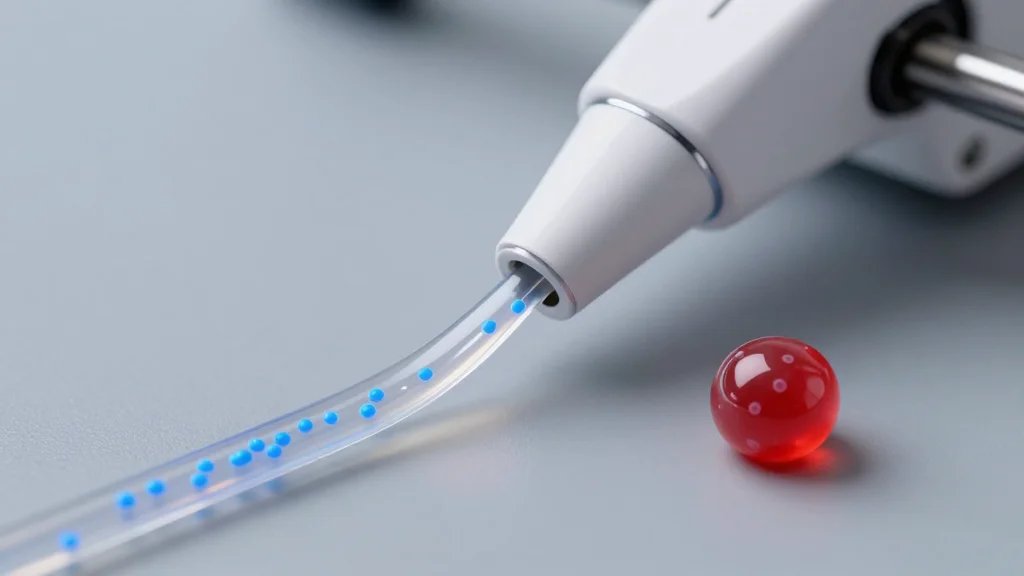
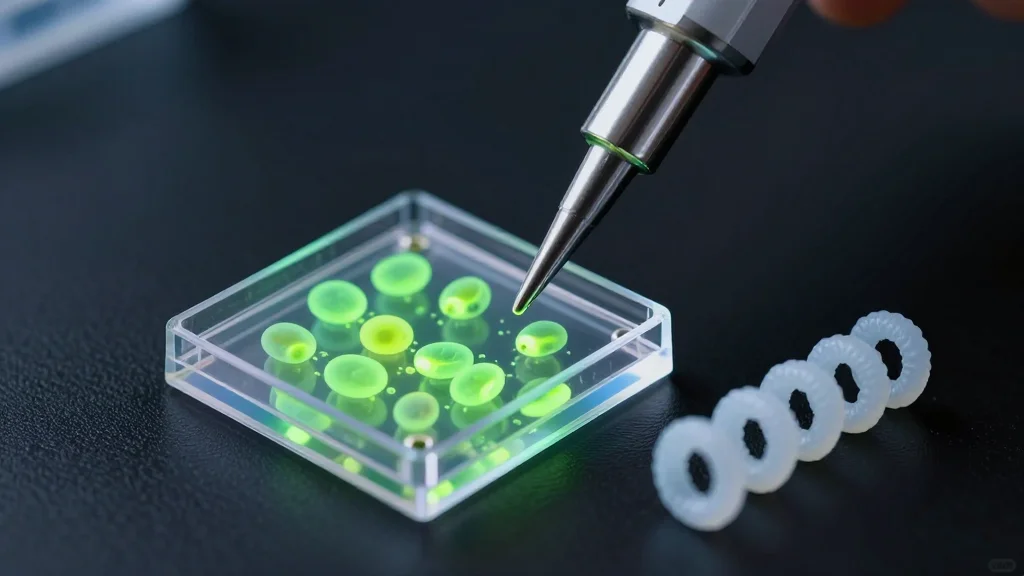
3D-Printed 'Spanlastics' Could Change How Cancer Drugs Reach Tumors
University of Mississippi researchers unveiled a FRESH 3D‑printing technique that fabricates hydrogel‑based spanlastic nanocarriers, 200–300 nm in size, loaded with anticancer drugs such as doxorubicin. The printed implants can be placed directly onto tumor sites, delivering high‑dose therapy locally while shielding healthy tissue. In vitro experiments on breast cancer cells demonstrated potent cytotoxicity, suggesting a pathway to reduce the systemic side effects typical of conventional chemotherapy. The study marks a first step toward personalized, localized cancer treatment, pending in‑vivo validation.
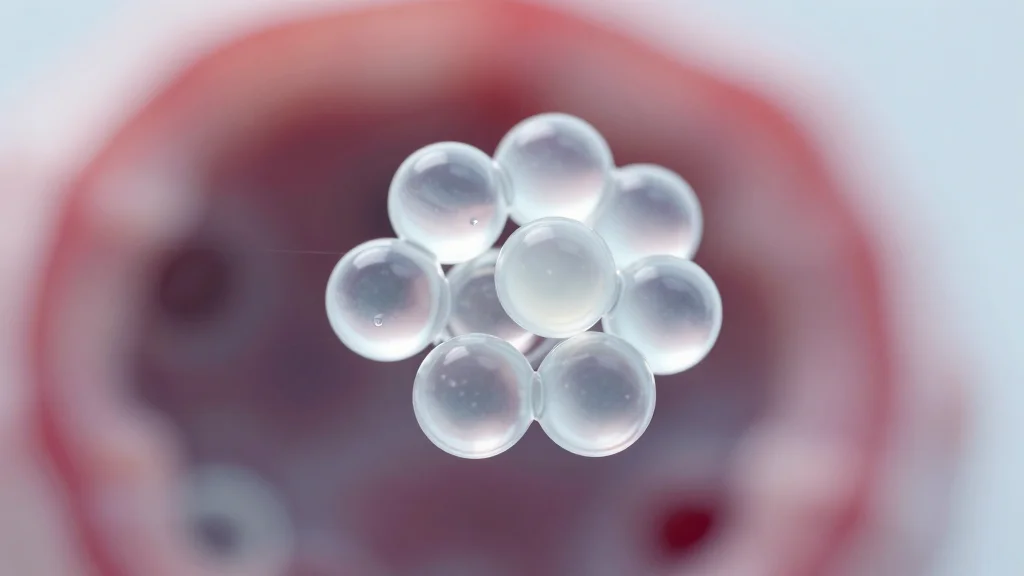
3D‑Printed Spanlastics Directly Target Tumors, Cut Side Effects
3D-printed spanlastics, microscopic carriers for cancer drugs, show potential to target tumors directly and reduce side effects by concentrating therapy at the tumor site rather than throughout the body. nanotechnology

Foundation of HER2 Gastric Cancer ASCO/CAP Guidelines—Overview of the Differences Between ASCO-CAP Breast and Gastric Cancer Scoring-January 28, 2026
The on‑demand webinar hosted by CAP TODAY on January 28, 2026, featured Dr. Josef Rüschoff, a leading authority on HER2 testing, to explain the newest ASCO/CAP guidelines for HER2 assessment in gastric cancer. It highlighted why HER2 is a critical therapeutic target beyond breast...
2Q Markets Preview, Tariffs and Biotech Takeouts — a BioCentury Podcast
Biotech remains resilient as markets wobble, with M&A activity persisting. The BioCentury podcast highlighted two major pending deals: Eli Lilly’s $6.3 billion acquisition of Centessa Pharmaceuticals and Biogen’s proposed $5.6 billion purchase of Apellis. Analysts also examined the lingering effects of Trump-era tariffs...
Creating Autonomous Health via Real-Time Multi‑Omic Monitoring
I've measured my body a lot. I'm about to dwarf what we've done by building real-time continuous multi-omic monitoring and intervention. Cars drive themselves. Software writes itself. I'm building Autonomous Health. First for me, then for you. > Peptides...
A Nanoparticle Therapy to Treat Lung Cancer and Associated Muscle Wasting at the Same Time
Researchers at Oregon State University have engineered lipid nanoparticles that carry follistatin messenger RNA to lung tumors, simultaneously attacking the cancer and the muscle‑wasting cachexia that often accompanies it. In mouse models the nanocarriers bind circulating vitronectin, home to integrin‑rich...

Cell 'Snowball' May Be Answer to Large-Scale Tissue Engineering
Researchers at Penn State have created bio‑hybrid cell spheroids that self‑assemble like a snowball, rapidly increasing in size while preserving oxygen and nutrient flow. By embedding living cells in microgel particles, the new spheroids overcome diffusion barriers that traditionally limit...

Treating Previously Untreatable Cancers: How CAR-T Cell Therapy Could Be Made Accessible to More Patients
CAR‑T cell therapy, a personalized immunotherapy that re‑programs a patient’s T cells, has transformed treatment for certain leukemias and lymphomas but remains prohibitively expensive in Canada, with commercial products costing roughly $325‑$466 k USD per patient and requiring 4‑6 weeks for manufacturing....

Immunotherapy Enhanced by Restoring Mitochondrial Function in Dendritic Cells
A new study in Science by St. Jude researchers reveals that tumors suppress dendritic cell function by crippling mitochondrial fitness, undermining the body’s antitumor immunity. Restoring mitochondrial activity in dendritic cells reactivates their ability to prime immune responses and dramatically...

DIG-RHD: Digoxin Effective in Rheumatic Heart Disease
The DIG‑RHD trial, presented at ACC 2026, randomized 1,759 patients with symptomatic rheumatic heart disease in India to digoxin or placebo. Over a median 2.1‑year follow‑up, digoxin achieved a 4.1‑percentage‑point absolute reduction in the composite of all‑cause death or new‑onset/worsening...

Mayo Clinic's Dual-Drug Nanotherapy Doubles Survival in Glioblastoma Models
Mayo Clinic scientists have created a lipid‑based nanocarrier that transports everolimus and vinorelbine across the blood‑brain barrier, extending survival more than twofold in patient‑derived glioblastoma models. The preclinical breakthrough could reshape treatment strategies for the deadliest brain cancer.

IPLEDGE Risk Evaluation and Mitigation Strategy (REMS)
The FDA approved a set of modifications to the iPLEDGE Risk Evaluation and Mitigation Strategy on February 9, 2026, aimed at easing administrative burdens while preserving safety for isotretinoin users. Key changes include allowing home pregnancy tests during and after...

Opioid Analgesic Risk Evaluation and Mitigation Strategy (REMS)
The FDA’s Opioid Analgesic Risk Evaluation and Mitigation Strategy (REMS) now requires manufacturers to supply prepaid mail‑back envelopes (MBEs) for safe opioid disposal, with pharmacies able to order them starting March 31 2025. The REMS education program, funded by unrestricted grants, offers...

Low-Cost, Single Sample Blood Test Detects Different Cancers, Liver Disorders, and Other Diseases
UCLA researchers unveiled MethylScan, a low‑cost blood test that analyzes cell‑free DNA methylation to detect multiple cancers and liver disorders in a single sample. By using methylation‑sensitive enzymes to strip away background DNA, the assay reduces sequencing needs to about...

Data Platform Unifies Blood Cancer 'Omics' And Clinical Data to Accelerate Discovery
Scientists from St. Jude Children’s Research Hospital, the American Society for Hematology and the Munich Leukemia Laboratory launched the ASH HematOmics (ASHOP) platform, uniting genomics, transcriptomics and clinical data from 5,960 blood‑cancer patients. The open resource combines whole‑genome and whole‑transcriptome...

Detecting Multiple Cancers and Other Diseases From a Single Blood Sample
UCLA researchers introduced MethylScan, a blood test that reads cell‑free DNA methylation to flag multiple cancers and liver diseases in a single assay. In a cohort of 1,061 participants the test achieved 98% specificity, detecting about 63% of cancers overall...

Pharmaceutical Executive Daily: Neurocrine Biosciences Agrees to Acquire Soleno Therapeutics
Neurocrine Biosciences announced a $2.9 billion cash acquisition of Soleno Therapeutics, paying $53 per share—a 34% premium to Soleno’s closing price and 51% premium to its 30‑day VWAP. The deal brings Vykat XR, the only FDA‑approved therapy for hyperphagia in Prader‑Willi syndrome,...

Under-the-Skin Tepezza Comparable to Infused Version in Key Study, Amgen Says
Amgen announced that its subcutaneous on‑body injector version of Tepezza, called Tepezza OBI, met both primary and key secondary endpoints in a late‑stage trial, showing 77% of patients achieved a meaningful reduction in eye bulging. The efficacy was comparable to...
VDyne Secures FDA Nod to Start Pivotal Trial for Tricuspid Valve
VDyne received FDA approval for an investigational device exemption to launch its pivotal TRIVITA trial of a transcatheter tricuspid valve replacement system. The study will assess safety and efficacy in patients with severe symptomatic tricuspid regurgitation, a condition affecting roughly...

FDA Public Meeting: FDA-Led Patient-Focused Drug Development Meeting for Nonhealing Chronic Wounds - 08/25/2026
On August 25, 2026 the FDA will host a hybrid Patient‑Focused Drug Development meeting dedicated to non‑healing chronic wounds. The public session runs from 10 a.m. to 4:30 p.m. ET and combines a virtual webcast with an in‑person gathering at the White Oak Campus....

Proposed FDA Budget Sets Makary up to Boost US Biotech
The White House’s FY budget proposes a sizable increase for the FDA, positioning Dr. Robert Makary to spearhead regulatory reforms aimed at accelerating U.S. biotech development. The plan includes policy changes that would shorten clinical‑trial timelines, lower fees for early‑stage...

Immune Training Against Senescent Cells Reduces Tumor Growth
Here's my take on your paper: Cellular senescence or "zombie cell" events happen when a cell experiences too much genetic or epigenetic noise, caused by cellular damage (e.g. DNA breaks) or telomere erosion Your study of a Lewis Lung Carcinoma (LLC) mouse...

Academic Clinical Trials for Rapamycin to Answer Questions on Dosing for Anti-Aging Use
Researchers at UT Health San Antonio have launched a multi‑phase academic clinical trial to evaluate rapamycin’s biological effects in older adults. The program begins with a younger‑cohort benchmark study, then seeks the optimal dose that restores immune and metabolic markers...

Expanded MAGIC Toolkit Makes Genome-Wide Single-Cell Mosaic Analysis Possible in Drosophila
Cornell researchers have expanded the MAGIC (Mosaic Analysis by gRNA‑Induced Crossing‑over) toolkit to provide genome‑wide coverage across all Drosophila chromosomes, including the historically recalcitrant fourth chromosome. The new kit accelerates single‑cell mutant generation, cutting analysis time from months to weeks,...

Can Medicinal Cannabis Help Kids’ Autism, ADHD or Tourette’s? Here’s What We Know so Far
Interest in medicinal cannabis for children with autism, ADHD and Tourette's has grown, prompting the Australian Therapeutic Goods Administration to confirm that doctors are prescribing it for these conditions. While parents hope it could reduce reliance on stimulants, antidepressants and...

Global Cohort Data Bolster Confidence in Dolutegravir for Pediatric HIV Care
A new analysis presented at CROI 2026 used International Epidemiology Databases to Evaluate AIDS (IeDEA) data from Africa, Asia‑Pacific, and Latin America, covering over 90% of the world’s children with HIV. The study found that dolutegravir initiates modest early weight gain...

Tozorakimab Scores Double Win in Phase III COPD Trials
AstraZeneca announced that its IL‑33 monoclonal antibody tozorakimab achieved its primary endpoints in two Phase III COPD trials, Oberon and Titania. The drug significantly reduced the annual rate of moderate‑to‑severe exacerbations versus placebo across former and current smokers. AstraZeneca positions tozorakimab...
New Technique Identifies Proteins that Trigger Immune Responses in Transplants and Implants
Mayo Clinic researchers introduced a new method called the Ratio of Immunogenicity (ROI) to identify proteins that provoke strong immune responses. By measuring protein abundance and immune activation, the ROI ranks proteins from most to least immunogenic, revealing that mitochondrial...

Holland Foundation Looks to Expand Vision-Saving Transplants
The Holland Foundation for Sight Restoration is scaling its Cincinnati Protocol for ocular surface stem‑cell transplantation by establishing “centers of excellence” and expanding surgeon education. To date, five centers—from Cincinnati to UC Irvine, Virginia Eye Consultants, and Massachusetts Eye and...

Diabetes Drug Replicates Exercise Effects in Prostate Cancer Patients
How a Diabetes Drug May Echo the Benefits of Exercise in Prostate Cancer Care “From a clinical standpoint, seeing a metabolic signal that mirrors what we associate with intense exercise was striking… For patients whose treatments or symptoms limit physical activity,...

Biotech M&A Poised for Second‑Busiest Year, Early Deals Surge
Biotech M&A on track to have 2nd most active year in history w/ more early stage deals. Nice summary of which funds have benefited from recent M&A from Stifel @TimOpler $XBI $IBB https://t.co/udCuhIcaIi

FDA Drug Info Rounds Video
The FDA’s Center for Drug Evaluation and Research has launched the Drug Info Rounds video series, a free on‑demand library that educates health‑care professionals on drug safety, regulatory processes, and compliance. The series covers topics such as MedWatch reporting, Medication...

One Psychedelic Dose Quickly Eases Depression, Trial Shows
Single dose of a psychedelic drug can rapidly reduce depressive symptoms, clinical trial suggests https://t.co/c72qpfwQPm https://t.co/xR7hbj4v8e

Repurposing Approved Drugs: Small Trials Explore New Uses
Great reporting from @Philanthropy on our grantee @CuresWReach, which runs small trials testing whether existing, approved drugs might work for entirely different diseases.

Catalyst MedTech Establishes Full Access Neurology Solution for Brain PET Implementation in the U.S.
Catalyst MedTech announced the nationwide rollout of its Full Access Neurology solution, a bundled offering that enables health systems to deploy dedicated brain PET imaging without large capital outlays. The platform combines CareMiBrain‑powered scanners, quantification software, service and maintenance, and...

AI Cancer Vaccines Thrive While Cities Reject Data Centers
incredible seeing (actual, live) AI-designed cancer vaccines one minute, and local city councils unanimously voting down data centers the next. cognitive dissonance.

Egg Bioreactors Replace CHO Cells in Biologics Production
We’ve optimized drug design with AI. Why are we still manufacturing with CHO like it’s the 1980s? Neion Bio is rethinking biologics production using chicken eggs as bioreactors. Excited to hear Sam Levin at #SynBioBeta2026. Visit the SynBioBeta website to read the full...
Pfizer's Late-Stage Lyme Vaccine Shows 73% Efficacy but Misses Primary Endpoint
Pfizer announced a late‑stage Lyme disease vaccine candidate that achieved 73.2% efficacy 28 days after the fourth dose, but the study missed its primary endpoint because of lower-than-expected case numbers. The vaccine, a monoclonal antibody developed with MassBiologics, will now...

Autonomous Labs Revolutionize Research: Insights From Ginkgo, OpenAI, PNNLab
awesome talk about Autonomous labs from @Ginkgo, @OpenAI, and @PNNLab ! Check it out 👇
Study Shows Subcutaneous Tepezza Matches Infused Version
Under-the-skin Tepezza comparable to infused version in key study, Amgen says https://t.co/rOQqbRmEpL $AMGN $VRDN - 25%
CRISPR Therapeutics' One‑Time Gene Edit Cuts LDL by 49% in Early Trial
CRISPR Therapeutics reported that a one‑time CRISPR‑based therapy turned off the liver gene ANGPTL3, dropping LDL cholesterol by 49% and triglycerides by 55% in the highest‑dose cohort of a 15‑patient trial. The results, published in the New England Journal of...

Resveratrol Alters Sperm Epigenetics, Boosts Offspring Metabolism
CRAZY IF TRUE PAPER: A new study in mice reports that resveratrol intake by old male mice (or directly treating their sperm) changes the metabolism of embryos & pups, "potentially through alterations in sperm telomere length and epigenetic modifications"...

Meta-Analyses Reveal Psychedelics Boost Brain Circuits
Two systematic, meta-analyses on psychedelic drugs today Brain Circuit Function @NatureMedicine https://t.co/nBmjreYEXU Depression @NatMentHealth https://t.co/zJR06AsC0Z https://t.co/qpWdjzBR4D
Neurocrine to Acquire Soleno Therapeutics for $2.9 Billion in Cash
Neurocrine Pharmaceuticals has signed a definitive agreement to buy Soleno Therapeutics for $2.9 billion in cash, paying $53 per share—a 34% premium to Soleno’s April 2 close. The deal brings VYKAT XR, the only FDA‑approved drug for hyperphagia in Prader‑Willi syndrome, into Neurocrine’s...

Cell Senescence Drives Cancer Risk, Yet Can Be Modulated
Cell senescence and how it predisposes to cancer, no less how we may be able to modulate it. An outstanding @CellCellPress review https://t.co/bmAXOS1Ybz

BIO’s Comments for USTR Report Highlight Global Threats to Intellectual Property
The Biotechnology Innovation Organization (BIO) submitted comments to the USTR’s 2026 Special 301 Report urging stronger global enforcement of intellectual‑property (IP) rights for biotech. BIO argues that weak IP protections in markets such as Colombia, Russia and Brazil create barriers...

Other News to Note for April 6, 2026
Jiangsu and Shanghai Hengrui have patented selective Nav1.8 sodium‑channel blockers that show pre‑clinical analgesic efficacy with fewer side effects. New hematopoietic stem‑cell research links chronic inflammation to early leukemic transformation, identifying inflammatory pathways as therapeutic targets. Infinimmune presented pre‑clinical data...

Regulatory Actions for April 6, 2026
BioWorld’s April 6, 2026 regulatory snapshot aggregates the day’s key FDA and global health authority actions across biopharma, medical technology, and diagnostics. The page links to data snapshots, special reports, infographics and trend analyses covering topics such as mRNA vaccine research, GLP‑1...
Amgen Seeks to Buoy Tepezza with Injectable Data in Face of Incoming Competition
Amgen announced that its injectable formulation of Tepezza achieved its primary endpoint in a Phase 3 trial for thyroid eye disease (TED). The data suggest the drug can be administered subcutaneously, offering a more convenient alternative to the current intravenous...
Machine Learning and Single-Cell Technology Combined to Drive High-Performance Cell Line Development
OneCyte and Kemp Proteins have formed a strategic partnership that fuses OneCyte’s high‑throughput single‑cell cloning platform with Kemp’s machine‑learning‑driven protein design system, PROTiQ. The combined workflow uses in‑silico sequence evaluation to flag developability risks, then rapidly screens thousands of clones...