Know What's Happening in BioTech
Know What's Happening in BioTech
BioTech Pulse
EMAIL DIGESTS
Daily
Every morning
Weekly
Tuesday recap

Theolytics, an Oxford‑based biotech, secured an €8 million non‑dilutive Horizon Europe grant to fund its phase 2 OCTOPOD‑IV expansion trial of THEO‑260, a novel oncolytic therapy for advanced ovarian cancer. The grant, awarded after a rigorous review, will finance the phase 2a portion targeting high‑grade serous and endometrioid tumors. International collaborators include the Institute of Cancer Research and Clínica Universidad de Navarra. THEO‑260’s dual action aims to destroy cancer cells and cancer‑associated fibroblasts, addressing a major unmet need in stroma‑rich solid tumors.

In 2025, Europe’s EMA, China’s NMPA, and Japan’s PMDA each granted first‑time approvals for novel large‑molecule therapeutics. Oncology accounted for the largest share of these approvals, while endocrinology—driven largely by GLP‑1‑based agents—was the second biggest category. The approvals span a...

EveryONE Medicines announced its shutdown just a week after the FDA released draft guidance for its new bespoke pathway targeting personalized genetic therapies. The guidance requires each individualized drug to be submitted as a separate application, a burden that the...

Researchers demonstrated that black‑soldier‑fly (BSF) larvae can consume food waste, sewage sludge, and pig manure while substantially lowering the load of human‑pathogenic RNA viruses. In eight‑day trials, larvae grew across all feedstocks, and viral abundance dropped dramatically, though some viruses...

Researchers have identified that chronic cerebral hypoperfusion, a hallmark of vascular dementia, induces pathological TDP‑43 modifications—including cytoplasmic mislocalisation and hyperphosphorylation—in mouse and cell models. These changes mirror TDP‑43 proteinopathies observed in ALS, frontotemporal dementia, and Alzheimer’s disease, suggesting a shared...

Aspire Biopharma has teamed with particle‑engineering specialist Microsize to develop a sublingual powder formulation of alprazolam, aiming for faster anxiety relief than traditional tablets. The partnership leverages Microsize’s micronisation expertise and Pace’s excipient compatibility and stability services to accelerate development....

The FDA issued its first botanical drug guidance in 2004 and updated it in 2016, yet only four complex plant‑derived drugs have been approved to date. Ajna Biosciences, led by Joel Stanley, has secured clearance to begin a Phase 2 trial...

Vocxi Health has teamed with Forj Medical to shrink its MyBreathPrint breath‑analysis system from a tabletop prototype to a handheld device the size of a deck of cards. The platform leverages graphene‑based nano sensors and AI‑driven algorithms to detect disease‑linked...
Extracellular vesicles (EVs) are emerging as a versatile platform for regenerative medicine and targeted drug delivery. Researchers highlight their ability to transfer proteins, RNA, and lipids between cells, mimicking natural signaling pathways. Recent advances in isolation, engineering, and loading techniques...

The UK government announced nearly £48 million in equipment funding to accelerate commercial clinical trials across the NHS. The money will equip 51 NHS trusts and 79 primary‑care organisations with diagnostic tools, scanners and mobile research vans, with 60 % directed to...

Researchers from ETH Zurich, the Friedrich Miescher Institute and the Cantonal Hospital of Lucerne have 3D‑printed elastic ear cartilage that closely replicates natural tissue mechanics. Using patient‑derived cartilage cells embedded in a bio‑ink, they produced ear‑shaped constructs that retained shape...

Pharmaceutical firms are accelerating onshoring efforts, with more than $270 billion pledged for U.S. manufacturing since early 2025 and 80% of global producers weighing domestic or near‑shore options. Legislative moves such as the BIOSECURE Act and incentives like BARDA Project NextGen are...

Federal budget approvals for 2026 dramatically increase nondilutive capital for biotech, raising NIH funding to $49 billion and expanding programs like CDMRP and ARPA‑H. Seed‑stage companies are responding, with 38 % planning to rely on grants and other non‑equity sources. Simultaneously, mission‑driven...

In this episode, Daniel Levine talks with Feng Nying Zhang, a partner at McKinsey Shanghai, about Asia’s rapid rise as a global hub for biopharmaceutical innovation, highlighted by a McKinsey report showing the region’s share of innovative drug pipelines jumping...

Researchers using advanced imaging in mice discovered that oligodendrocyte precursor cells continuously differentiate into mature oligodendrocytes across the brain and spinal cord, regardless of myelin injury. This overturns the long‑standing belief that these cells only mature in response to damage....

MIT engineers have unveiled SAFARI, a biodegradable ingestible sensor that confirms pill ingestion using a bioresorbable Faraday cage and RFID tag. The device activates once the cage dissolves in the gastrointestinal tract and transmits a signal within about ten minutes....

Merck’s Keytruda remained pharma’s top‑selling drug in 2025, generating $31.7 billion and a 7 % year‑on‑year increase. However, the combined sales of GLP‑1 franchises—Eli Lilly’s tirzepatide line and Novo Nordisk’s semaglutide portfolio—totaled over $36 billion, overtaking Keytruda for the first time. The GLP‑1 market is...
A new Nature study sequenced the whole genomes of 1,364 breast cancers and linked the data to transcriptomics and real‑world treatment outcomes. The analysis showed that genome‑wide signatures such as homologous recombination deficiency, intratumoral heterogeneity, and copy‑number instability correlate with...

New Device Detects Brain Waves in Mini Brains Mimicking Early Human Development by @ShellyFan https://t.co/oMMhx9ux8Q https://t.co/6321RBopxf

Early-stage testicular cancer patients could soon benefit from a blood‑based assay that detects microscopic disease after orchiectomy. The CLIMATE study, led by WEHI and ANZUP, identified the microRNA marker miR‑371 as a reliable predictor of relapse, which occurs in roughly...

EyePoint reported Q4 2025 results, highlighting rapid enrollment of over 900 patients in two pivotal wet AMD Phase 3 trials (Lugano and LUCHIA) with top‑line data expected mid‑2026. The company also initiated Phase 3 DME studies (COMO and CAPREIT) slated for first...

Karyopharm Therapeutics reported Q4 2025 total revenue of $34.1 million, an 11.8% year‑over‑year increase, driven primarily by XPOVIO net product revenue of $32.1 million. The company trimmed R&D and SG&A expenses, improving operating performance by roughly 43% quarter‑over‑quarter, yet posted a GAAP...

Verastem reported $1.7 million in COPIKTRA net revenue for Q1 2019, a 38% increase over the prior quarter, and achieved reimbursement coverage with more than 92% of targeted health plans. Clinical updates highlighted the Phase 3 DUO trial, which showed...

Niagen Bioscience reported Q2 2025 revenue of $31.1 million, a 37% year‑over‑year increase, and lifted full‑year revenue guidance to 22‑27%. Gross margin improved to 65% driven by a higher‑margin sales mix and inventory efficiencies. The company expanded its Niagen Plus clinic...
![[Comment] Considerations for Improving Non-Inferiority Trials](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/0f1a72bb75d13d20256f6082d5abd222.webp)
Non‑inferiority trials are increasingly employed to evaluate new therapies that may offer advantages beyond efficacy, such as safety or convenience. However, their design and analysis present unique methodological challenges that can compromise study validity. The commentary outlines essential considerations, including...

Sino Biopharm’s subsidiary Chia Tai Tianqing Pharmaceutical Group has signed an exclusive global licensing deal with Sanofi for its blood‑cancer drug rovadicitinib. The agreement provides Sanofi with worldwide rights to develop, manufacture and commercialize the oral JAK/ROCK inhibitor, and includes...
An international team led by the University of Göttingen has created a microfluidics platform that streamlines multiplexed super‑resolution microscopy. The system automates precise fluid exchange, replacing manual pipetting, which enhances image quality and reproducibility across long imaging cycles. It was...
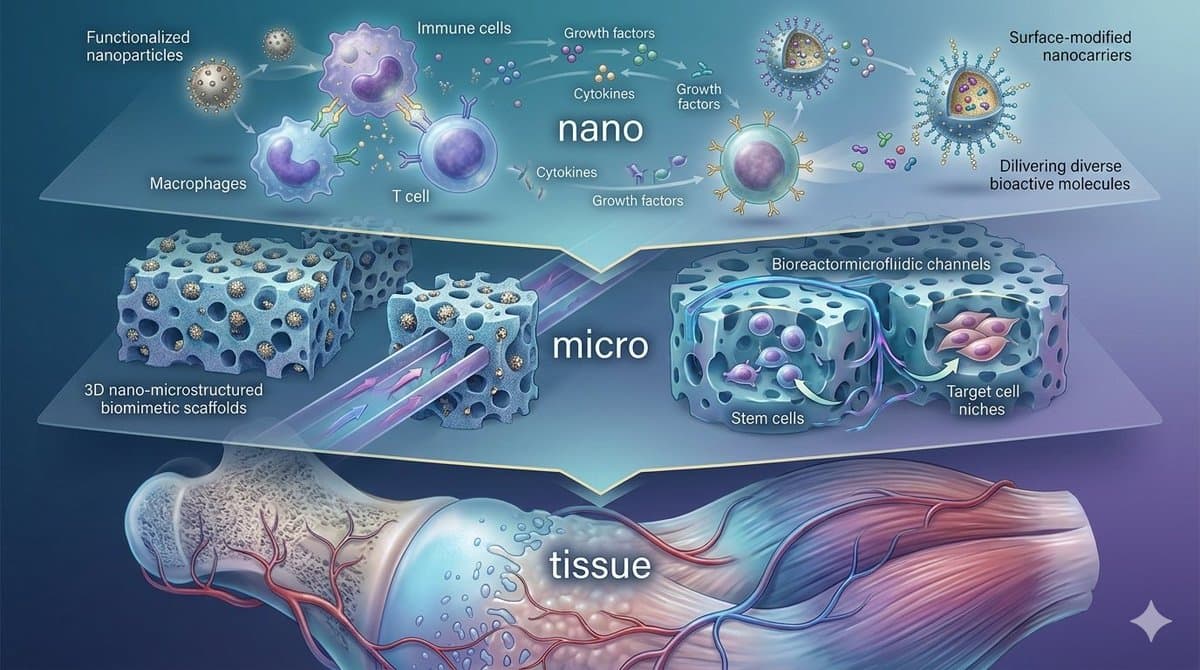
🚀 Call for Papers | Special Issue in Frontiers in Bioengineering and Biotechnology Nano–Microtechnology Enabled Immuno-Engineering and Multiscale Fabrication for Next-Generation Regenerative Medicine Join us in shaping the future of regenerative medicine at the convergence of nanotechnology, immuno-engineering, advanced biomaterials, and translational...
Following strong interest in the ASH25 hematologic malignancy intelligence report, Biotech Strategy Blog released a new strategic intelligence series covering the ASCO GU meeting data on prostate, bladder, and renal cell cancers. The first installment focuses on advanced prostate cancer,...

A team led by Associate Professor Tetsuya Muramoto at Toho University has created a CRISPR‑based genome‑editing platform that works in multiple social amoeba species. The vector, combined with donor DNA, dramatically improves editing efficiency, allowing gene disruptions in previously intractable...

The FDA has approved the first generic version of Flovent HFA, a fluticasone propionate inhalation aerosol delivering 44 µg per actuation for asthma maintenance in patients aged four and older. The generic, produced by Glenmark Specialty SA, matches the brand’s safety and efficacy...

AnaptysBio announced it will spin off its biopharma division into a new public company, First Tracks Biotherapeutics, in Q2 2026, separating it from the royalty‑focused parent that will retain the AnaptysBio name. The royalty side highlighted strong performance from GSK’s Jemperli,...

Oculis Holding AG reported a transformative 2025, highlighted by a $268.7 million cash position that extends its runway to 2029 and a $210 million equity raise. The company secured FDA Breakthrough Therapy designation for Privosegtor, its neuro‑protective candidate for optic neuritis, and...

A phase‑1 pilot of low‑dose intralesional cemiplimab in early‑stage cutaneous squamous cell carcinoma (CSCC) demonstrated rapid tumor regression with visual objective response rates of 66.7%–75% and pathologic complete responses of 58.3%–66.7%. Safety was favorable, with no grade ≥ 3 events and minimal...

Researchers identified a unique chromatin accessibility signature in peripheral blood mononuclear cells of centenarians, marked by widespread chromatin openness across multiple immune subsets. Unlike typical aging, which often links increased accessibility with senescence, centenarians maintain open promoters and enhancers in...

Tirzepatide: In the SURMOUNT-1 trial, weight reduction percentages were dose-dependent: 5 mg: ~15.0% weight reduction. 10 mg: ~19.5% weight reduction. 15 mg: ~20.9% weight reduction (Jastreboff et al., 2022). https://www.nejm.org/doi/10.1056/NEJMoa2206038 https://www.gatlan.com/ @GatlanHealth 

If you were Moderna, would you have gambled going in front of a jury made up of the general public these days? I don’t think so.

The FDA granted accelerated approval to navepegritide (Yuviwel), a once‑weekly CNP prodrug, for children aged two years and older with achondroplasia and open growth plates. In the phase 2b ApproaCH trial, the drug raised annualized growth velocity to 5.89 cm/year, a 1.49 cm/year...
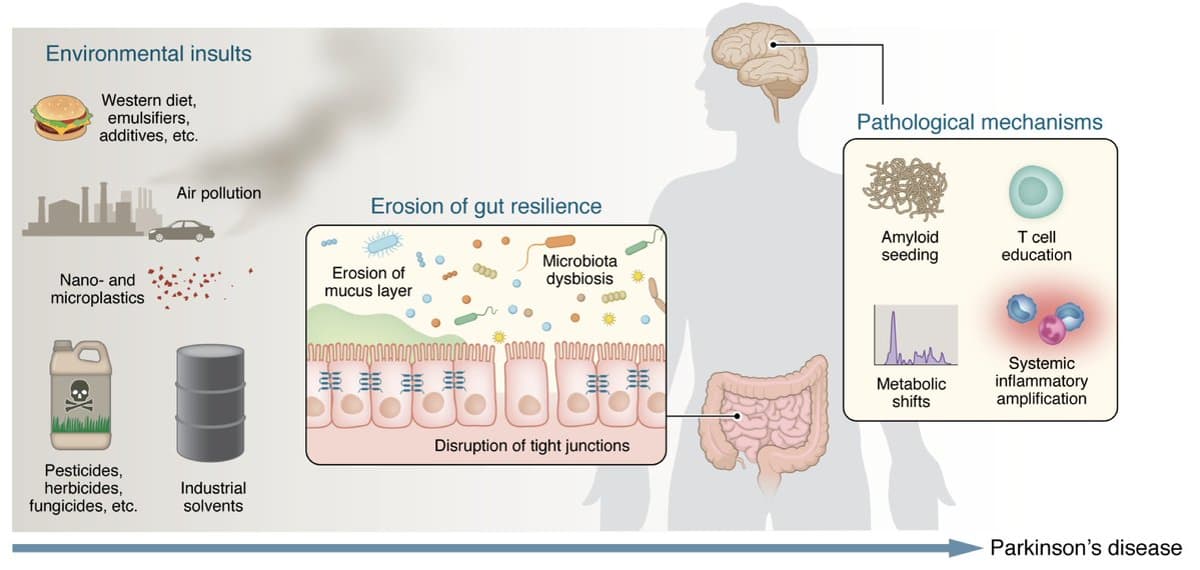
The gut and environmental toxins are key underpinnings of the Parkinson's disease pandemic @jclinicalinvest https://t.co/e8o4UuIafG https://t.co/7634Yg2Qo5

Join the Timmerman Traverse. WATCH alumni Ted Love, Amy Abernethy, Doug Fambrough and Bonnie Anderson. https://t.co/49ZC5b4mKy
New post‑hoc analyses of the phase 3 STOP‑HS1 and STOP‑HS2 trials show that oral povorcitinib, a selective JAK1 inhibitor, delivers rapid, high‑threshold lesion clearance in patients with severe hidradenitis suppurativa. In the 75 mg arm, up to 57% of participants achieved complete...
Prime Medicine $PRME to seek approval for gene editing treatment after two-patient trial https://t.co/nBnrUzqSCN via @Jasonmmast
Pierre Fabre seeks to revive US approval chances for spurned cell therapy https://t.co/gJeklT8asd @ByJonGardner $ATRA $QURE $RGNX
Kyowa Kirin announced it will cease all clinical trials of rocatinlimab, an anti‑OX40 antibody once touted as a potential eczema blockbuster, after a safety review identified emerging malignancy signals. The review, conducted with former partner Amgen, uncovered a confirmed and...

Scientists at Ludwig Maximilian University discovered that loss of the DJ-1 protein triggers ATP deficiency in human dopaminergic neurons, leading to reduced VMAT2 levels and impaired dopamine vesicle loading. The resulting dopamine oxidation fuels accumulation of pathological α‑synuclein species, a...

Did the FDA’s individualized therapy guidance put a spike in the heart of a startup biotech? Fake news, says the HHS. Yeah, it did, says an insider. Exclusive from @RLCscienceboss and @Jared_Whitlock https://t.co/IN9o4VIp03

Real‑world evidence from Duke’s PRECISION platform shows Pluvicto (Lu‑177 vipivotide tetraxetan) delivers a median progression‑free survival of 13.5 months in PSMA‑positive metastatic castration‑resistant prostate cancer (mCRPC) after androgen‑receptor pathway inhibitor (ARPI) therapy, matching the pivotal PSMAfore trial. Patients who received Pluvicto...

The FDA granted priority review to Takeda’s rusfertide, a first‑in‑class hepcidin mimetic for polycythemia vera, after Phase III trials more than doubled response rates. A regulatory decision is expected in Q3 2026. The agency also expanded Novo Nordisk’s once‑weekly Sogroya to treat children...

A March 2026 study in Pediatric Research applied whole exome sequencing to seven patients with methylmalonic acidemia (MMA) and cystathionine beta‑synthase (CBS) deficiency, uncovering both known and novel pathogenic variants in MMUT, MMAA and CBS genes. The researchers linked specific mutation...
Researchers have engineered a nanocomposite incorporating silver (Ag) nanoparticles that markedly improves anticancer efficacy. The hybrid material boosts reactive oxygen species generation and enhances tumor cell uptake while sparing normal tissue. In vitro studies show synergistic activity with standard chemotherapeutics,...