DNA-Containing Extracellular Vesicles Boost Antitumor Responses in Mice
Researchers at Weill Cornell Medicine discovered that extracellular vesicles released by activated T cells contain DNA that can be transferred to dendritic and tumor cells, enhancing antigen processing and presentation. In mouse models of glioblastoma, pancreatic and triple‑negative breast cancer, these DNA‑laden vesicles slowed tumor growth and synergized with checkpoint inhibitors. The study suggests a new acellular immunotherapy that could boost immunity against immunologically cold tumors and serve as a non‑viral gene‑delivery platform.

It’s Time to Take Genetic Testing Off the Pedestal
Genomic testing, once a specialist‑only tool, is now technologically mature and affordable, yet it remains underused in routine care. Advances in sequencing speed, AI‑driven interpretation, and large data sets have removed most technical barriers. Patients are increasingly seeking molecular insights,...

Pharmaceutical Executive Daily: FDA Approves Auvelity
The FDA approved Axsome Therapeutics’ Auvelity for agitation associated with Alzheimer’s disease, marking the first non‑antipsychotic and only the second drug cleared for this indication. Earlier, the FDA’s Oncologic Drugs Advisory Committee gave AstraZeneca’s Truqap a 7‑1 favorable benefit‑risk vote,...

Allosteric Switch in TB Enzyme Opens New Drug Target
A newly identified allosteric switch in the ICL2 enzyme of Mycobacterium tuberculosis reveals a potential pathway for targeting drug-resistant TB, offering insight into protein regulation unique to the bacterium. structuralbiology

The Next Alzheimer’s Breakthrough Will Take More Than Just Science
Alzheimer’s research has moved from theory to treatment as anti‑amyloid antibodies like Lecanemab and Donanemab receive regulatory approval and begin reaching patients. These drugs can clear existing amyloid plaques and modestly slow cognitive decline, extending the disease trajectory from roughly...
Restoring Protein Recycling Reverses T-Cell Exhaustion in Mice
Scientists at UC San Diego discovered that impaired protein recycling drives T‑cell exhaustion in mice. Restoring the activity of specific E3 ligases—NEURL3, RNF149, and WSB1—reestablished proteostasis, cleared misfolded proteins, and revived T‑cell anti‑tumor function. The findings, published in Cell, suggest...

Moderna in Talks with FDA over Phase 4 Covid Vaccine Data
Moderna is actively collaborating with the U.S. Food and Drug Administration to submit Phase 4 post‑marketing data on its COVID‑19 vaccines. The company hopes the additional safety and efficacy evidence will persuade regulators to broaden the current, narrowed product labels that...

AI, Gene Therapies Drive Market Trends in Eye Care
In 2026 the ophthalmology market is being reshaped by gene‑therapy breakthroughs and the emergence of agentic AI, according to Boston Consulting Group’s Long Sha. Gene‑based treatments are moving beyond rare retinal disorders into chronic conditions such as wet age‑related macular...
Common Cholesterol Medications Do Not Alter Long-Term Dementia Risk
A massive target‑trial emulation study of more than 320,000 older adults found that statin use does not change long‑term risk of dementia. While statin users showed a 46% spike in dementia diagnoses during the first year after initiation, researchers attribute...

Ensorcell Debuts VersaWeld Platform, Wins Best New Product at INTERPHEX 2026
Ensorcell introduced the VersaWeld sterile tube welding platform at INTERPHEX 2026, marking its entry into cell and gene therapy (CGT) manufacturing equipment. The system automates thermoplastic tubing welding in closed‑system environments, delivering consistent weld integrity across multiple tube sizes and materials...

Amgen Launches Late-Stage Obesity Trial in Patients Who Switch From Rival Drugs
Amgen is initiating three Phase III trials for its long‑acting obesity injection MariTide, including a pivotal study that enrolls about 1,200 patients switching from Eli Lilly’s semaglutide or Novo Nordisk’s tirzepatide. The primary goal is a minimum 10% body‑weight loss after 68 weeks,...

TMDX’s OCS Tech Drives Rapid Adoption, Pressures Valuation
$TMDX reports Tuesday after the close This is the only company on earth that keeps a donor heart, lung, or liver beating outside the body Not a metaphor... the organ stays warm and oxygenated e2e The number to watch on Tuesday isn't rev,...

Update on Brad Stanfield's Rapamycin Clinical Study in NZ
Brad Stanfield’s New Zealand rapamycin trial enrolled older adults on a 12‑week protocol, with participants typically taking 6 mg every other week. The study measured functional outcomes such as the chair‑stand test, sparking debate over whether short‑term dosing can reveal longevity benefits. Commentators...

Shield Therapeutics Posts $2.5M EBIT in Q1, CFO Resigns Amid 11% Share Drop
Shield Therapeutics plc posted a $2.5 million EBIT for the first quarter, a swing from a $4.4 million loss a year earlier, thanks to a $7.9 million development milestone from ASK in China and a 54% jump in ACCRUFeR sales. At the same...

F.D.A. Grants Early Access to Promising Drug for Pancreatic Cancer
On May 1, the FDA granted expanded‑access permission for daraxonrasib, an experimental oral drug from Revolution Medicines, allowing patients with previously treated metastatic pancreatic cancer to obtain the therapy outside clinical trials. The drug, taken as three pills daily, has produced...

Pacira Q1 2026 Earnings Highlight COO Brendan Teehan's Product Launch Drive
Pacira BioSciences posted a 7% rise in EXPAREL sales to $143.3 M and double‑digit growth for ZILRETTA and ioverao in Q1 2026, attributing the momentum to COO Brendan P. Teehan’s accelerated product launches and expanded sales teams. The company also repurchased...

Nanjing King‑Friend Biochemical Pharma Posts 4.9% Q1 Revenue Rise to $130 M
Nanjing King‑Friend Biochemical Pharmaceutical Co., Ltd. announced first‑quarter revenue of RMB928.66 million ($130 million), up 4.9% from a year earlier, and profit of RMB89.01 million ($12.5 million). The modest earnings per share of RMB0.05 matched last year, signaling steady profitability amid growing demand for...

Regulatory Actions for May 1, 2026
BioWorld released a comprehensive “Regulatory Actions for May 1, 2026” roundup, collating FDA, EMA and other global regulator updates across biopharma, medical technology, and policy domains. The collection links to data snapshots, infographics on digital analysis, and special reports covering topics such...

The Cyborgs Commeth - EP 69 Connor Glass
In this episode, host Ashley Vance talks with Connor Glass, co‑founder and CEO of Phantom Neuro, about the company’s sub‑dermal neural interface that lets amputees control prosthetic limbs using muscle‑derived electrical signals—offering a less invasive alternative to brain‑computer interfaces like...

FDA Permits Expanded Access for Investigational Pancreatic Cancer Drug
The FDA issued a “safe to proceed” letter to Revolution Medicines, enabling an expanded access protocol for its experimental pancreatic cancer drug daraxonrasib. The request, received on April 28 and signed on April 30, targets patients with previously treated metastatic pancreatic ductal...

Redo TAVR: Supra-Annular, Intra-Annular Valves Linked to Comparable Outcomes
A study of 172 redo transcatheter aortic valve replacement (TAVR‑in‑TAVR) procedures from the international PANDORA registry shows comparable one‑year outcomes regardless of whether the initial and second valves are supra‑annular or intra‑annular. The median interval between the index and redo...

Acelyrin Founder Shao-Lee Lin Emerges at Cue, as It Becomes Latest to License From China
Cue Biopharma announced a strategic reboot, appointing Acelyrin founder Shao‑Lee Lin to its executive team and unveiling a new allergy drug candidate sourced from research labs in Taiwan and mainland China. The Boston‑based company, previously focused on T‑cell immunotherapies, will...

Asembia AXS26: How CGTs Are Reshaping Specialty Pharmacy
Cell and gene therapies (CGTs) are fundamentally changing the specialty pharmacy landscape, moving beyond traditional buy‑and‑bill models toward highly coordinated, patient‑specific workflows. The therapies demand specialized treatment‑center activities such as apheresis, conditioning, and strict cold‑chain logistics. High upfront costs create...
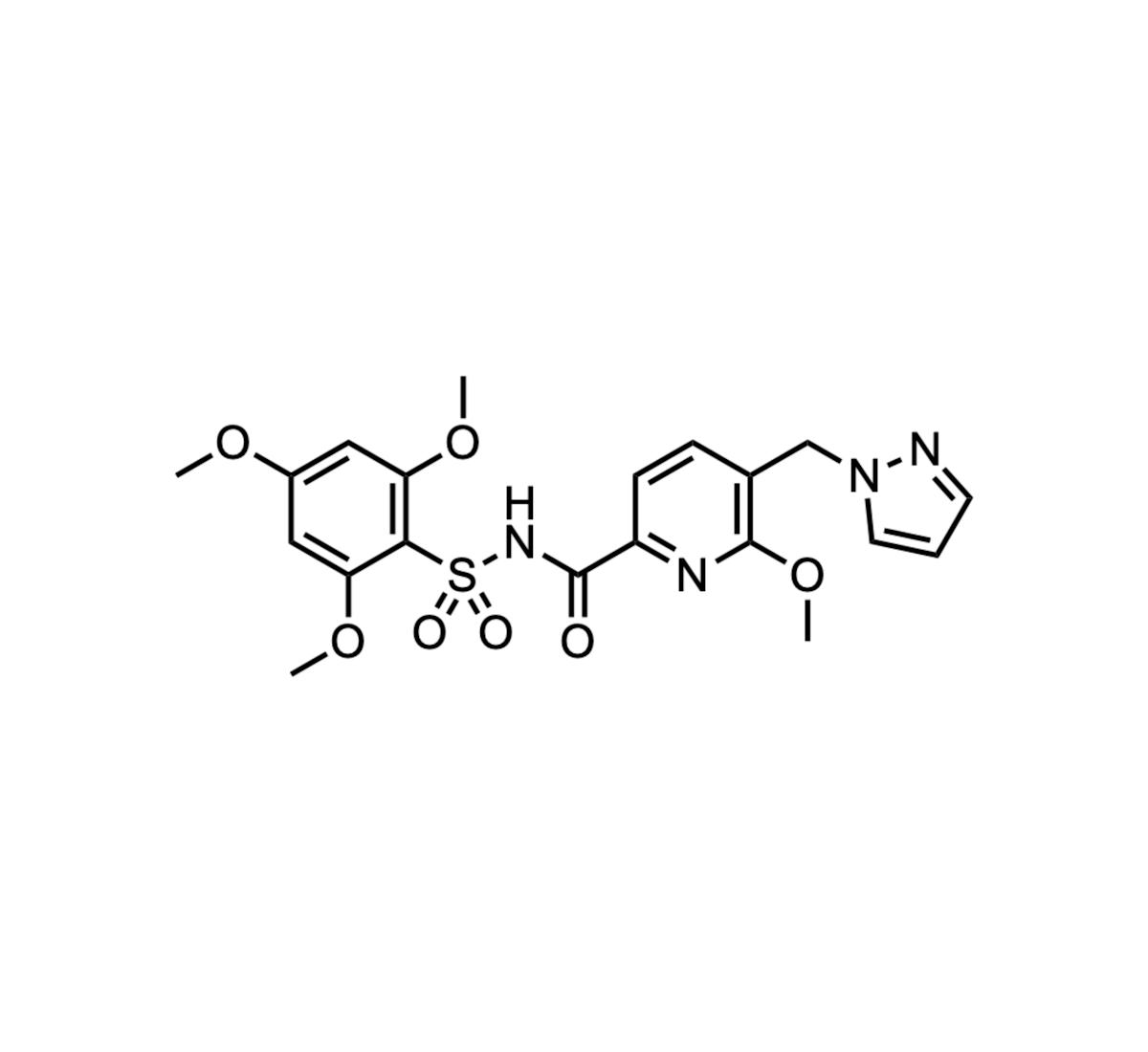
OP-3136
OP‑3136, a KAT6A‑selective inhibitor, entered Phase 1/2 trials for advanced hormone‑receptor‑positive breast cancer. The drug mimics the pyrophosphate of acetyl‑CoA using an acyl‑sulfonamide scaffold, delivering high specificity for the epigenetic writer KAT6A. Olema Pharmaceuticals is testing OP‑3136 in combination with SERDs...

Microbiome-Based Therapy Gains FDA Fast Track in Ulcerative Colitis
Belgium‑based MRM Health announced that its investigational microbiome‑based therapy MH002 has been granted FDA fast track designation for mild‑to‑moderate ulcerative colitis. MH002 is a live biotherapeutic composed of six defined commensal bacterial strains designed to restore gut microbial balance and...

Sydnexis to Present New Data From Phase 3 STAR Trial of SYD-101 at ARVO 2026 Annual Meeting
Sydnexis announced it will unveil new subgroup analysis data from the Phase 3 STAR trial of its low‑dose atropine eye drop SYD‑101 at the ARVO 2026 meeting in Denver. The analysis focuses on children with fast‑progressing myopia, a cohort that typically...

World Asthma Day 2026: New Anti-Inflammatory Drugs and More for Asthma Control
World Asthma Day on May 5 highlighted the persistent gap in access to anti‑inflammatory inhalers, a core theme of the Global Initiative for Asthma. The FDA approved AstraZeneca’s Breztri Aerosphere, the first single‑inhaler triple therapy that combines an inhaled corticosteroid, a...

New Semaglutide for Alcohol Use Disorder Trial Shows Big Drops in Drinking
A Lancet‑published, double‑blind, 26‑week trial found once‑weekly semaglutide markedly reduced alcohol consumption in participants with alcohol use disorder and obesity. Across primary drinking endpoints, the semaglutide arm showed statistically significant declines compared with placebo, despite both groups receiving identical cognitive‑behavioral...

Arrowhead Pharmaceuticals Receives TGA Approval of REDEMPLO® (Plozasiran) in Australia, Expanding Global Access for Patients with Familial Chylomicronemia Syndrome (FCS)
Arrowhead Pharmaceuticals announced that Australia’s Therapeutic Goods Administration approved REDEMPLO® (plozasiran), the first siRNA therapy for familial chylomicronemia syndrome (FCS) in the country. The drug, administered subcutaneously every three months, targets apoC‑III to dramatically lower triglycerides. In the Phase 3 PALISADE...

Policy Watch: FDA Looks to Expand Real-Time Drug Clinical Trials
The FDA has launched an AI‑driven pilot that streams clinical‑trial data in real time for AstraZeneca and Amgen’s lymphoma and lung‑cancer studies, and is now seeking public input to broaden the approach. The Ninth Circuit Court declined to compel the...
Gut Microbe’s Sulfated Bile Acid Eases Pediatric Sepsis
Researchers identified deoxycholic acid 3‑sulfate (DCA‑3S) as a gut‑derived metabolite that mitigates pediatric sepsis. Metabolomic and metagenomic analyses revealed Enterococcus raffinosus as the primary producer, accounting for over 80 % of DCA‑3S synthesis. In mouse models, DCA‑3S restored intestinal barrier integrity and dampened...
STAT+: Axsome Wins FDA Nod for Alzheimer’s Agitation
Axsome Therapeutics announced that the U.S. Food and Drug Administration has granted a regulatory nod for its investigational therapy aimed at treating agitation in Alzheimer’s disease. The agency’s decision clears the path for accelerated clinical development, potentially moving the drug...

Novel Pulsed Field Ablation Technology ‘Works’
A first‑in‑human trial of Pulse Biosciences' nanosecond pulsed‑field ablation (CellFX nsPFA 360) treated 177 patients with paroxysmal atrial fibrillation. The catheter delivered >10,000 V nanosecond pulses, achieving 100% acute lesion success and 91% durability at 2‑3 months. At one year, 89.7% of patients remained free...
Peptides Are Unproven as Health Aids. FDA May Unleash Them Anyway
The FDA is poised to broaden access to injectable peptides by allowing compounding pharmacies to produce them and by considering their inclusion in oral dietary supplements. Health and Human Services Secretary Robert F. Kennedy Jr. has publicly opposed what he...

Drug Trials Snapshot: TRYNGOLZA
TRYNGOLZA (olezarsen), an APOC‑III‑directed antisense oligonucleotide, received FDA approval on December 19, 2024 for adults with familial chylomicronemia syndrome (FCS). In the pivotal Phase 3 trial (NCT04568434), 66 patients from 11 countries received monthly 80 mg injections or placebo for 53 weeks. The drug achieved...
Programmable RNA Targeting via DNA-Guided CRISPR-Cas12a
A team of molecular biologists has reengineered the CRISPR‑Cas12a nuclease to cleave RNA using a DNA guide, creating a programmable RNA‑targeting platform. The DNA‑guided Cas12a system achieved up to 90% knockdown of endogenous transcripts in human cell lines and functioned...

Drug Trials Snapshot: ALYFTREK
Vertex Pharmaceuticals received FDA approval for ALYFTREK, a triple‑combination CFTR modulator, on Dec. 20, 2024. The drug targets cystic fibrosis patients aged six and older with at least one F508del or other responsive mutation. Approval was based on two 52‑week, non‑inferiority trials...

Drug Trials Snapshot: ROMVIMZA
Deciphera's ROMVIMZA (vimseltinib) received FDA approval on Feb 17 2025 for adult tenosynovial giant‑cell tumor (TGCT) when surgery is unsuitable. Approval was based on a global phase II trial of 123 patients, showing a 40% overall response rate versus 0% with placebo...

Rosen Law Firm Opens Securities Class Action Probe Into Disc Medicine After FDA Setback
Rosen Law Firm announced a securities class‑action investigation into Disc Medicine (NASDAQ: IRON) following an FDA Complete Response Letter that halted the company's bitopertin program and sent the stock down 22% on Feb. 13, 2026. The firm is urging shareholders to join...

Cerus Names Vivek K. Jayaraman as New COO, Raises 2026 Revenue Guidance
Cerus Corporation introduced Vivek K. Jayaraman as its incoming chief operating officer during its Q1 2026 earnings call. The leadership change coincides with a 24% jump in product revenue to $53.7 million and an upward revision of full‑year product revenue guidance...

Summit Slips on Ivonescimab's Apparent Interim Miss in Sign of Investor Frustration
Summit Therapeutics’ shares plunged almost 20% after an interim analysis of its Phase 3 trial of ivonescimab showed a statistical shortfall in a key efficacy endpoint. The miss was limited to a subgroup analysis, but investors interpreted it as a signal...
High‑dose Semaglutide Cuts Alcohol Use in RCT
Clinicians and patients saw big reductions in alcohol intake when starting GLP-1 medicine. This is the first randomized trial to look at the high dose of semaglutide. There was a study a couple years back looking at a lower dose....
CNS Gene Therapies Showcase Tau-Targeted VY170
CNS Gene Therapies Featured in Multiple Presentations at ASGCT 2026, Including Late Breaker on Tau-Targeted VY1706 for Alzheimer’s Disease https://t.co/oQ5MCd5piS https://t.co/8KeWSTJ1q7

Empa and HOCH Health Launch Light‑Activated Nanozyme Therapy for Brain Tumors
Empa and the HOCH Health Ostschweiz network have begun a research partnership to create a light‑activated nanozyme therapy for astrocytoma and other aggressive brain tumors. The project, funded by several Swiss foundations, targets the blood‑brain barrier challenge and plans to...
FDA Approves Early, yet ARVN, PFE Lack Partner
FDA approves vepdegrestrant one month early. $ARVN and $PFE still haven't found (or disclosed) a third-party company to commercialize it. What a world...

ARCH Backs Mental‑health IPO, Celebrating Early Investment Success
Congrats to Daphne Zohar and team, and Steve Paul for an amazing IPO to fuel new drugs for mental health and brain disorders. ARCH invested early here because of the team, platform, and clinical need.
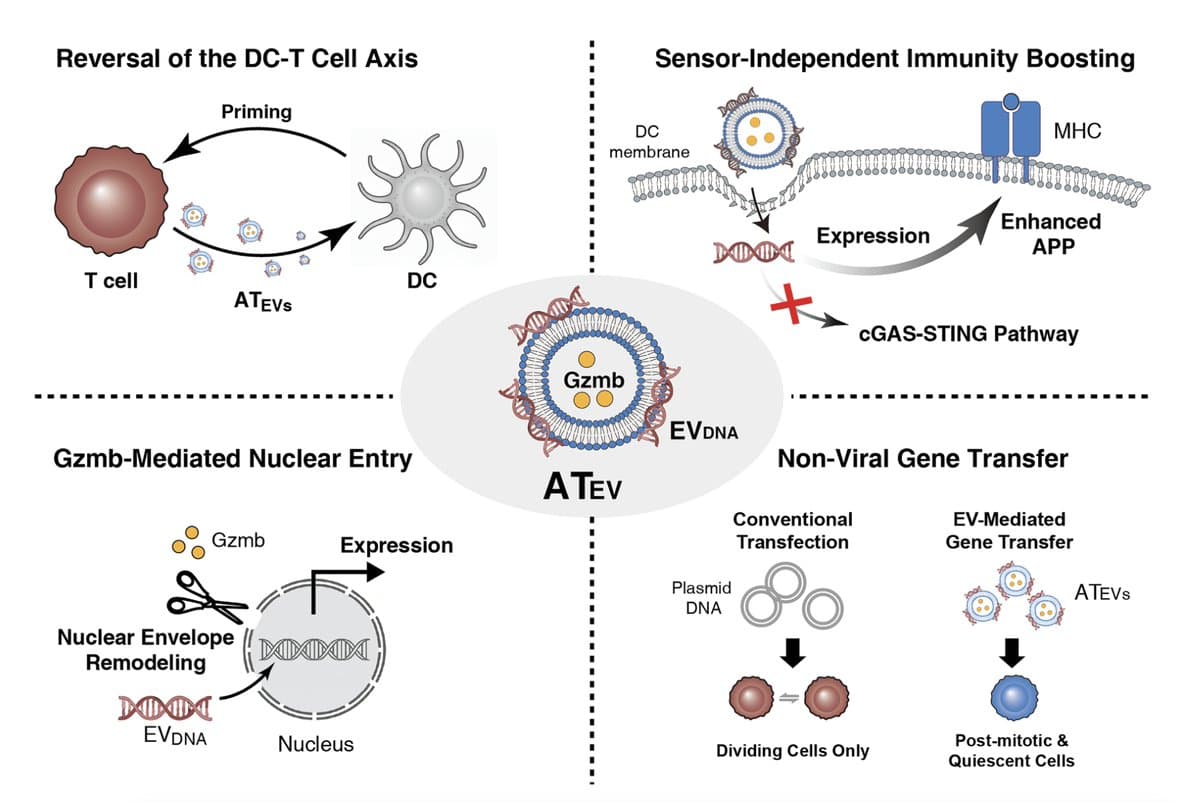
T‑cell Vesicles Deliver DNA, Converting Cold Tumors Hot
In cancer, activated T cells release abundant extracellular vesicles that transfer DNA to the nucleus of tumor and immune (dendritic) cells, turning tumors from cold to hot. An immunotherapy in the works. @Cancer_Cell https://t.co/mHcahijOJC https://t.co/oPT6d7lrFM https://t.co/I0egYxZold

Early Trial RNA‑seq Data: Exciting Yet Requires Caution
🧵 You just got your hands on early clinical trial RNA-seq data. Excited? You should also be cautious. Here's why. 👇 https://t.co/GjA8XIYcVQ

Biotech IPOs Surge, Exposing Wall St Underestimation
Three biotech IPOs this week. Each one raised its number of shares and priced atop range. Suggests that Wall St is slightly underestimating investor interest in new biotech issues.

AI Chatbots Capture Poorer Symptom Detail than Doctors
Symptoms reported to an AI chatbot were of lesser quality than reported to a physician, results of a randomized trial of 500 participants across multiple models https://t.co/WXP4R3y9HA https://t.co/l2hLT76AJB