Know What's Happening in BioTech
Know What's Happening in BioTech
BioTech Pulse
EMAIL DIGESTS
Daily
Every morning
Weekly
Tuesday recap

Researchers at Texas A&M have created injectable nanocomposite hemostats that cut blood clotting time from six‑seven minutes to one‑two minutes, slashing bleeding duration by up to 70% in internal hemorrhage models. The devices combine clay‑derived nanosilicates with a shape‑memory foam or micro‑ribbon matrix that expands or curls upon exposure to body heat, sealing wounds without dispersing particles. Funded by the DoD and NSF, the technology targets non‑compressible bleeding where traditional compression is impossible. The goal is a self‑applicable, field‑ready dressing that can extend the critical "golden hour" after trauma.

Researchers at St. Jude Children’s Research Hospital introduced M‑PACT, an AI‑driven liquid‑biopsy platform that classifies pediatric brain tumors from cerebrospinal fluid with 92% accuracy. The deep neural network was trained on over 5,000 DNA methylation profiles covering about 100 tumor...

Scientists used array tomography to reconstruct podocytes in three dimensions, revealing how these non‑regenerative kidney cells adapt to aging. In rats, podocyte numbers fell while surviving cells expanded 4.6‑fold, forming autocellular junctions and exporting waste extracellularly. The study provides the...

Biopharma senior leaders are increasingly shouldering operational work left behind by downsizing, stretching their capacity beyond strategic duties. The overload fragments focus, slows decisions, and erodes communication, while burnout remains hidden behind continued presence. As pressure builds, the organization’s decision‑making...

Peptris, a Bengaluru‑based AI drug‑discovery startup, closed a Rs 70 crore ($7.7 million) Series A round co‑led by IAN Alpha Fund and Speciale Invest. The capital will accelerate its pipeline, moving novel chemical entities and repurposing programs toward clinical readiness within 24 months. Peptris’ platform...

Venture capital in biopharma rebounded in 2025, delivering $33.8 billion across 1,171 deals, but the capital is increasingly concentrated in late‑stage companies. Early‑stage startups received only 32% of the funding, down from over 40% during the pandemic surge, while median deal...

Regulatory uncertainty is rising as the FDA shifts leadership, adopts a single‑trial approval pathway, and tightens its benefit‑risk framework. While the one‑trial standard promises faster, cheaper market entry, recent surprise complete response letters (CRLs) show that even successful Phase 3 results...

Ionis Therapeutics announced that its GSK‑partnered antisense drug bepirovirsen achieved a statistically significant functional cure rate of roughly 15‑20% in a Phase 3 trial, far surpassing the 1‑3% rate of existing hepatitis B therapies. The company also reported a strong first‑year launch...

A new analysis in Health Affairs Scholar reveals that brand‑name drugmakers are exploiting the Hatch‑Waxman framework through serial patent litigation and continuation patents to extend market exclusivity. By filing multiple overlapping patents, companies can repeatedly sue generic challengers, triggering 30‑month...

Resveratrol protects kidneys from fat accumulation and diabetic kidney disease by activating SIRT1, similar to its effects on fatty liver 🐁 www.sciencedirect.com/science/article/abs/pii/S1098882326000067

ArgenTag, a Buenos Aires‑based startup, has entered PacBio’s Compatible Partner Program, confirming that its Single‑Cell RNA Library Kit works with PacBio’s Kinnex RNA kits for long‑read isoform sequencing. The partnership eliminates the need for expensive cell‑partitioning instruments by using ArgenTag’s...
Glaukos Corp reported record fourth‑quarter 2025 sales of $143.1 million, a 36% year‑over‑year increase, and annual net sales of $507.4 million, up 32%. The company reaffirmed its 2026 revenue outlook of $600‑$620 million, indicating more than 20% growth. Growth was powered by the...

A pooled analysis of five double‑blind, placebo‑controlled intranasal esketamine trials (n≈1,016) confirms that esketamine significantly reduces Montgomery‑Åsberg Depression Rating Scale (MADRS) scores versus placebo. Female participants showed larger total MADRS improvements at later visits (days 22 and 28) and greater reductions in...
The study engineered 3D microvascular networks from Rett syndrome (RTT) patient‑derived iPS cells carrying MeCP2[R306C] or MeCP2[R168X] mutations and discovered markedly increased vascular permeability. Transcriptomic profiling linked this defect to up‑regulation of endothelial‑specific miR‑126‑3p, which suppresses tight‑junction regulators such as...

Ocular Therapeutix reported that its VEGF1/2/3 inhibitor Axpaxli outperformed aflibercept in the Phase III SOL‑1 trial, with 74.1% of patients maintaining visual acuity at 36 weeks versus 55.8% for the comparator. The study also met a 52‑week durability endpoint, showing 65.9%...

The BioCentury This Week podcast highlighted the rapid expansion of oral selective estrogen receptor degraders (SERDs) as a leading application of targeted protein degradation, noting several candidates advancing toward late‑stage trials. Host Lauren Martz examined the competitive landscape and the therapeutic...

Kobe University bioengineers have re‑programmed the gut bacterium Escherichia coli to synthesize orsellinic acid, the core of Rhododendron‑derived meroterpenoids, at 202 mg per liter—a 40‑fold increase over prior microbial attempts. This marks the first time the complex eukaryotic pathway has been...

Embryos and old people have a lot in common: the PRC2 protein complex, which controls DNA methylation during embryogenesis, is a key participant in age reversal via epigenetic reprogramming link.springer.com/content/pdf/10.1038/s44320-026-00195-9.pdf

Researchers at IIT Gandhinagar have merged scanning electron microscopy (SEM) with artificial intelligence to automate pollen grain identification. Using a dataset of 28 medicinal plant species, they built a computer‑vision pipeline that segments pollen via YOLOv11n and classifies it with...

Researchers at Southern University of Science and Technology have unveiled an optoelectrowetting platform that uses programmable light patterns to dispense nanoliter droplets with unprecedented precision. By projecting dynamic illumination onto a microfluidic chip, the system creates virtual electrodes that guide...

Gilead Sciences has agreed to pay $80 million upfront to Suzhou Genhouse Bio for worldwide rights to GH31, a clinic‑ready oncology molecule. GH31 employs synthetic lethality to inhibit the MAT2A enzyme, a target implicated in several cancers. The deal follows regulatory...

In a BioCentury interview, RA Capital Managing Partner Peter Kolchinsky warned that staffing cuts and a growing conservative stance at the FDA are adding at least a three‑month delay to even the most promising drug programs. He says sponsors are now...

Researchers at NYU Langone Health discovered that pancreatic ductal adenocarcinoma cells sense extracellular matrix (ECM) components via the surface protein integrin α3, which determines whether they adopt a proliferative or autophagic state. Cells anchored to laminin‑rich ECM keep autophagy low and...

The FDA’s Biomarker Qualification Program (BQP) offers a three‑stage pathway—Letter of Intent, Qualification Plan, and Full Qualification Package—to secure regulatory endorsement of biomarkers as drug development tools. Under the 21st Century Cures Act, the process has been modernized, allowing qualified...

Enlil unveiled Milestone View, the industry’s first evidence‑based milestone interface for medical device product development. The new capability embeds directly within Enlil’s unified traceability platform, linking each milestone to underlying requirements, design outputs, risk controls, change histories and regulatory evidence....

The European Journal of Pediatrics retracted the 2024 randomized trial that claimed folinic acid reduced autism symptoms, citing data that did not support its conclusions. The study had been the largest of its kind, influencing clinical recommendations and regulatory guidance....

Swedish researchers have created a photo‑patternable solid‑state hydrogel electrolyte using i‑carrageenan and PEGDA, achieving ionic conductivity above 10 mS cm⁻¹ and feature sizes down to 15 µm. The material replaces liquid electrolytes in organic electrochemical transistors (OECTs), enabling fast, dense, and flexible circuits...

Researchers used UK Biobank data to map vascular phenotypes across the retina, carotid artery, aorta, and brain, revealing consistent cross‑organ correlations. Retinal vascular density showed modest but significant negative links with white‑matter hyperintensities, carotid intima‑media thickness, and aortic lumen size,...

Incretin (GLP-1) based therapies have dramatic effects on many aspects of metabolic disease. Can this occur independent of the effect on obesity? https://www.nature.com/articles/s44324-024-00030-5

.@Vero_Bioscience is working to end age related chronic disease. With a simple blood test, Vero uses proteomics and machine learning to measure the biological age of each organ, revealing where the body is aging fastest so people can intervene early...

A European biotech is challenging the ADC status quo by prioritising novel targets and payloads over sheer potency. In an interview, the company’s CSO argues that the industry’s obsession with ultra‑potent cytotoxins like MMAE and DM1 is stalling progress, creating...

With all the attention on China for TOPO-I ADCs of late, how about the potential for European agents, especially ones involving novel targets? Our latest expert interview drops with some hard hitting and candid opinions from a battle hardened...

Paris‑based AGS Therapeutics signed a strategic agreement with INITS SMO, a GMP‑qualified shared manufacturing organization, to transition its microalgae‑derived extracellular vesicle (MEV) platform to full GMP compliance. The partnership allows AGS to run its proprietary MEV manufacturing, loading, and analytics...

The article outlines a growing strategy to develop central nervous system (CNS) therapeutics that remain pharmacologically active only within the brain, termed "BrainOnly" pharmacology. By leveraging selective transport mechanisms, pro‑drug designs, and peripheral clearance pathways, researchers aim to minimize off‑target...

Researchers at the University of Missouri have identified the protein SRFR1 as a key regulator of root depth in plants. By editing the SRFR1 gene, they induced significantly longer roots in test crops. The modified plants demonstrated enhanced water acquisition...

ArgenTag, now a PacBio Compatible Partner, has launched a grant that provides free access to its Single‑Cell RNA Library Kit for Long‑Read Sequencing. The kit uses a gravity‑based microfluidic chip to isolate individual cells, capture full‑length mRNA, and prepare barcoded...

The FDA has completed the BsUFA II deliverables, publishing a final assessment of the Biosimilar User Fee Act review program on February 21, 2022 and an interim assessment on January 27, 2021. Alongside these reports, the agency released annual updates...

Boehringer Ingelheim is expanding its cardio‑renal‑metabolic (CRM) platform beyond its recent idiopathic pulmonary fibrosis (IPF) successes to target obesity and related metabolic disorders. President Brian Hilberdink says the company is prioritizing partnerships rather than large‑scale M&A to broaden its pipeline...

Recruitment of BRD4 to the ASXL1 genomic targets depends on the extra-terminal domain of BRD4 https://t.co/sRbcAAMqPx https://t.co/IbD0EM56SS
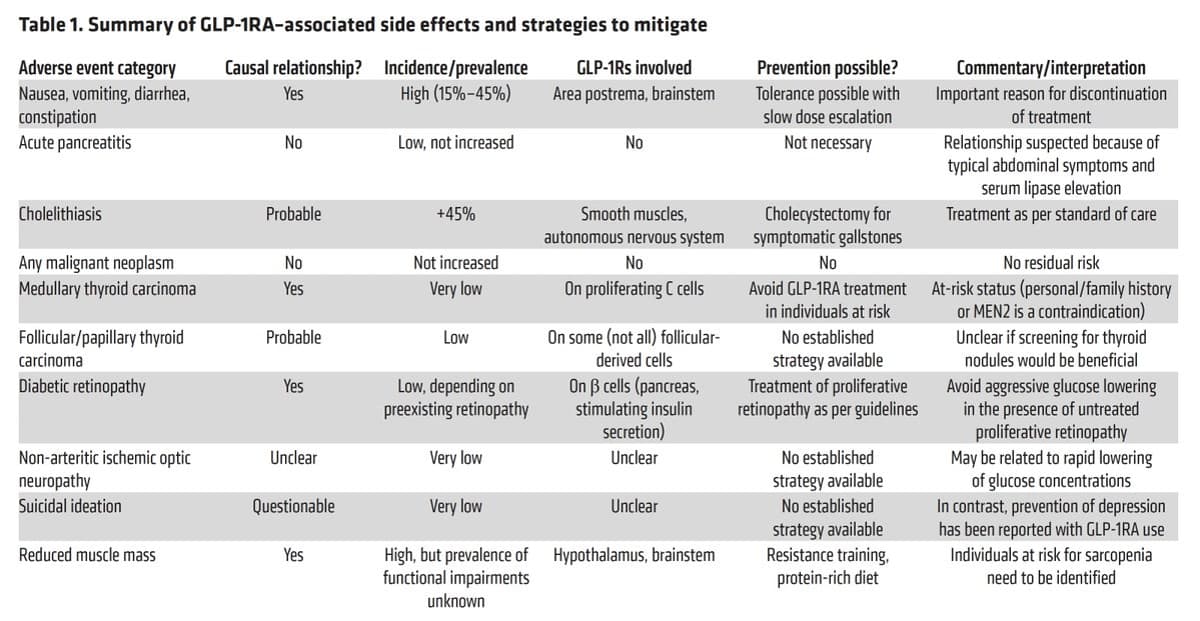
A short thread on GLP-1s based on new publications. 1. The safety story Table of potential side effects and very good description of mechanisms, especially w/r to retina https://t.co/L5GNZyArrn https://t.co/MyXVOF3SYv
Vaxart is advancing an oral, bivalent norovirus vaccine pill (VXA‑G1.1‑NN) that demonstrated a 30% reduction in infection in a Phase 2b challenge study. The vaccine uses a non‑replicating adenovirus vector loaded with a dsRNA adjuvant to stimulate gut immunity, and a...

🧵 Need gene lengths for every mouse gene? Bioconductor annotation packages make this simple. Here's how to get gene lengths using TxDb.Mmusculus and https://t.co/FiqDAsSPa0.db. https://t.co/VyE36hp3om

AI biology firm Prima Mente has launched Pleiades, a 7‑billion‑parameter epigenetic foundation model that reads DNA methylation in cell‑free DNA to diagnose early‑stage Alzheimer’s and Parkinson’s. In real‑world cohorts the model achieved 89% detection accuracy, rising to 97% when combined...
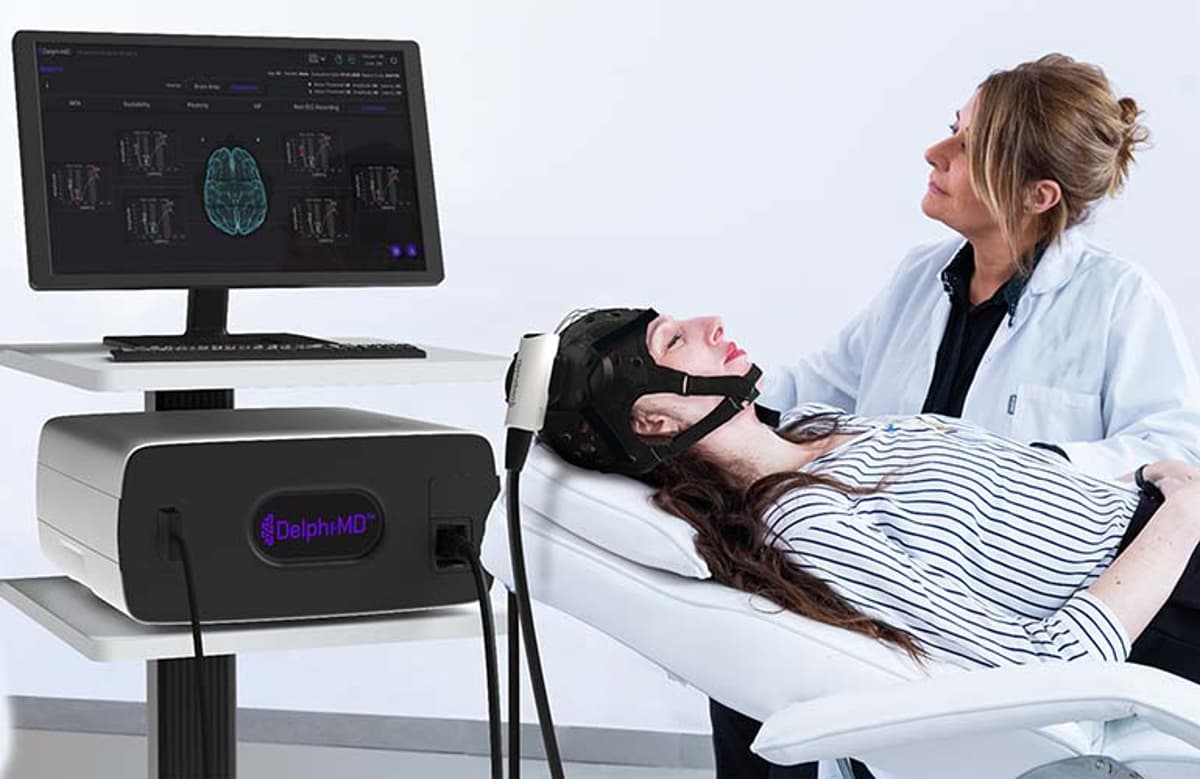
QuantalX secured FDA de novo clearance for Delphi‑MD, a first‑of‑its‑kind device that fuses transcranial magnetic stimulation (TMS) with electroencephalography (EEG) to deliver real‑time brain network function assessments. The system leverages proprietary algorithms and an age‑correlated normative database to generate actionable...

Regulatory T‑cell (Treg) therapies are moving from discovery to clinic, with multiple biotech firms reporting early clinical data. Sonoma Biotherapeutics’ CAR‑Treg candidate SBT‑77‑7101 achieved a 67% reduction in rheumatoid‑arthritis swelling in a Phase 1 trial, while Coya Therapeutics secured IND approval...

The FDA’s Office of Generic Drugs has launched a public, bi‑weekly list of all approved abbreviated new drug applications (ANDAs) that carry a Competitive Generic Therapy (CGT) designation. The list details each product’s reference listed drug, NDA number, applicant, active...

Never‑smokers now account for 10‑20% of U.S. lung‑cancer cases, a share that is climbing despite overall declines. Current USPSTF guidelines limit low‑dose CT screening to heavy‑smoking adults, leaving most never‑smokers unscreened. Shira Boehler’s incidental finding on a whole‑body MRI prompted...

The FDA is actively phasing out animal‑testing requirements for certain therapies, a move echoed by the NIH and codified in the pending FDA Modernization Act. This regulatory shift opens the door for artificial intelligence and human‑derived organoid models to become...

As I’ve written @BakerInstitute MAHA = a system of pseudoscience designed to undermine scientific biomedicine in America for the benefit and profits of the wellness influencer industry. It’s incredibly corrupt and endangers the health of Americans. Vaccines are just the...

Cogent Biosciences announced that its lead drug bezuclastinib is on track for multiple FDA submissions in 2026, including NDAs for non‑advanced systemic mastocytosis (Non‑AdvSM), advanced SM (AdvSM) and second‑line GIST. The company reported $901 million in cash, sufficient to fund operations...