Know What's Happening in BioTech
Know What's Happening in BioTech
BioTech Pulse
EMAIL DIGESTS
Daily
Every morning
Weekly
Tuesday recap

The FDA has rejected a pediatric myopia drug, citing a lack of substantial evidence for its efficacy. Pediatric ophthalmologists argue the drug, already used in compounded form, effectively slows myopia progression in children. The decision follows a series of recent, unexpected FDA rejections of other therapies, prompting clinicians to call for a reversal. The controversy highlights tensions between regulatory standards and real‑world clinical practice.

The U.S. Food and Drug Administration has reversed its earlier decision to decline a review of Moderna's mRNA influenza vaccine candidate. Following a Type A meeting with the company, the agency set an August 5, 2026 deadline for completing its evaluation. The...

Researchers have engineered a wet‑adhesion Janus hydrogel (WAJH) that mimics oral mucosa to treat diabetic oral ulcers. The hydrogel features an antifouling agar/polyacrylamide layer and a tannic‑acid‑rich adhesive layer, delivering adhesion energies of 15 J m⁻² and 316 J m⁻² respectively. Its antioxidant tannic...

The FDA and UTMB are hosting a free, week‑long training course on data quality and integrity for BSL‑4 laboratories from July 28 to August 1, 2025, with in‑person seats in Manhattan, Kansas and a virtual option. The curriculum covers GLP requirements, Animal Rule...

Researchers discovered that elevated circulating APOE in older mice suppresses bone regeneration by inhibiting osteoblast differentiation. Liver‑specific knockout of APOE or a single dose of a neutralizing antibody lowered serum APOE, restored Wnt/β‑catenin signaling, and markedly improved fracture callus density...

The NewCo model, where Asian biotech assets are spun out into Western start‑ups backed by Western capital, surged in 2025 with 13 new companies—more than double the six disclosed in 2024. Early 2026 already sees three additional launches, including Kidswell...
The FDA has reversed its earlier refusal and will now evaluate Moderna’s mRNA‑1010 seasonal flu vaccine using a two‑track approach: a full Biologics License Application review for adults 50‑64 and an accelerated approval pathway for those 65 and older, contingent...

The nova‑Institut report shows bio‑based polymers reached 4.5 million tonnes in 2025, representing about 1 % of total polymer output. Production is projected to grow at an 11 % compound annual growth rate through 2030, potentially doubling the market share to 2 %. Capacity...

Aceclidine (Vizz®) received FDA approval in 2025 as an ophthalmic solution for presbyopia, targeting age‑related near‑vision loss. The drug acts as a pupil‑selective muscarinic agonist, inducing miosis without significant ciliary muscle activity, thereby enhancing depth of focus through a pinhole...

The National University of Singapore Academy for Healthy Longevity is hosting the Geromedicine Conference on February 26-27, 2026, bringing together geroscience researchers, clinicians, and industry leaders. The event emphasizes clinical translation of molecules such as NAD+ precursors, urolithin A, and ergothioneine,...

Eli Lilly has agreed to pay CSL Ltd. $100 million to license the monoclonal antibody clazakizumab, which targets interleukin‑6. Under the agreement, CSL will keep development rights for the drug’s use in preventing cardiovascular complications in end‑stage kidney disease patients, currently...

Rapid functional testing is turning ambiguous genetic variants into actionable medical decisions. A zebrafish model proved a newborn‑identified SMN1 mutation benign, allowing clinicians to defer costly SMA therapy. This proof‑of‑concept shows whole‑genome sequencing can move from data to diagnosis within...

The FDA’s Center for Drug Evaluation and Research created the Office of Pharmaceutical Quality (OPQ) to standardize drug quality oversight across all manufacturing sites, both domestic and foreign. OPQ’s mandate spans new drugs, biologics, generics, biosimilars, over‑the‑counter products, and certain...

The FDA has accepted Bristol Myers Squibb’s protein‑degrading multiple myeloma candidate iberdomide for review, with a decision expected by Aug. 17 after a Phase 3 trial showed higher minimal residual disease‑negative rates. In contrast, the agency rejected Disc Medicine’s accelerated‑approval bid for...

Pharma launches are missing the mark 58% of the time, a failure rate driven by poor market insight and fragmented decision‑making. Accelerating market‑access planning from pre‑Phase 2 and integrating real‑world data can give companies a strategic north star. Purpose‑built AI tools...

Researchers at the Salk Institute demonstrated that the dynamic three‑dimensional folding of the genome directly influences gene expression and cell identity. By depleting the cohesin‑loader protein NIPBL in human iPSC‑derived neurons and cardiomyocytes, they observed region‑specific unfolding of chromatin loops....

The FDA initially issued a refuse‑to‑file letter for Moderna’s mRNA influenza vaccine, then reversed course within a week and agreed to review the product. The agency will now consider two separate pathways: full approval for adults 50‑64 and accelerated approval...

Vision loss remains a leading disability, prompting biotech firms to shift from symptom‑management to disease‑modifying therapies. Gene‑replacement approaches using AAV vectors dominate the inherited retinal disease pipeline, highlighted by Luxturna’s success and multiple late‑stage candidates such as Beacon’s laru‑zova and...

A wave of clinical trials is testing stem‑cell, gene‑editing and bionic‑eye technologies to treat blindness, especially retinitis pigmentosa and dry age‑related macular degeneration. Early‑stage studies from UC Davis, BlueRock Therapeutics and the University of Michigan report measurable vision gains, such as...

Cryogenic electron microscopy enabled researchers to solve five structures of Vibrio cholerae transcription activation complexes, revealing how the bacterium initiates virulence in the human gut. The study shows ToxR and TcpP transcription factors bind the RNA polymerase α‑C‑terminal domain via...

Professor Segun Fatumo highlights the stark under‑representation of African genomes in global research, where over 86% of GWAS participants are of European ancestry despite Africa housing the greatest genetic diversity. He explains how this gap limits the accuracy of polygenic...

Researchers identified interferon regulatory factor 7 (IRF7) as a master transcriptional driver that pushes vascular smooth muscle cells (SMCs) into a pro‑inflammatory, macrophage‑like state, a key step in plaque destabilisation. Single‑cell RNA sequencing and trajectory analysis uncovered an intermediate stem‑endothelial‑monocyte...
Novartis announced that its oral BTK inhibitor Rhapsido (remibrutinib) achieved significantly higher complete response rates than placebo in the phase 3 RemIND trial for the three most common forms of chronic inducible urticaria (CINDU). The drug, already approved for chronic spontaneous...

A “Variant of Unknown Significance” is a genetic finding that can’t yet be interpreted, leaving families with uncertainty instead of answers. Now, AI agents can help connect sequencing data with scientific literature and biological context to turn VUS into traceable, evidence-backed...

What's ruffling? Large veins interfacing the skull (venous sinuses) that patrol immune cells and help drain the brain https://t.co/WsML3Jd1yA
Korsana Biosciences announced a $175 million financing round led by Fairmount, Venrock, Wellington Management and TCGX to develop its Alzheimer’s antibody candidate KRSA‑028. The startup positions KRSA‑028 as a next‑generation “brain shuttle” designed to clear amyloid plaques more efficiently than existing...

FDA chief Marty Makary says 'everything should be over the counter' unless drug is unsafe or addictive https://t.co/HiYBuxvyWC

Upcoming meeting of federal vaccine panel may be postponed after HHS missed deadlines https://t.co/zfAl7owoQg via @statnews

Carterra unveiled Vega, the industry’s first 48‑channel high‑throughput SPR platform, delivering roughly 12‑fold higher screening capacity for small‑ and large‑molecule drug candidates. Covaris introduced the truCOVER® Total RNA Library Prep Kit, enabling robust RNA‑seq libraries from as little as 10 ng...

The unfounded move by @HHSGov against mRNA vaccines will hurt our future potent immune therapy vs cancer. Another point of progress for triple-negative breast cancer with individualized neoantigen mRNA vaccines today @Nature Adds to successful pancreatic, renal cell, melanoma reports...
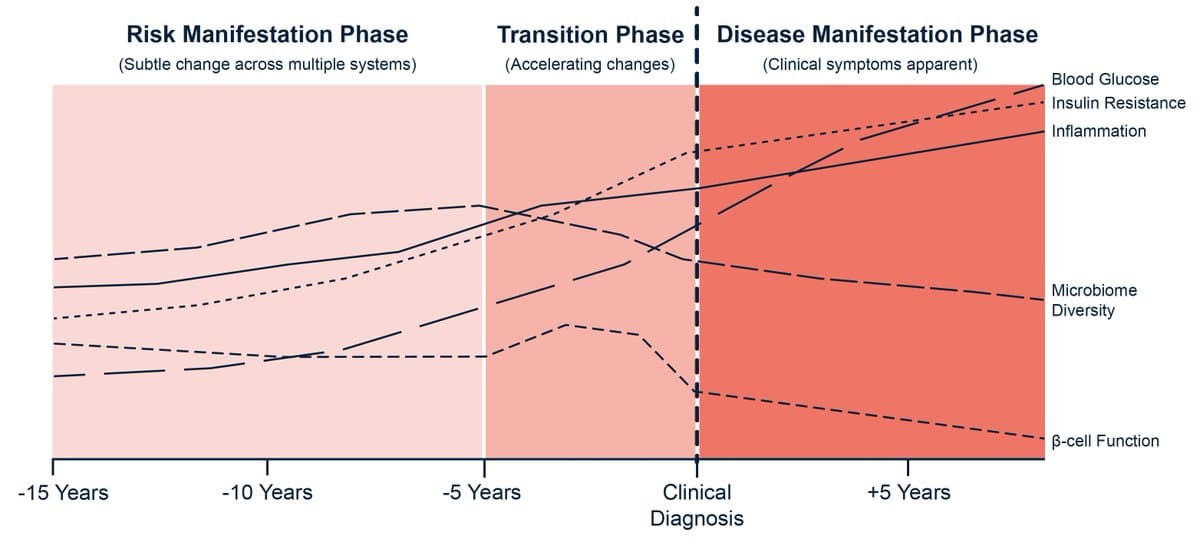
We could prevent diseases so much better but we haven't learned to listen to when biology whispers long (10 + years) in advance. A key theme in Super Agers, too. https://t.co/0pSap3f32N @NathanPriceSci @ @nrappapo @BuckInstitute example below for Type 2...

Immedica’s pegzilarginase (Loargys), the first enzyme replacement therapy for arginase‑1 (ARG1) deficiency, has received NICE endorsement for NHS use in the UK. The weekly IV or subcutaneous treatment cuts blood arginine levels by roughly 80% and is recommended for patients...
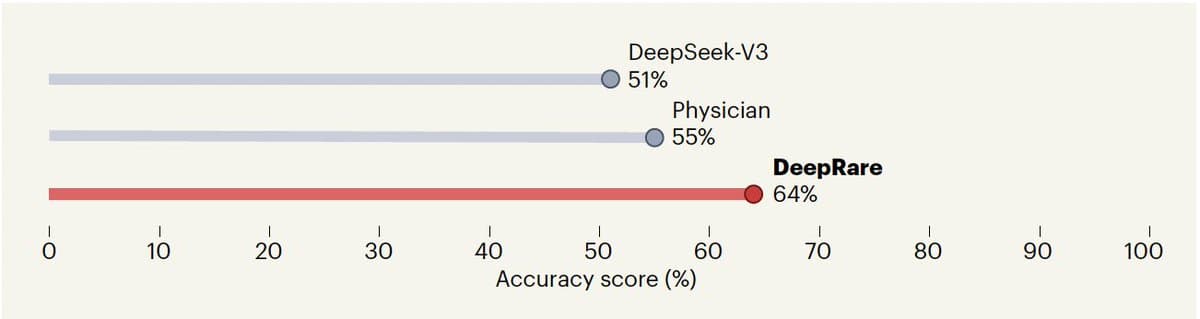
Another good use case for agentic AI in medicine: diagnosis of rare diseases. Exemplified by DeepRare https://t.co/XBd68V3iDl https://t.co/rBl5yn95sI
The pharmaceutical industry needs stability & clear guidance from #FDA as it attempts to bring through licensure new drugs & vaccines. Vinay Prasad, FDA's head of biologics, has brought drama to a setting where it's not welcome, @matthewherper.bsky.social writes. https://t.co/Qaw1QmYNpL

The UK government, via Innovate UK, has launched a £20 million funding programme to accelerate development of medicines, medical devices, wearables, virtual‑reality therapies and AI‑enabled tools for drug and alcohol addiction. Grants of up to £10 million for late‑stage projects and up to...

The FDA keeps spinning, spinning, spinning round and round. Just what you want from a drug regulator: Erratic decision-making and complete unpredictability. https://t.co/q5abiPwgkW

🧵 That V-plot showing transcription factor binding? It might be an artifact. New research shows chromatin fragmentation methods create patterns even on naked DNA. Here's what went wrong. https://t.co/79B5c0ulgD

Portal Diabetes has secured FDA breakthrough device designation for its implantable Portal Pump, an insulin delivery system that pairs an abdominal pump with continuous glucose monitoring and concentrated insulin. The company also launched a Phase I study of temperature‑stable Portal Insulin...

1/ Claude Code just hallucinated ENSEMBL IDs for my volcano plot. (it happened last week for real) I asked it to highlight specific genes. The data matrix uses ENSEMBL identifiers. Instead of flagging that it couldn't map the gene symbols, it invented...
🧵 10 free bioinformatics tools you should know in 2026. These will save you time, money, and headaches. https://t.co/t8amdLE4MX

Janux Therapeutics has dosed the first participant in a Phase I, open‑label, dose‑escalation trial of JANX011, its inaugural candidate from the adaptive immune response modulator (ARM) platform. The bispecific antibody targets CD19‑expressing B cells to achieve a deep, lasting immune reset...

The FDA’s Moderna $MRNA pirouette is one more sign of chaos at the agency https://t.co/zThJqhT2BS via @matthewherper

#FDA backs down on #Moderna: The agency had refused to review the company's licensure application for an mRNA flu vaccine, but after significant pushback has relented. https://t.co/omo7bEBQGW

Johnson & Johnson received FDA approval for a once‑monthly subcutaneous regimen of Rybrevant Faspro combined with Lazertinib for first‑line treatment of advanced EGFR‑mutated non‑small cell lung cancer. The new schedule delivers clinical outcomes comparable to the earlier bi‑weekly dosing while preserving...

$MRNA says FDA will now review its flu vaccine. PDUFA date is Aug. 5. https://t.co/npZzeEa5nk

FDA agrees to review Moderna flu vaccine application, splitting review into standard approval for those under 65 and accelerated approval for those 65 and above. $mrna https://t.co/HMMlPm0PGh

Barcelona is emerging as a premier European life‑sciences hub, contributing 7.6 % of Catalonia’s GDP and ranking sixth in scientific output. Six local biotech firms—Accure Therapeutics, Oryzon Genomics, SpliceBio, Peptomyc, Ona Therapeutics and Integra Therapeutics—are advancing diverse modalities from small‑molecule neuroprotectors...

Researchers published in Nature Communications that iron deficiency triggers a selective loss of mature pancreatic β‑cells while sparing immature cells. Using lineage tracing and single‑cell transcriptomics, the team showed that iron scarcity impairs mitochondrial function, elevates ROS, and down‑regulates key...

Researchers at KAIST unveiled an AI‑driven method that extracts wavelength‑dependent information from standard optical coherence tomography (OCT) to automatically detect lipid‑rich coronary plaques. The weakly supervised deep‑learning model learns from frame‑level labels, eliminating the need for pixel‑wise annotations and works...
UCLA researchers have engineered lipid nanoparticles to co‑deliver CRISPR/Cas9 components and a full‑length CFTR gene, achieving precise, mutation‑agnostic insertion in human airway cells. The non‑viral system corrected 3‑4% of cells yet restored up to 100% of normal chloride channel function,...