Replimune Reports 3‑Year Survival Benefit for RP1 + Nivolumab in Anti‑PD‑1‑Resistant Melanoma
Replimune Group presented IGNYTE trial results at the 2026 ASCO meeting, showing a median overall survival of 32.9 months and nearly half of patients alive at three years. The RP1 plus nivolumab regimen delivered a 33.6% response rate with durable responses, offering a new option for melanoma patients who have progressed on anti‑PD‑1 therapy.

FDA Clears MannKind's Afrezza Inhalable Insulin for Kids 6+, Expanding Pediatric Diabetes Options
MannKind Corp. announced that the U.S. Food and Drug Administration has approved Afrezza inhalable insulin for children and adolescents aged six and older with type 1 or type 2 diabetes. The decision, based on the pivotal INHALE‑1 trial and two decades of...

This Discovery Could Change How Often You Need Ozempic Shots
A NIH team published in Nature Metabolism that semaglutide’s weight‑loss power depends on how long it sustains cyclic AMP (cAMP) spikes in the brain’s area postrema. Real‑time imaging of mouse neurons showed some cells keep elevated cAMP for hours while...
Long-Term Leukemia Trial Reveals MRD-Triggered Treatment May Slow or Prevent Relapse
The RELAZA2 trial has released its long‑term data, confirming that azacitidine administered at the first sign of measurable residual disease (MRD) can significantly delay relapse in myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML). As the world’s first prospective MRD‑triggered...

Tempus Unveils the Next-Generation of Lens, Expanding Its Agentic AI Platform for Oncology Drug Development
Tempus AI launched the next‑generation Lens, an agentic AI platform that unifies its multimodal real‑world oncology data, high‑performance computing and validated AI agents. The cloud‑based system lets drug developers create research plans in plain language, execute analyses on more than...
New Pancreatic Cancer Drug Shows Survival Boost
People who don't follow cancer research often ask me why we haven't cured cancer. That perception masks a wonderful reality: We make amazing, stepwise progress every year, and the result is that many people live much longer today than they...

Standing Ovation for Daraxonrasib Survival Benefit at ASCO26
Incredible #ASCO26 moment. Dr. Brian Wolpin, presenter of the daraxonrasib study, received a standing ovation DURING his talk after he stated the survival benefit for PDAC patients. It was sustained. Cheering. I have never see anything like it in the...
Pfizer’s BRAFTOVI Regimen Nearly Doubles Median Progression-Free Survival in Metastatic Colorectal Cancer
Pfizer reported that its BRAFTOVI (encorafenib) regimen combined with cetuximab and FOLFIRI halved the risk of disease progression or death in patients with BRAF V600E‑mutant metastatic colorectal cancer. Median progression‑free survival more than doubled to 15.2 months versus 8.3 months for...

Kelonia Therapeutics Presents Updated First-in-Human Data From Phase 1 inMMyCAR Study of KLN-1010 in Vivo BCMA CAR-T Therapy at the...
Kelonia Therapeutics reported updated Phase 1 inMMyCAR data for its in‑vivo BCMA CAR‑T candidate KLN‑1010 at ASCO 2026. All 18 patients dosed showed a 100% overall response rate with MRD‑negative bone marrow, and the earliest‑treated patient remains disease‑free beyond ten months....
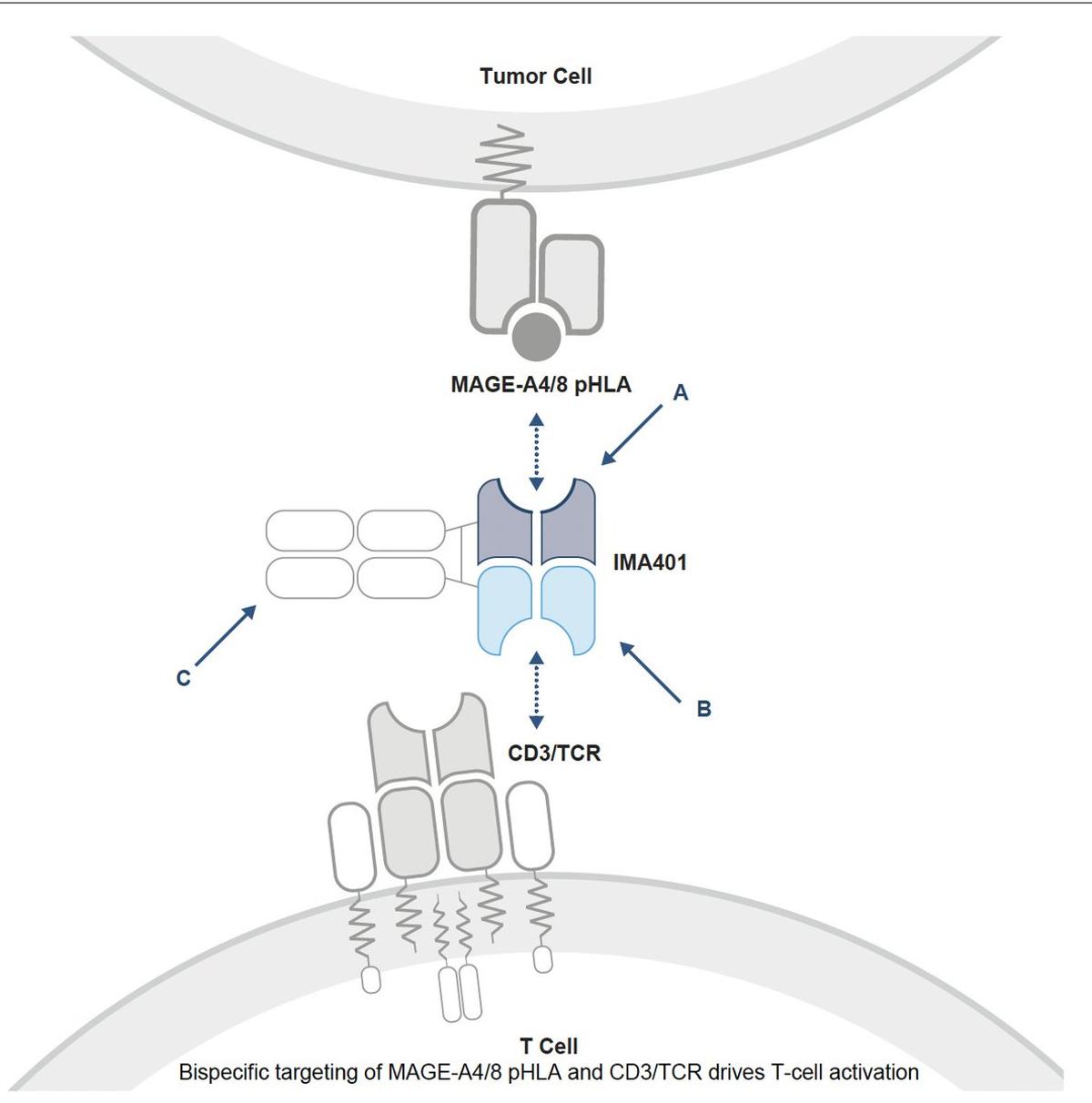
Bispecific T‑cell Engager Shows Promising Responses in Refractory
Just published @NatureMedicine and presented @ASCO Advanced, refractory solid cancers treated with a bispecific T cell engager (BiTE, Figure), Phase 1 trial, with or without Keytruda, in 61 patients. Some very favorable responses with "manageable safety profile" https://t.co/MkzQXY1psA
Lanatoside C Shows Senolytic Activity and Cuts Atherosclerosis in Mice
Researchers screened 2,150 FDA‑approved drugs and identified lanatoside C as a senolytic that eliminated senescent cells and lowered atherosclerotic plaque in mice, highlighting a new therapeutic avenue for age‑related vascular disease.

Eli Lilly's Retatrutide Triggers 30%+ Weight Loss in Late‑Stage Trial
Eli Lilly announced that its next‑generation GLP‑1 candidate Retatrutide produced weight loss of 30% or more in a late‑stage study, eclipsing results from existing injectables. The outcome could redefine the company's growth trajectory as it battles Novo Nordisk in the...

Pfizer’s TALZENNA‑XTANDI Combo Cuts Progression Risk 52% in HRR‑Mutated Prostate Cancer
Pfizer announced that its Phase 3 TALAPRO‑3 study found the TALZENNA (talazoparib) and XTANDI (enzalutamide) combination reduced radiographic progression or death by 52% versus XTANDI alone in men with HRR‑mutated metastatic castration‑sensitive prostate cancer. The data, presented with three‑year rPFS...

On KRAS Inhibitors and Why Potency Doesn’t Equal Durability
The latest ASCO data on second‑generation KRAS inhibitors reveal that higher potency does not automatically translate into longer patient responses. While first‑gen G12C agents such as sotorasib showed modest durability, new G12D compounds demonstrate improved binding but still face resistance...
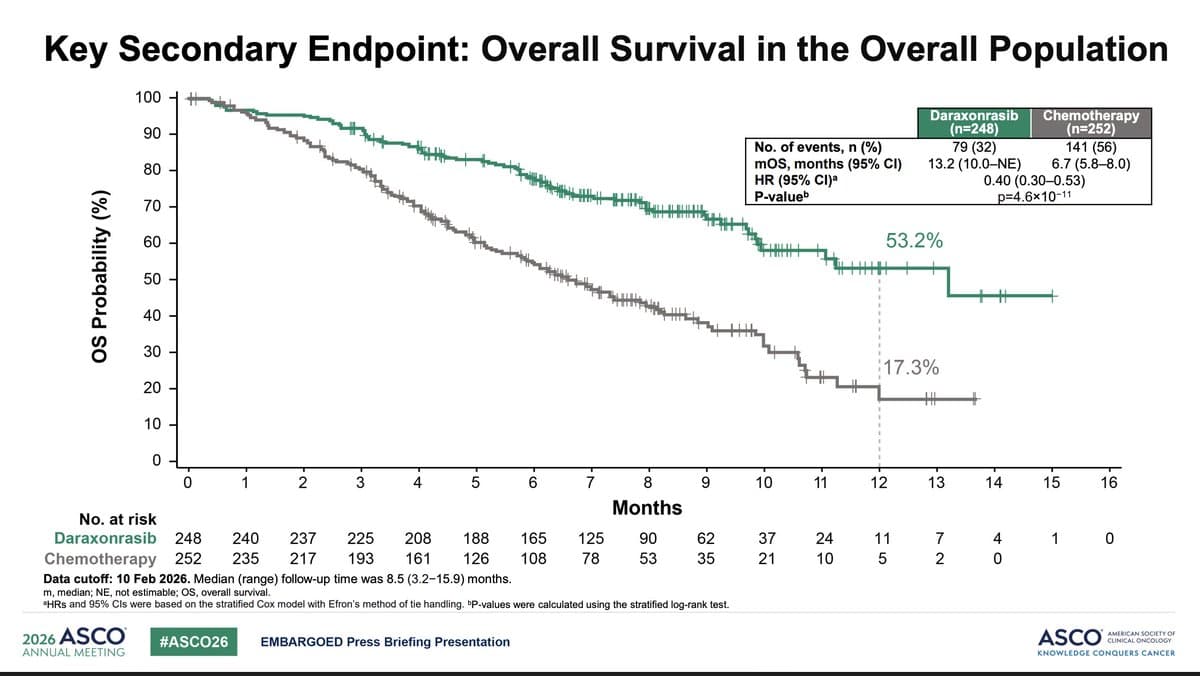
Experimental Pill Promises New Hope for Deadly Pancreatic Cancer
Researchers reported that the experimental pill daraxonrasib, which blocks mutated KRAS proteins, nearly doubled median survival for patients with advanced pancreatic cancer to 13.2 months versus 6.7 months on chemotherapy. The drug showed fewer severe side effects and improved quality...
RASolute 302 Brings a “Transformative" Moment in Pancreatic Cancer: A 60% Improvement in Overall Survival
Revolution Medicines’ RASolute 302 phase‑3 trial showed that the RAS(ON) inhibitor daraxonrasib improved overall survival by 60% in patients with previously treated metastatic pancreatic ductal adenocarcinoma. Median overall survival more than doubled to 13.2 months versus 6.6 months on standard chemotherapy,...
STAT+: For Prostate Cancer Patients Set on Surgery, New Hormone Regimen May Improve Outcomes, Study Finds
A phase‑3 PROTEUS trial found that administering two hormone therapies before and after prostatectomy outperformed a single‑hormone regimen in high‑risk, early‑stage patients. The dual approach reduced biochemical recurrence and improved margin‑negative resection rates. Oncologists view the data as a potential...
Oculis' Phase 3 OCS-01 Eye‑Drop Trial Fails in Diabetic Macular Edema, Halts FDA Push
Oculis Holding AG announced that its Phase 3 DIAMOND-1 and DIAMOND-2 trials of OCS-01 eye drops failed to meet the primary visual‑acuity endpoint and the key secondary 15‑letter gain endpoint in diabetic macular edema. The company will not pursue an...

NeuExcell Therapeutics Announces Encouraging Data of NXL-004 in Recurrent Malignant Glioma at 2026 ASCO
NeuExcell Therapeutics announced that its first‑in‑human gene therapy NXL‑004 for recurrent malignant glioma earned a Rapid Oral presentation slot at ASCO 2026. The AAV‑NeuroD1 platform was tested in eleven patients, showing a favorable safety profile with no drug‑related serious adverse...

Retatrutide Is Bringing a Hard Look at Health Outcomes
Eli Lilly’s phase‑3 TRIUMPH‑1 trial showed retatrutide delivering unprecedented weight loss, with participants on the top dose shedding an average of 28.3% of body weight after 80 weeks and 45.3% achieving at least a 30% reduction. In a two‑year extension...
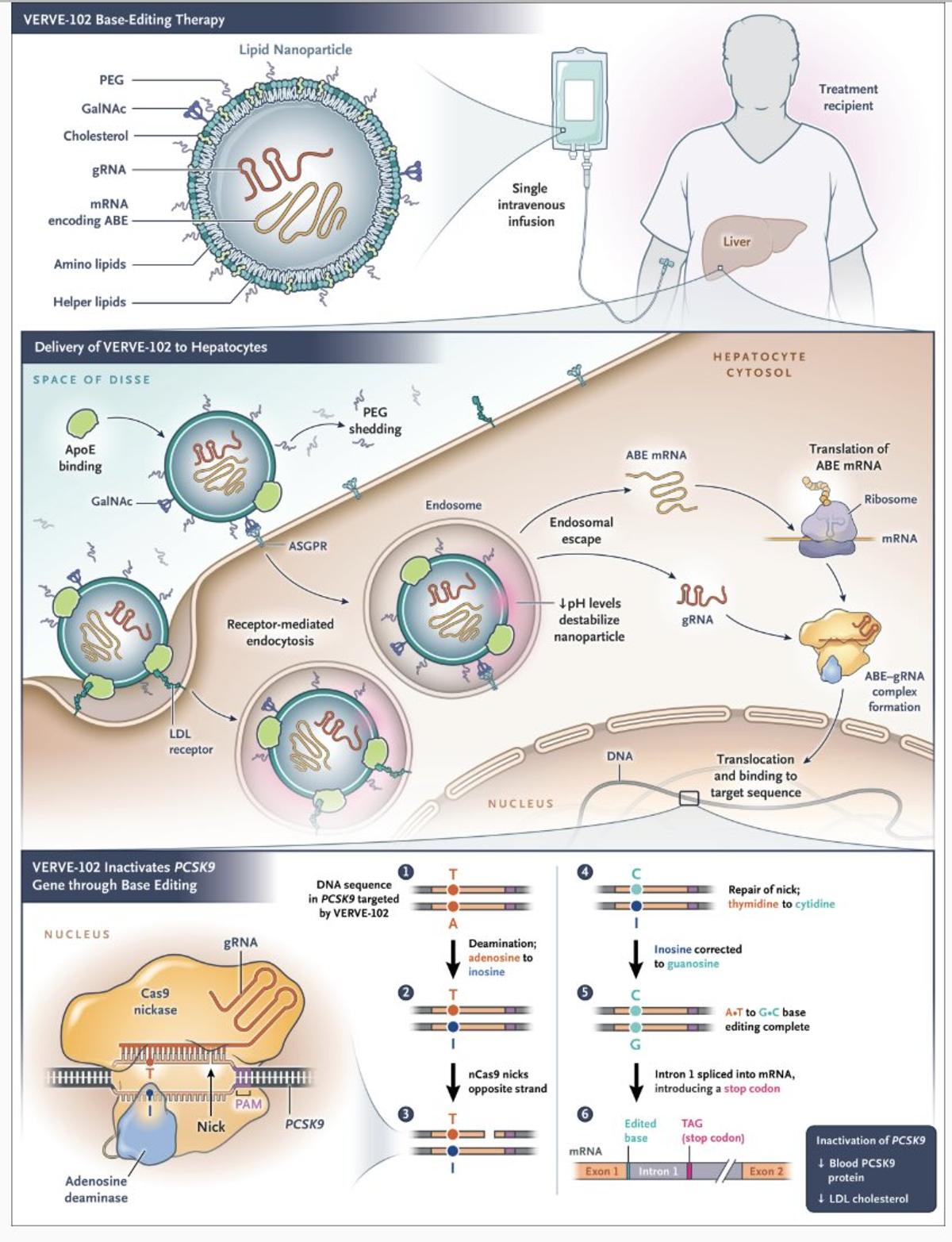
Weekly Reads: Effective LDL Gene Editing, Chinese Genetics Guidelines, Human Embryo Models in Space
A single‑infusion base‑editing drug (VERVE‑102) lowered LDL cholesterol in a small human study, sparking talk of a potential one‑time cure for high cholesterol. Parallel research unveiled universal transcriptomic aging clocks that can forecast mortality, advancing precision longevity metrics. Meanwhile, China...
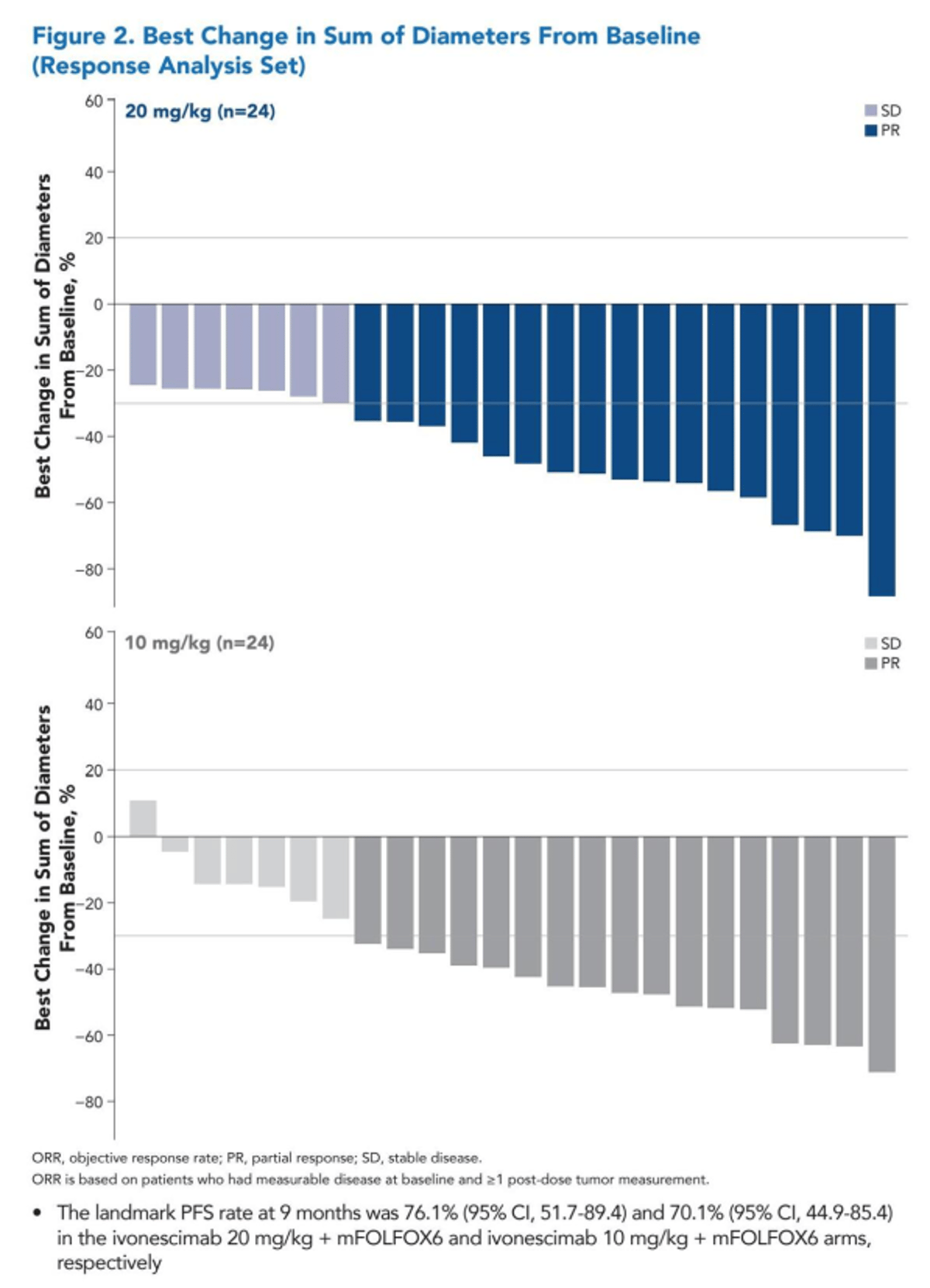
Ivonescimab Plus Chemo Yields 71% Response In
Akeso ph2 trial of ivonescimab + chemo in 1L MSS/pMMR colorectal cancer (incl KRASm & w/t). ORR 71% (32/48). Via David Berz #ASCO26 $SMMT https://t.co/i5NW6fpKAu
Real-World Predictors of Survival and Response in Advanced Melanoma
Immunotherapy has transformed advanced melanoma care, yet many patients fail first‑line treatment. Researchers applied machine‑learning to a large, real‑world electronic health record database to build interpretable models for overall survival, progression‑free survival, and response. ECOG performance status, PD‑L1‑based first‑line therapy,...
HIV in South Africa: Why Rolling Out a Groundbreaking New Shot Will Miss a Critical Group of Men
The U.S. shipped the first batch of lenacapavir, a long‑acting injectable HIV‑prevention shot, to South Africa in early April 2026, with rollout slated for June. Clinical trials show close to 100% efficacy with just two doses per year. The national...

Lupin Wins FDA Nod for Generic Sutab Colon Prep, Targeting $132.8M Market
Lupin Limited has secured U.S. FDA approval for its generic version of Sutab colon‑preparation tablets, becoming the exclusive first‑to‑file applicant and earning a 180‑day exclusivity period. The product targets a market that generated roughly $132.8 million in U.S. sales in 2026,...

Johnson & Johnson Gains FDA Approval to Expand TREMFYA Label for Psoriatic Arthritis
Johnson & Johnson announced that the FDA approved a supplemental Biologics License Application expanding TREMFYA’s label to include inhibition of structural joint damage in adults with active psoriatic arthritis. The update makes TREMFYA the only IL‑23 inhibitor with proven disease‑modifying...

Biomerica Signs $1.75 Million Service Deal to Build New IVD Assays
Biomerica, Inc. (BMRA) entered a three‑year Master Services Agreement with a private life‑science firm to develop proprietary in‑vitro diagnostic (IVD) assays, with an initial development target fee exceeding $1.75 million. The work will be performed at Biomerica’s FDA‑licensed facility, positioning the...
ASCO 2026: Bayer Shares New Findings for NUBEQA® (Darolutamide) Regarding Cognitive Decline in Men with Advanced Prostate Cancer Compared to...
Bayer presented Phase II ARACOG trial data at ASCO 2026 showing that NUBEQA® (darolutamide) significantly limited cognitive decline in men with advanced prostate cancer compared with enzalutamide. Over 24 weeks, the median maximal cognitive domain change was –15.8% for darolutamide versus...
NeuroPace Secures FDA Clearance for ECoG Assistant, First AI‑Driven Epilepsy Tool
NeuroPace announced FDA approval of its ECoG Assistant, an AI‑enabled clinician tool that scans intracranial EEG data to flag episodes of interest. Developed on 124,450 expert‑labeled recordings, the clearance paves the way for faster, data‑driven epilepsy treatment decisions.
Phase III Trial Shows Sacituzumab Tirumotecan Boosts Lung Cancer Survival
Sichuan Kelun-Biotech reported that its antibody‑drug conjugate sacituzumab tirumotecan, combined with pembrolizumab, more than doubled 12‑month progression‑free survival in a Phase III trial of 413 NSCLC patients. The data, unveiled at the ASCO meeting in Chicago, could reshape first‑line treatment...
Altimmune Reports 23.7% Triglyceride Drop in Phase 2b MASH Trial
Altimmune Inc. released 48‑week Phase 2b IMPACT trial results showing pemvidutide cut triglycerides by 23.7% and cholesterol by 15.4% in 212 MASH patients, while participants lost an average of 7.5% body weight. The data were unveiled at the 2026 EASL...
COXFA4L2 Boosts Cytochrome C Oxidase in Leigh Syndrome
A new Nature Communications study reveals that the mitochondrial protein COXFA4L2 is up‑regulated in cells with COXFA4 mutations, preserving cytochrome c oxidase activity in Leigh‑like encephalopathy. Cryo‑EM shows COXFA4L2 integrates into complex IV, maintaining electron transfer despite the genetic defect. Functional assays...
Tobevibart + Elebs
Vir’s tobevibart + elebsiran hit 65.9% undetectable HDV RNA at week 24 in the SOLSTICE trial. Big step toward a potential accelerated approval for hepatitis D. HepatitisD
Salubris Biotherapeutics Announces Updated Phase 1/2 Data for JK06, a 5T4-Targeted Antibody Drug Conjugate, at the 2026 American Society of...
Salubris Biotherapeutics presented updated Phase 1/2 data for JK06, a 5T4‑targeted antibody‑drug conjugate, at the 2026 ASCO meeting. The study treated 173 patients across dose‑escalation and expansion cohorts, reporting a 50% objective response rate (ORR) in squamous non‑small cell lung cancer...

Rapamycin Signalling Profile on THP-1
A 24‑hour, 10 µM rapamycin treatment of the THP‑1 monocytic leukemia cell line triggers a complex transcriptional rewiring. Negative feedback loops in the PI3K/AKT/mTOR axis are released, leading to up‑regulation of PIK3R1, AKT isoforms and mTORC1 scaffolds while FOXO3 mRNA is...

Predicting Drug Side Effects via LLM Pharmacology
A new study published in Scientific Reports introduces PromptSE, a framework that uses large language models (LLMs) to predict drug side effects from textual pharmacological descriptions. By converting chemical and mechanistic data into prompts, PromptSE outperforms traditional classification models in...
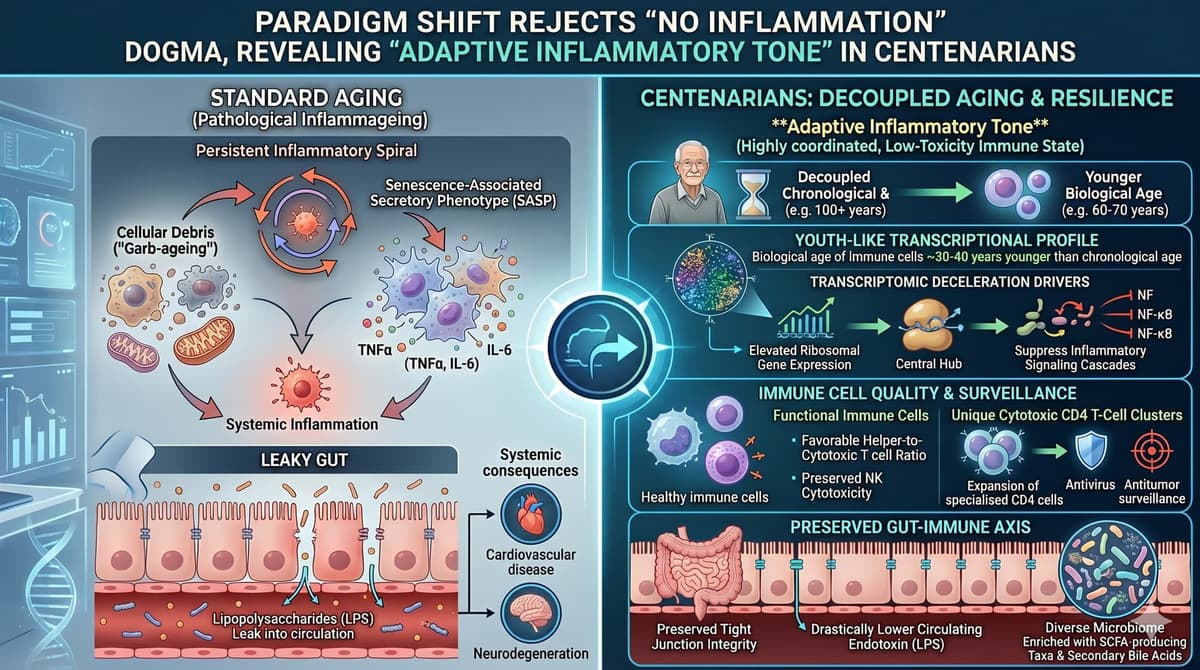
The Century-Old Immunome: Learning From the Adaptive Shield of Human Centenarians
The article outlines translational strategies to mimic centenarians’ elevated RNASEH2C activity, which clears cytoplasmic RNA:DNA hybrids and dampens chronic inflammation. It proposes four therapeutic levers: epigenetic maintenance to prevent RNASEH2C hyper‑methylation, delivery of centenarian‑derived extracellular vesicles, upstream protection of mitochondrial...

Russia Unveils 24‑Nanometer Quantum‑Dot Platform for Early Cancer Detection
Russian scientists at Sirius University of Science and Technology announced a nanoplatform that uses 24‑nanometer quantum‑dot particles to illuminate tumors in the infrared spectrum, enabling earlier cancer detection. The device also triggers necrotic cell death, opening a pathway toward novel...
Labcorp Teams with Alliance for Clinical Trials to Boost Genetic Testing in Colorectal Cancer
Labcorp Holdings announced a collaboration with the Alliance for Clinical Trials in Oncology to launch a national, multicenter trial focused on expanding germline and multigene panel testing for newly diagnosed colorectal cancer patients. The study, sponsored by the National Cancer...
Targeting BCL2: New Hope for Pancreatitis Therapy?
Researchers have identified the anti‑apoptotic protein BCL2 as a therapeutic target for acute pancreatitis, a condition that currently lacks disease‑modifying drugs. Preclinical studies using a selective BCL2 inhibitor demonstrated a marked reduction in pancreatic inflammation and cell death. Building on...

ARACOG Trial Links Enzalutamide to Greater Cognitive Decline Compared With Darolutamide: Alicia Morgans, MD, MPH
The phase‑2 ARACOG trial directly compared the cognitive impact of two androgen‑receptor pathway inhibitors in advanced prostate cancer. At 24 weeks, patients on darolutamide experienced a 15.8% decline on a neuropsychological test versus a 36.1% decline with enzalutamide. The advantage...

Eikon Therapeutics Presents Data on Clinical-Stage Programs at the 2026 Annual Meeting of the American Society of Clinical Oncology
Eikon Therapeutics presented six ASCO 2026 abstracts highlighting late‑stage data for its lead candidates. In the Phase 2 TeLuRide‑005 trial, the TLR7/8 dual agonist EIK1001 combined with pembrolizumab and chemotherapy achieved a 63.1% objective response rate and a 90.8% disease‑control rate...

Global Data for BioNTech and Bristol Myers Squibb’s PD-L1xVEGF-A Bispecific Pumitamig Shows Encouraging Efficacy in Patients with Non-Small Cell Lung...
BioNTech and Bristol Myers Squibb reported interim Phase 2 data from the global ROSETTA Lung‑02 trial of their bispecific PD‑L1×VEGF‑A immunomodulator pumitamig combined with chemotherapy in treatment‑naïve advanced NSCLC. Among 40 evaluable patients, the regimen yielded a confirmed objective response rate...
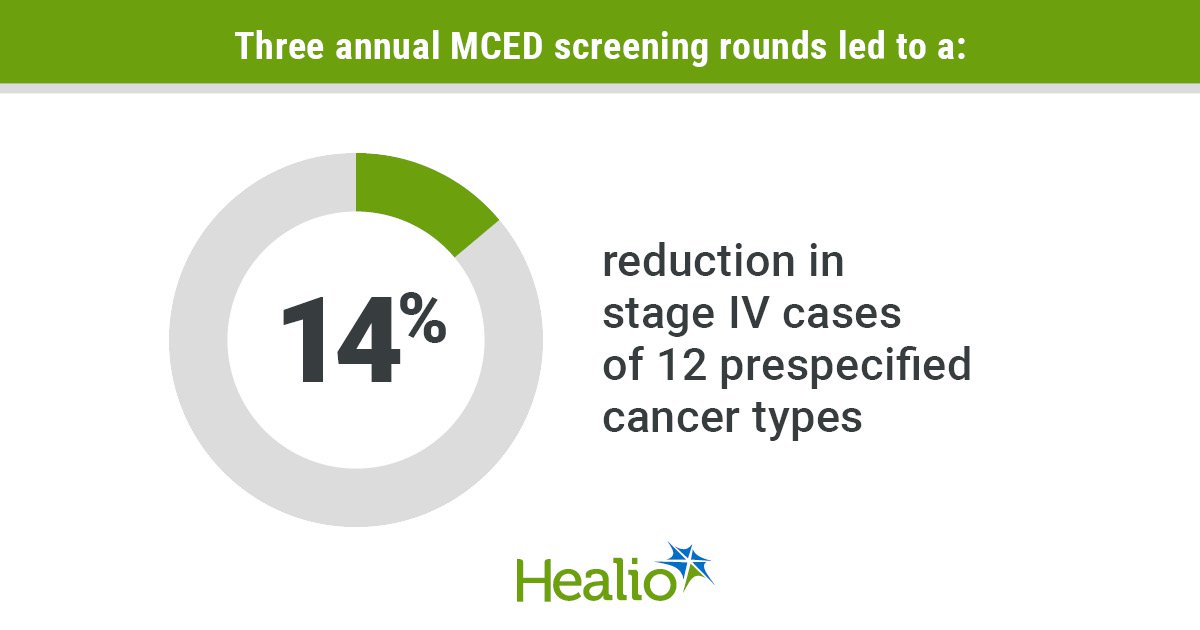
Trial of Early Cancer Detection Test Misses Primary Endpoint
The NHS‑Galleri randomized trial evaluated a multicancer early detection (MCED) blood test in 142,942 adults aged 50‑77. After three annual screens, the study failed to meet its primary endpoint of reducing combined stage III/IV diagnoses for 12 prespecified cancers. However, secondary...
Tezepelumab Helps Severe Asthma Patients Reduce Oral Steroids over 28 Weeks
A Phase III SUNRISE trial published in The Lancet Respiratory Medicine shows tezepelumab enables severe asthma patients to sharply cut their reliance on oral corticosteroids. Over 28 weeks, 69% of participants on the drug achieved at least a 50% dose reduction...

ITM Announces Phase 3 COMPETE Patient-Reported Quality of Life Data with N.c.a. ¹⁷⁷Lu-Edotreotide (ITM-11) Vs. Everolimus at ASCO 2026
ITM Isotope Technologies presented Phase 3 COMPETE patient‑reported quality‑of‑life data at ASCO 2026, showing that non‑carrier‑added ¹⁷⁷Lu‑edotreotide (ITM‑11) outperformed everolimus in gastroenteropancreatic neuroendocrine tumors (GEP‑NETs). In a cohort of 309 patients, the radiopharmaceutical maintained QoL scores (+0.9) while everolimus saw a decline...
Exelixis Announces Results From Subgroup Analysis of Phase 3 CABINET Pivotal Trial Evaluating CABOMETYX® (Cabozantinib) in Non-Functional and Functional Neuroendocrine...
Exelixis presented a subgroup analysis of the phase 3 CABINET trial at ASCO 2026, showing that CABOMETYX (cabozantinib) significantly extends progression‑free survival (PFS) in advanced neuroendocrine tumors (NET) regardless of functional status. In non‑functional NET, median PFS was 9.4 months versus 3.1 months on...

Massive Bio Announces Next-Generation Reticulum Nexus™ Product Suite at ASCO 2026
Massive Bio unveiled the next‑generation Reticulum Nexus™ suite at ASCO 2026, integrating six AI‑driven agents—Patient Connect™, TrialRelay™, NexusPulse™ calculators, Sentinel Agents™, DrArturo AI™ and Phoebe AI™—into a real‑time orchestration layer for precision oncology. The platform moves patients from fragmented trial...
GSK’s Bepirovirsen Achieves Functional Cure in 19% of Hepatitis B Patients in Phase III Trials
GSK and Ionis reported that bepirovirsen produced a functional cure in 19% of participants across two Phase III studies of chronic hepatitis B. The antisense oligonucleotide cleared viral DNA for at least six months after treatment stopped, marking the first such result...
Tempus AI Shows Zero‑Shot Predictive Power in Oncology with Multimodal Foundation Model
Tempus AI announced early results from its multimodal foundation model at the 2026 ASCO meeting, showing zero‑shot stratification of overall and progression‑free survival in EGFR‑mutant lung cancer. The model, trained on 2.5 million patient records, outperformed traditional Cox‑PH approaches and could...