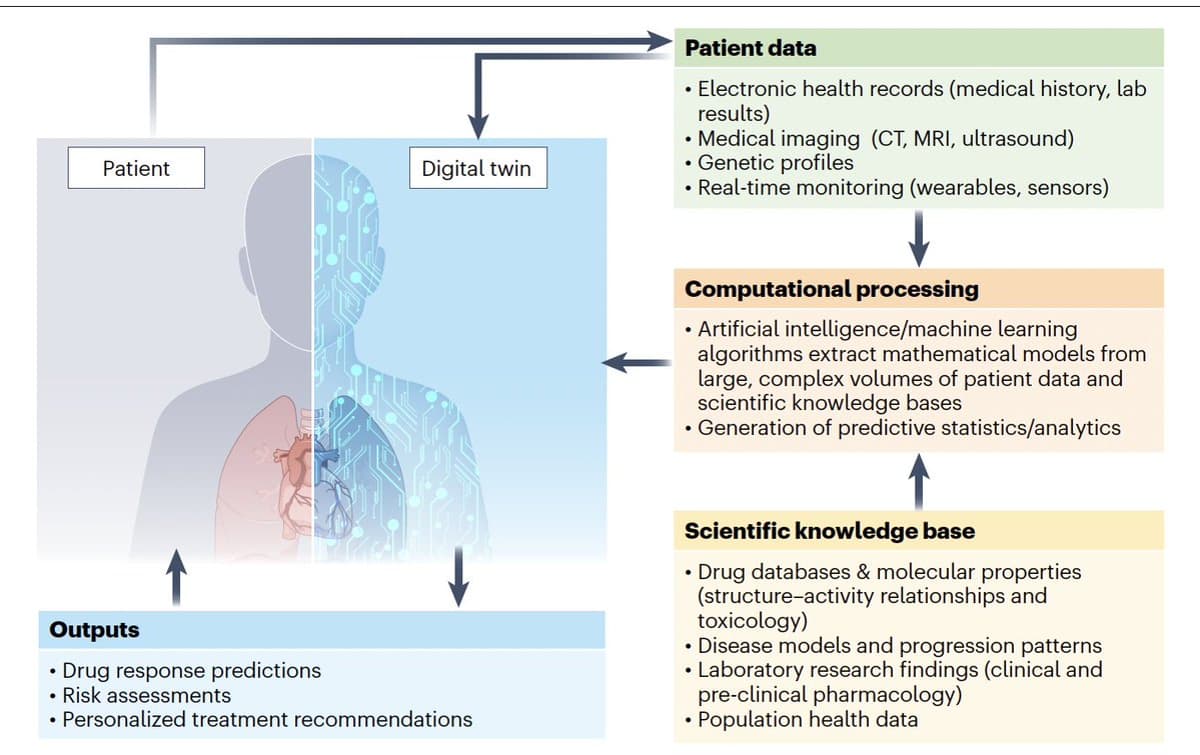
Digital Twins Will Accelerate Drug Development via Virtual Trials
How digital twins and in silico clinical trials can change the future of drug development https://t.co/lEhJNwrSlH

Motif Neurotech Receives FDA IDE Approval to Initiate RESONATE Trial of Motif XCS System in Treatment-Resistant Depression
Motif Neurotech has secured FDA Investigational Device Exemption (IDE) to launch the RESONATE early feasibility study of its Motif XCS System in patients with treatment‑resistant depression who have failed at least two medications. The trial will monitor 12‑month safety, symptom...

ERAS Pan‑Ras Data Released; RVMD Alleges Patent Infringement
$ERAS pan-Ras data update this afternoon after market close. also, $RVMD sent Erasca a letter claiming ERAS-0015 violates certain RevMed patents. https://t.co/QTmyUhT89n

Don't Reinvent Broken Wheels—Use Existing Bioinformatics Tools
1/ Bioinformaticians: Before you heroically code your own method... STOP. You might be about to reinvent a very broken wheel. https://t.co/c4FT7VXeM6
GSK/Tesaro Claim Denied in Jemperli Lawsuit with AnaptysBio
A Delaware court rejected GSK and its Tesaro unit's attempt to terminate the 2014 licensing agreement with AnaptysBio, preserving the existing royalty rate for the PD‑1 cancer drug Jemperli. The ruling affirms that AnaptysBio has not breached the contract, while...

T‑cell Engagers Need Missing Design Tricks for Success
With the rise and rise of ADCs, T cell engagers have taken something of a back seat of late. What if the bigger problem is some of them may be missing a key trick? Here we highlight some design...

NTLA's HAE Results Fall Short, Funding Needed for Weak Launch
$NTLA HAE study results were both expected and unimpressive relative to current treatments. The company will need to raise a lot of money to sell a therapy that is not going to launch well.
STAT+: Oruka’s Long-Acting Psoriasis Therapy Posts Strong Results in Mid-Stage Study
Oruka Therapeutics reported that its long‑acting injectable, ORKA‑001, achieved complete skin clearance in 63% of plaque‑psoriasis patients during a mid‑stage trial. The data suggest the drug can be administered just once per year, a stark contrast to current biologics that...

Biosimilar T
Me writing scripts to completely control 90% of my psoriasis patients with biosimilar Taltz >2 years before $ORKA even reads out P3: https://t.co/TVaA10pHOe
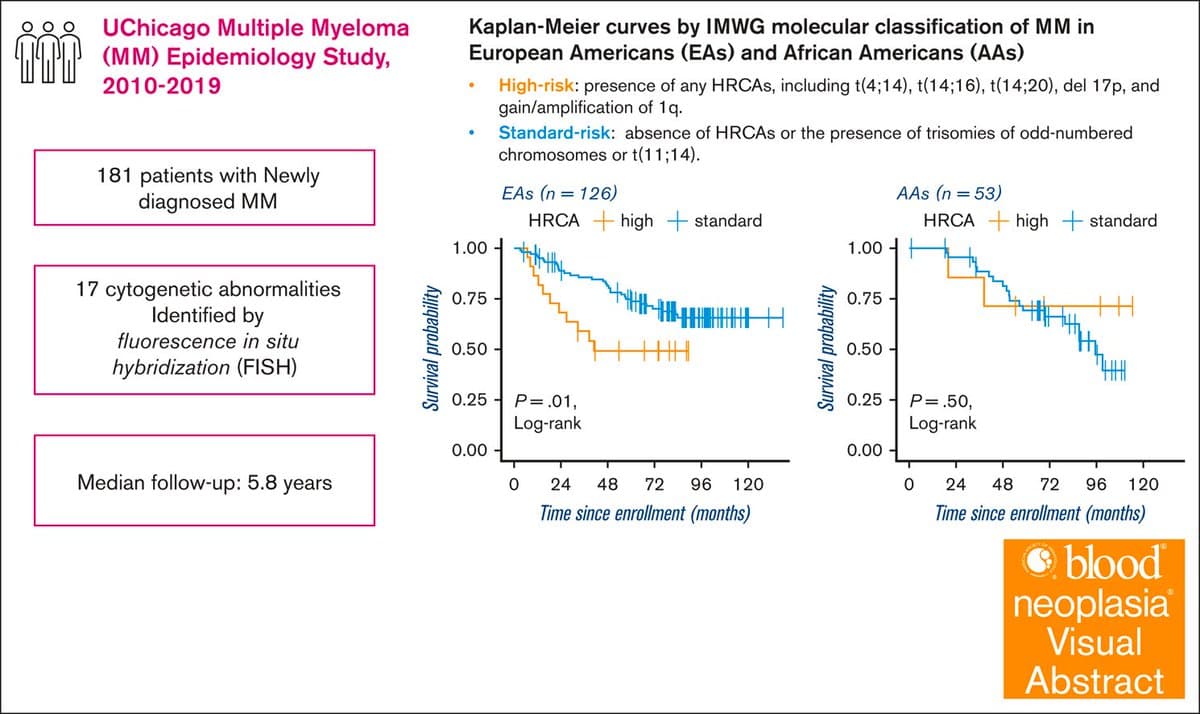
Chromosomal Abnormalities Impact Myeloma Survival Differently Across Populations
Population differences in the associations between chromosomal abnormalities and overall survival of multiple myeloma [Jan 31, 2025] Bei Wang et al. @Bloodneoplasia https://t.co/IFPgCwpGo6 #mmsm #PrecisionMedicine #cancerdisparities https://t.co/vpNoEupRPG

STAT+: Veradermics’ Hair Loss Drug Succeeds in Late-Stage Trial
Veradermics announced that its oral hair loss drug VDPHL01 met primary endpoints in a Phase III trial. Over six months, participants taking the pill grew 30‑33 hairs per square centimeter versus seven in the placebo group. Patient‑reported improvement reached 79‑86%, and...

Fathom Therapeutics Secures $47M Series A for AI‑Physics Drug Discovery
Exclusive: Fathom Therapeutics, formerly Atommap, has raised a $47M Series A, building a pipeline with its strategy of blending AI and physics approaches to drug discovery: https://t.co/iAlmJwCQK7
CRISPR Therapy Cuts Swelling Attacks 87%, 60% Attack‑Free
$NTLA Intellia says CRISPR-based treatment for rare disease reduced swelling attacks in pivotal trial Attack rates dropped 87%, relative to placebo. Just over 60% were entirely attack-free over the period, compared to 11% for the placebo patients. https://t.co/ljPC7Lww0D via @Jasonmmast
Zepbound’s and Ozempic’s Greatest Benefit May Be Their Anti-Inflammatory Power
GLP‑1 drugs such as Ozempic and Zepbound are gaining recognition for anti‑inflammatory effects that go beyond weight loss and glucose control. Clinical data show semaglutide reduces C‑reactive protein by about 40% independent of weight loss and improves liver inflammation in...
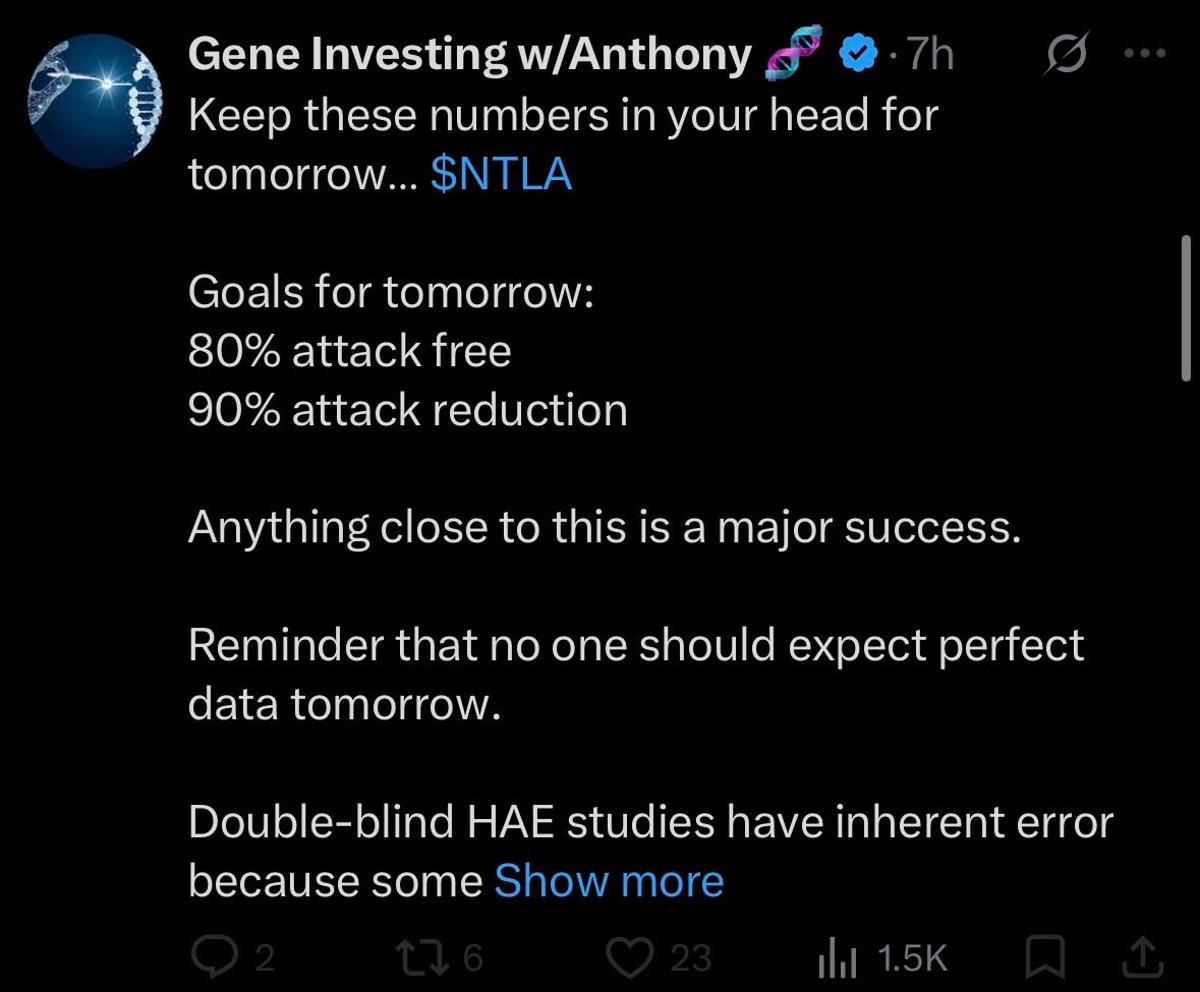
NTLA's Attack Metrics Fall Short of Expectations
$NTLA 87% attack reduction and only 62% attack free rate. Attack free rate especially widely missed the bar set ahead of time by @GeneInvesting. Legitimately worse than I expected. https://t.co/wmNFYfsQSj
Oruka's Long-Acting Psoriasis Drug Achieves 63% PASI‑100
$ORKA Oruka’s long-acting psoriasis therapy posts strong results in mid-stage study 63% PASI 100 score at 16 weeks, very strong data. https://t.co/RJxnbuU0e3 via me

Ascletis Completes Enrollment in U.S. Phase‑II Trial of Oral GLP‑1 Agonist ASC30
Ascletis Pharma announced that its 13‑week U.S. Phase‑II trial of ASC30, an oral small‑molecule GLP‑1 receptor agonist for type‑2 diabetes, has enrolled 100 participants. The company expects topline results in the third quarter of 2026, a milestone that could reshape...

Veradermics' Hair Loss Drug Clears Late-Stage Trial
Ok, here we go... $MANE Veradermics' hair loss drug succeeds in late-stage trial. A report from @ADeAngelis_bio https://t.co/jtSB36sPGO

Can We Truly Deliver Gene Therapies to Patients Without Solving the Cost and Scale Challenges of AAV Production?
Gene‑therapy developers are confronting a manufacturing bottleneck as adeno‑associated virus (AAV) vectors must be produced at commercial scale. FUJIFILM Biotechnologies proposes 2,000‑liter single‑use bioreactors combined with an integrated CDMO model to boost yield, cut cost per dose, and preserve product...

AstraZeneca Reports FDA Approval of Saphnelo for Systemic Lupus Erythematosus (SLE)
The U.S. Food and Drug Administration has granted approval for AstraZeneca’s Saphnelo autoinjector (anifrolumab, 120 mg weekly) for adult patients with systemic lupus erythematosus (SLE) when used alongside standard of care. The decision follows the Phase III TULIP‑SC trial, which enrolled 367...

Novartis Secures Pair of Regulatory Wins for Skin Disease and Malaria Treatments
Novartis won two regulatory milestones: the European Commission approved its oral BTK inhibitor Rhapsido for adults with chronic spontaneous urticaria who have failed antihistamines, and the World Health Organization granted prequalification to Coartem Baby, an artemether‑lumefantrine formulation for infants weighing 2‑5 kg....
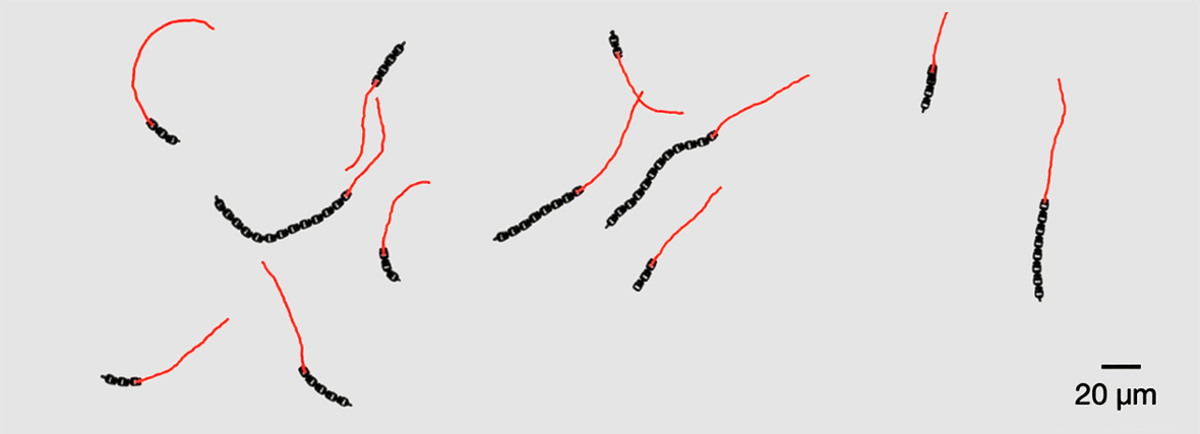
Leiden’s Sensor-Free Microrobots Move Like Living Organisms
Researchers at Leiden University have created soft, chain‑like microrobots that move and adapt without sensors, software, or external control. Each 5 µm segment is linked by 0.5 µm joints and powered solely by an electric field, allowing the robot’s shape to dictate...

Longevity Medicine’s New Vanguard: 11 Innovators Poised to Drive an $8 Trillion Market
Business Insider unveiled its 2026 Rising Stars of Longevity, naming 11 innovators whose AI‑driven diagnostics, biotech breakthroughs and gerotherapeutics aim to capture a market expected to grow to $8 trillion by 2030. The list, vetted by experts like Andrea Maier and...

Key Insights Await From Tovecimig Companion-002 Data
$CMPX data from Companion-002 study of tovecimig coming before 8am ET today. What to look out for, via @ByMadeleineA -> https://t.co/6IDCqMyNPE
Evidence for MLKL to Be Important in Hematopoietic Stem Cell Aging
Researchers have identified the RIPK3‑MLKL signaling axis as a central driver of hematopoietic stem cell (HSC) aging. Activated MLKL accumulates in HSC mitochondria, impairing self‑renewal and lymphoid differentiation without causing necroptotic cell death. The study links multiple stress responses—such as...
Reviewing the Inability of Anti-Amyloid Immunotherapies to Affect Alzheimer's Disease
A recent Cochrane meta‑analysis of ten anti‑amyloid monoclonal antibodies—including aducanumab, lecanemab and donanemab—shows only trivial cognitive gains and modest functional improvement in patients with mild Alzheimer’s disease, despite clear plaque clearance. The studies also reveal an elevated risk of amyloid‑related...

New NIHR-Funded TRC for Parkinson’s Disease
The National Institute for Health and Care Research (NIHR) has launched the Parkinson’s disease Translational Research Collaboration (PD‑TRC), the first of eight UK TRCs dedicated to Parkinson’s. Backed by NIHR and four major charities, the hub links 17 centres of...

Lilly Falls on Slower Start for Foundayo versus Wegovy
Eli Lilly’s oral GLP‑1 agonist Foundayo launched in early April with modest uptake, recording 1,390 prescriptions in week 1 and 3,707 in week 2, far below Novo Nordisk’s oral Wegovy, which logged over 3,000 fills in its first days and 18,410 in the second...

Sun Pharma to Acquire Organon for $11.75 B, Boosting Women’s Health and Biosimilars
Sun Pharmaceutical Industries announced an all‑cash deal to acquire Organon & Co. for $14 per share, valuing the U.S. firm at $11.75 billion. The merger will lift Sun Pharma into the top‑25 global drugmakers, give it a top‑3 position in women’s...
UK Biotech Could Be Bound for Recovery, BIA Report Reveals
UK biotech venture financing rebounded in Q1 2026, with total equity raised climbing 18% to £552 m ($746 m). Venture‑capital inflows also rose 17% to £516 m ($699 m), and 25 companies secured funding, up from 15 a year earlier. While public‑market activity stayed flat—no...