
Pfizer, Arvinas Win FDA Nod for VEPPANU, First PROTAC Cancer Drug
Pfizer and Arvinas have secured U.S. FDA approval for VEPPANU (vepdegestrant), the first orally bioavailable PROTAC drug, targeting estrogen‑receptor positive, HER2‑negative, ESR1‑mutated advanced breast cancer after endocrine therapy failure. The decision validates the targeted protein degradation platform while raising questions about commercial uptake and broader applicability.

Review Flags Neurotoxic Risks of Silver Nanoparticles in Rodent Studies
Researchers from Çanakkale Onsekiz Mart University and University Montpellier published a review on 29 April 2026 that links silver nanoparticle exposure to multiple neurotoxic mechanisms in rodent models. The paper warns that expanding biomedical applications—such as neural implants and drug‑delivery systems—must contend...
Loyal's Canine Longevity Pill Nears Market After FDA Deems Likely Effective
Loyal, the biotech startup founded by Celine Halioua, has moved its canine longevity pill closer to market after the FDA deemed it likely effective in February 2025. The drug, which could add a year to a dog’s healthy lifespan, arrives amid...

Redefining Success in Transplant Medicine
In this episode of Signals KOLs, transplant nephrologist Dr. Deirdre Sawinski, industry veteran Steve Risk (SVP Medical Affairs at Veloxys), and patient‑physician Sejal Patel discuss the stagnant long‑term outcomes in kidney transplantation despite excellent short‑term success. They highlight how current...

RESEARCH: NICLOSAMIDE in CANCER and Other Diseases - 2025 Review Paper From Henan, China
A 2025 review paper from Henan, China, evaluates niclosamide—a decades‑old anti‑parasitic—as a repurposed oncology agent. The analysis compiles pre‑clinical data across breast, lung, pancreatic and colorectal cancers, and highlights early‑phase clinical trials showing modest tumor responses. Researchers also discuss formulation...

Liquid Biopsy Predicts Response to Breast Cancer Immunotherapy
Researchers at Vanderbilt-Ingram Cancer Center demonstrated that serial liquid biopsies analyzing peripheral blood RNA can predict response to pembrolizumab in high‑risk early‑stage HER2‑negative breast cancer. The study examined 546 blood samples from 160 patients in the I‑SPY2 trial, showing transcriptional...

Electronics‑Free Smart Lens Monitors Glaucoma and Releases Medication
Researchers have developed a prototype electronics-free smart contact lens that can track glaucoma in real time and deliver drugs in response. https://spectrum.ieee.org/smart-contact-lens-glaucoma-microfluidics

Spaceflight Is Hard on the Heart, yet Artificial Ones Grow Better in Space than on Earth
Researchers demonstrated that miniature human hearts grown from stem cells mature faster in microgravity than on Earth. The International Society for Heart and Lung Transplantation meeting highlighted data from ISS experiments showing a significant boost in organoid production without the...

The Biosimilar Market Isn’t Failing, It’s Finding Its Purpose
The biosimilar market is not collapsing; it is transitioning from a price‑driven scramble to a purpose‑focused strategy. Recent HHS data show that markets become uneconomic once five competitors vie for the same molecule, prompting a shift toward diversified product portfolios....

FDA Closes the 503B Bulks Door on Semaglutide, Tirzepatide, and Liraglutide
On April 30, 2026 the FDA issued a proposal to exclude semaglutide, tirzepatide and liraglutide from the 503B Bulks List, arguing there is no clinical need for outsourcing facilities to compound these GLP‑1 drugs. The move follows the resolution of...

FDA Rejects RP1 Melanoma Therapy After Fast‑Track Review
The FDA has denied approval of RP1, an oncolytic immunotherapy for advanced melanoma, despite its breakthrough designation. The agency cited a heterogeneous study population and lack of a control arm, overturning an earlier panel recommendation.
Gene Therapy Gives Deaf Toddlers Hearing After One Injection
Regeneron’s Otarmeni received FDA accelerated approval after an international trial showed toddlers with congenital OTOF‑related deafness responding to a single injection. The study, led by Mass Eye and Ear and Fudan University, recorded measurable hearing in 80% of participants, sparking...

Rethinking Blood Thinners for Atrial Fibrillation Patients
At the American College of Cardiology meeting, a three‑year trial demonstrated that the Watchman left‑atrial‑appendage closure device provides stroke protection comparable to lifelong anticoagulation while causing far fewer bleeding events. The findings challenge the entrenched belief that atrial fibrillation patients...
Blocking a Cellular Inflammation Process Could Result in Effective Therapy for Pancreatic Cancer
Scientists at The Wistar Institute and ChristianaCare identified a vulnerability in pancreatic cancer where defective mitochondria release double‑stranded RNA, triggering the TLR3/TRAF6 inflammatory pathway. The tumor cells become dependent on this inflammation for growth and survival, and blocking the pathway...

Stopping and Restarting Certain GLP-1s to Lose Weight May Make the Drug Less Effective
A preclinical study from the University of Pennsylvania found that stopping and restarting GLP‑1 weight‑loss drugs, such as semaglutide, markedly diminishes their efficacy. Overweight mice on a stop‑and‑start regimen regained weight during off periods and never recaptured their initial loss,...

T‑cell Engagers Face Uncertain Future Amid Mixed Readouts
T cell engagers for I&I will have a lot of clinical readouts this year. This update (report from MS) from $AMGN on blinatumomab last week doesn't bode well for the class and it's unclear if next gen formats will...
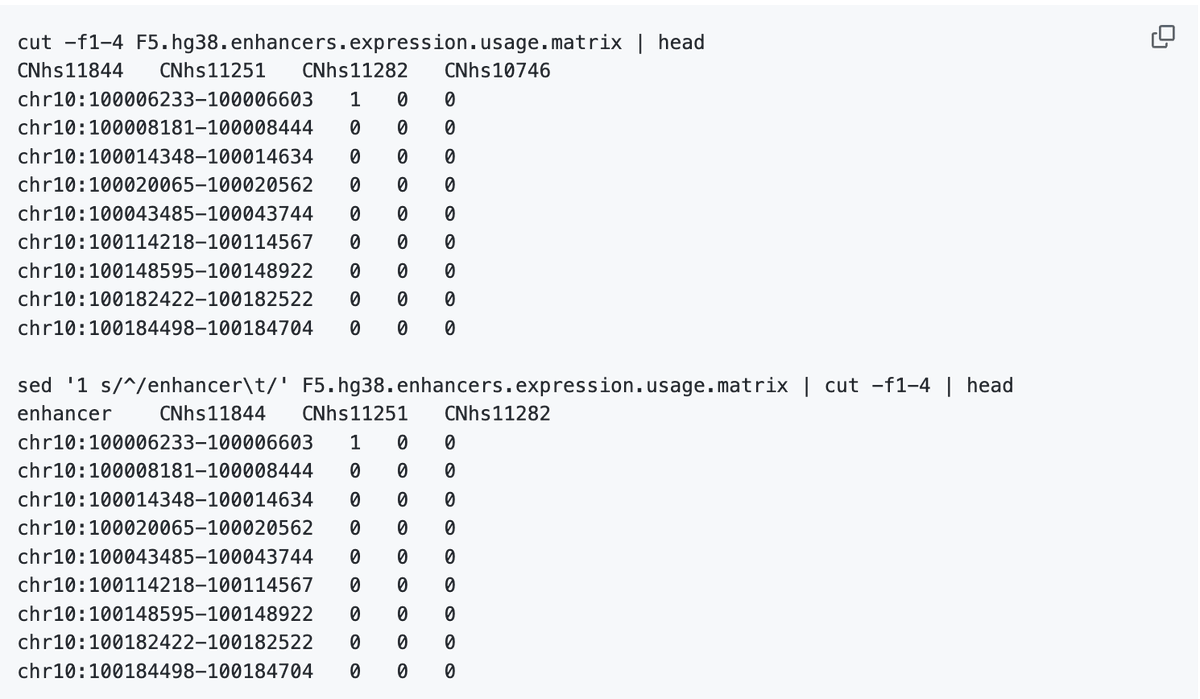
Missing Enhancer ID Header in 20GB Matrix
You just downloaded a 20GB enhancer x sample matrix. One problem: the first column header is missing. 1/ You stare at a giant matrix. Rows = enhancers. Columns = samples. But there’s no header for the enhancer ID column. https://t.co/rC6ciWOg9Q
AI‑Driven STAR System Enables First Biological Fatherhood for Azoospermic Man
Columbia University’s STAR (Sperm Tracking and Recovery) platform used artificial intelligence to isolate viable sperm from a man diagnosed with azoospermia, resulting in the first AI‑assisted conception and a baby expected in 2026. The breakthrough could reshape treatment for the...
Blood Test May Forecast Alzheimer’s Years Before Symptoms
A new study by researchers at Harvard-affiliated Mass General Brigham has found that a blood test has the potential to predict progression of Alzheimer’s disease ***years before*** symptoms or brain scan changes. https://t.co/WtghaB12r2

Vaccines Must Evolve for Climate, Megacities, Anti‑Science Era
May 14 I’m at @ucsc Univ California Santa Cruz delivering the annual Sinsheimer Distinguished Lecture in Biology on Global Vaccines in a time of Climate Change, Megacities, and Anti-science https://t.co/R55Ya6D6ys https://t.co/wc3KmeEFeO

Syngene International Names Abhijit Zutshi as Chief Commercial Officer to Drive Global CRO Growth
Syngene International Limited has appointed Abhijit Zutshi as Chief Commercial Officer. The 27‑year pharma veteran will oversee sales, marketing, client partnerships and digital initiatives, positioning the contract research organization for accelerated revenue growth and expanded global reach.

Trump’s Executive Order Spurs Ibogaine Hype as FDA Greenlights First Clinical Trial
President Donald Trump signed an executive order on April 18 to accelerate psychedelic access, and the FDA announced it will permit the first clinical trial of ibogaine. The political push has ignited a wave of public enthusiasm that far exceeds the...

The Bias in Medical Research: Africa Carries a Huge Disease Burden but Is Missing From Clinical Trials
A new analysis of 2,472 randomized controlled trials published between 2019 and 2024 reveals a stark under‑representation of Africa in top medical research. Only 3.9% of trials in the most prestigious general journals were conducted exclusively on the continent, and...

Truist Reaffirms Buy on Eli Lilly (LLY) After Trial Success
Truist Securities reaffirmed its Buy rating on Eli Lilly, keeping a $1,281 price target after the company reported positive Phase 3 data for its insulin candidate Foundayo. The ACHIEVE‑4 trial involving over 2,700 type 2 diabetes patients demonstrated non‑inferiority to insulin glargine and...

After Heart Attack, Therapeutic Plasma Exchange (TPE) Rescues the Aging Heart
Researchers at UC Berkeley demonstrated that therapeutic plasma exchange (TPE) performed 24 hours after a heart attack can nearly reverse damage in aged mice, the equivalent of humans in their 60s. By replacing half of the plasma with saline‑albumin solution, the...
NomosLogic Founder Matt Hardy Launches Lyceum and Odyssey on Dendrite Lite
NomosLogic unveiled two consumer‑facing experiences, Lyceum and Odyssey, on its Dendrite Lite platform. Lyceum delivers a personalized genomic‑literacy quiz generated from a user’s own DNA, while Odyssey presents the genome as a seven‑chapter narrative. Both tools leverage NomosLogic’s Hardy Bridge...

High-Dose Vitamin D Cuts Diabetes Risk in Genetically Susceptible, Lowers Alzheimer Biomarkers
Two peer‑reviewed studies published in April 2026 reveal that high‑dose vitamin D supplementation can slash type‑2 diabetes risk by 19% in people with specific vitamin‑D‑receptor gene variants and is associated with lower tau protein, a key Alzheimer’s biomarker. The findings...

ByteDance’s Anew Labs Unveils First AI‑Designed Small‑Molecule Immunotherapy
ByteDance’s drug‑discovery unit Anew Labs presented its first AI‑designed small‑molecule immunotherapy at the American Association of Immunologists conference in Boston. The generative‑AI compound targets IL‑17, a protein‑protein interaction long labeled undruggable, and could pave the way for oral treatments that...

Epigenetic Reprogramming Enters Human Trials for Vision
Aging biotech has made TIME's "New Frontiers" list. @lifebiosciences is using Yamanaka factors, and the FDA just cleared their first-in-human trial for optic neuropathies. Partial epigenetic reprogramming is no longer theoretical. It's in human trials. https://t.co/ZtVT5PxbHZ https://t.co/OhqLtNfqGT
Low Leukocyte mtDNA and High Inflammation Predict Elderly
Association of leukocyte mitochondrial DNA copy number and inflammation with mortality among older adults https://t.co/AeqAZIv3er
Celcuity's Gedatolisib Triplet Beats Alpelisib in Phase 3 PIK3CA‑Mutant Breast Cancer Trial
Celcuity Inc. announced that its Phase 3 VIKTORIA‑1 trial met its primary endpoint, showing a statistically significant and clinically meaningful progression‑free survival benefit for the gedatolisib‑fulvestrant‑palbociclib regimen over alpelisib‑fulvestrant in PIK3CA‑mutant HR+/HER2‑ advanced breast cancer. The data will be presented at...

Scientists Cautiously Suggest GLP-1s Are Safe to Use Around Pregnancy
A systematic review of more than 49,000 pregnancies over two decades found that exposure to GLP‑1 receptor agonists such as Ozempic, Wegovy and Mounjaro around conception does not increase the risk of major birth defects. The meta‑analysis of ten cohort...

Intellia’s In‑Vivo CRISPR Therapy Clears Phase 3, Prompting $1.9 B Chiesi Deal
Intellia Therapeutics announced that its single‑dose in‑vivo CRISPR therapy lonvoctocogene‑z cut hereditary angioedema attacks by 87% in a Phase 3 trial, the first such success for any in‑vivo CRISPR product. Two days later, Italy’s Chiesi Group acquired KalVista Pharmaceuticals for $1.9 billion,...

Blood Factors Drive Aging or Rejuvenation; Therapies Emerging
Aging is increasingly understood as a system-wide process shaped by factors in the blood, which can actively drive either decline or rejuvenation rather than just reflect it. Emerging therapies that modify the circulatory environment show promise in reversing aspects of...
New Test Promises to Detect Cancer Earlier, From Tiny Particles in Bodily Fluids
Researchers at the University of Calgary have unveiled EXOSense, a patent‑pending platform that electrically isolates small extracellular vesicles from blood or urine for cancer screening. These vesicles carry molecular signatures that appear long before conventional biomarkers, offering a potential route...

FDA Approves First‑In‑Human Trial of Motif Neurotech’s Depression Brain Implant
The U.S. Food and Drug Administration has granted Motif Neurotech permission to begin a first‑in‑human study of its miniature brain‑computer interface designed for treatment‑resistant depression. The Houston‑based startup says the device, the size of a blueberry, can be implanted in...

Vitamin D Study Split: 2,000 IU Boosts Aging Cells, While D2 May Undermine Immunity
A recent study found that daily 2,000 IU vitamin D supplementation helps preserve telomere length in older adults, suggesting a potential anti‑aging benefit. At the same time, researchers in the UK reported that vitamin D2 supplements can suppress vitamin D3...
Meta‑Analysis Shows GLP‑1 Drugs Cut Cardiovascular Events by 13% Over Three Years
Researchers at Anglia Ruskin University analyzed data from more than 90,000 patients and found GLP‑1 receptor agonists lower major adverse cardiovascular events by roughly 13% over an average three‑year follow‑up. The benefit appears across diabetic and non‑diabetic high‑risk groups, reinforcing...

FDA ODAC Backs AstraZeneca’s Truqap Combo for PTEN‑deficient Prostate Cancer
The FDA’s Oncologic Drugs Advisory Committee voted 7‑1 to recommend AstraZeneca’s Truqap (capivasertib) combined with abiraterone and androgen deprivation therapy for PTEN‑deficient metastatic hormone‑sensitive prostate cancer. The recommendation follows the CAPItello‑281 Phase III trial, which showed a 19% reduction in radiographic...

Esperion to Be Bought by ARCHIMED in $1.1 B Deal, Shares Surge 56%
Esperion Therapeutics agreed to be acquired by funds managed by ARCHIMED for up to $1.1 billion. Shareholders will receive $3.16 in cash per share—a 58% premium—plus contingent milestone payments that could add $100 million. The announcement sent the stock up 56% in...
FDA Grants Expanded Access to Revolution Medicines' Pancreatic Cancer Pill Daraxonrasib
The U.S. Food and Drug Administration approved an expanded‑access program for Revolution Medicines' experimental pancreatic cancer drug daraxonrasib, allowing eligible patients to receive the pill before formal approval. The move follows trial data that doubled median survival versus chemotherapy and...

BREAKING: 146 NEUROLOGICAL AND PSYCHIATRIC CDC/FDA SAFETY SIGNALS WERE BREACHED WITH COVID SHOTS
A recent Substack post by Nicolas Hulscher alleges that COVID‑19 mRNA vaccines breached 146 CDC/FDA safety signals, citing astronomical relative‑risk figures such as a 3,000‑fold increase in brain clots and a 7.4% national rate of cognitive disability. The author claims...

Weekly Biotech Roundup: Top Insights Curated by BiopharmIQ
Here is my favourite weekly 🧵- made by my dear friend @BiopharmIQ. This post is a comprehensive recap of the past week in BioTech and gathers the best related posts written by some of the smartest & brightest 𝕏 Bio...

Biotech Expands Beyond Disease, Merging Tech and Medicine
Excited to be speaking on Monday at Mass General Brigham MESH Core 2026 about how biotechnology is moving beyond just treating disease ! Will do my best to bridge tech and medical cultures -- should be fun/spicy🌶️ wish me luck...
Development Analysis and Strategic Insights From a 505(b)(2) Reformulation Product: Enzalutamide
Astellas used the 505(b)(2) pathway to reformulate enzalutamide (XTANDI®) from a 40 mg lipid‑filled soft capsule into 40 mg and 80 mg film‑coated tablets, aiming to cut pill burden. Five biopharmaceutic studies demonstrated comparable AUC between the two dosage forms, while the tablet...

NMN Curbs Antibody Attacks, Eases Bleeding Disorder
NEW CLINICAL TRIAL RESULT: NMN suppresses harmful antibody-driven immune attacks to alleviate a common bleeding disorder. Authors suggest it may help other autoimmune diseases like lupus and rheumatoid arthritis 🙏🧵 https://t.co/eMvefTFxZo

Longevity Pill May Redefine Human‑dog Bonds
YOUR NEXT DOG A new pill could soon extend dogs’ lives. How will that change our relationship with our pets? By @andersen 🔗https://t.co/u2cuBfKGuU https://t.co/Iaw4uoZfZe
Ten-Year Pivotal Data Demonstrate Long-Term Durability of Edwards Lifesciences’ Resilia Tissue
Edwards Lifesciences released 10‑year results from the COMMENCE aortic trial, confirming that its RESILIA tissue surgical valves retain high durability. At ten years, 97.9% of patients were free from structural valve deterioration and 97.8% avoided reoperation. The data also show...

AI's Measured Impact on Biopharma R&D Remains Limited
My 2c on this highly relevant @Loftus @WSJ on ai in biopharma R&D, an article that offers generally measured assessment of ai impact so far, and will not come as surprise to regular @timmermanreport readers…-> https://t.co/KPigSAw5RC
Zuckerberg Funds $500M AI to Model Human Cells
Mark Zuckerberg backs $500 million push to build AI models of human cells as part of long-term effort to cure disease. https://t.co/nLBDaUVj9Z