Phase 3 Data Highlight Hair Regrowth With Deuruxolitinib in AA
Phase 3 THRIVE‑AA2 data demonstrate that oral deuruxolitinib, a selective JAK1/2 inhibitor, significantly improves scalp hair regrowth in adults with severe alopecia areata. At 24 weeks, 33.0% of patients on 8 mg and 38.3% on 12 mg achieved a SALT20 response versus just 0.8% on placebo. Patient‑reported satisfaction rose to roughly half of treated participants, while safety remained manageable with mostly mild adverse events. The trial enrolled 517 participants across the US, Canada and Europe, supporting deuruxolitinib as a new therapeutic option.

Reprogram Biosciences Secures Financing for Solid Tumor mRNA Work
Reprogram Biosciences announced a $45 million Series B financing round to accelerate its mRNA platform targeting solid tumors. The capital infusion comes from a mix of strategic pharma partners and venture investors, underscoring confidence in the company’s antigen‑discovery pipeline. Reprogram plans to...

Statistical Approaches to Establishing Bioequivalence
The FDA has released a final guidance titled “Statistical Approaches to Establishing Bioequivalence,” superseding the 2001 version and formalizing the December 2022 draft. The document outlines recommended statistical methods for bioequivalence (BE) assessments across INDs, NDAs, ANDAs, and related amendments. It...

Ironwood Pharmaceuticals Reports the US FDA Approval of Linzess to Treat Pediatric Patients (≥2yrs.) With Functional Constipation
U.S. FDA has approved an expanded label for Ironwood Pharmaceuticals’ Linzess (linaclotide) for children aged two years and older with functional constipation. The decision follows a 12‑week Phase III trial in 2‑ to 5‑year‑old patients, where the 72 µg dose significantly increased...
3D-Printed Lymph Nodes Could Widen Access to CAR T-Cell Therapy
Researchers have shown that 3D‑printed lymph‑node scaffolds can grow CAR‑T cells more quickly and at a lower cost. The bioprinting method compresses the manufacturing timeline from several weeks to just a few days, potentially cutting expenses by up to 70...

Perceptic Raises £9m to Boost AI Drug Discovery
Perceptic announced a $12 million seed round, led by Accel, Air Street Capital and Elder Gull, to develop a unified AI platform for the entire drug lifecycle. The system aims to integrate asset scouting, scientific evaluation and clinical data into a...

FDA Approves Ironwood's LINZESS for Constipation in Kids 2‑5
Ironwood Pharmaceuticals announced that the U.S. Food and Drug Administration has approved LINZESS for functional constipation in children aged 2 to 5 years. The decision adds a new pediatric indication to a drug already used by more than 5.5 million patients...

How ASGCT and OTXL Are Working to Revive Shelved Cell and Gene Therapies
The American Society of Gene & Cell Therapy (ASGCT) and Orphan Therapeutics Accelerator (OTXL) have launched CGTxchange, an AI‑driven matchmaking platform designed to revive cell and gene therapies that were shelved for ultra‑rare diseases. By aggregating confidential and public data...

Basilea Secures $13.3 Million BARDA Grant to Push Novel Urinary‑Tract Antibiotic
Basilea Pharmaceutica Ltd. announced that the U.S. Biomedical Advanced Research and Development Authority (BARDA) awarded the company a $13.3 million grant to advance its novel antibiotic ceftibuten‑ledaborbactam etzadroxil. The funding lifts BARDA’s total commitment to $25 million and opens the door to...

Wedbush Boosts Apogee Therapeutics to Outperform, Sets $135 Target Amid $1.3 B Blackstone Deal
Wedbush reaffirmed an Outperform rating on Apogee Therapeutics and lifted its twelve‑month price target to $135, up from $120. The upgrade comes as Apogee announced a financing package with Blackstone Life Sciences that could deliver up to $1.3 billion in non‑dilutive...

The Evolving Biopharma Regulatory Landscape: Q&A with Harpreet Singh, MD
Harpreet Singh, former FDA oncology division director and now chief medical officer at Precision for Medicine, explains how the FDA is reshaping biopharma regulation through a shift to single pivotal trials for high‑risk cancers, the expanding but opaque National Priority...
PRINCE: A Small-Molecule Switch for Safer Gene Editing
Researchers unveiled PRINCE, a CRISPR‑Cas system whose nuclease and guide RNA are each activated by separate FDA‑approved small‑molecule drugs, delivering precise temporal control. The platform stayed functional for up to two years after genomic integration in human cells, and a...

AbbVie Acquires Imgen’s ADC After Jazz Passes
Remember Decnupaz (pivekimab sunirine, then coded IMGN632) was one of two Immunogen ADCs $JAZZ decided not to opt into c2020. $ABBV later bought $IMGN for Elahere.

In 2018, a Chinese Biophysicist Announced He Had Gene-Edited Twin Girls Using CRISPR. The Scientific Consensus Is that What He...
In November 2018 Chinese biophysicist He Jiankui announced that he used CRISPR‑Cas9 to edit embryos, resulting in the birth of twin girls, Lulu and Nana. Subsequent analysis showed the edits were novel CCR5 deletions, highly mosaic, and accompanied by off‑target...
Researchers Block Key Protein that Helps Parkinson’s Spread Through the Brain
Researchers at the University of Pennsylvania have pinpointed the brain immune protein GPNMB as a catalyst for the spread of alpha‑synuclein in Parkinson’s disease. In pre‑clinical experiments, monoclonal antibodies that block GPNMB prevented the protein’s propagation between neurons. Analysis of...
New Drug ‘Functionally Cures’ Many Hepatitis B Virus Infections
GSK’s antisense drug bepirovirsen (bepi) added to standard antivirals produced a functional cure in 19% of chronic hepatitis B patients in two phase 3 trials, rising to 26% among those with the lowest surface‑antigen levels. The cure, defined as undetectable HBV DNA...

Colossal Bio Grows Chicks in 3D-Printed Artificial Eggs. Breakthrough or Copycat Technology?
Colossal Biosciences unveiled a 3D‑printed, silicone‑lined “artificial egg” that allowed baby chicks to develop inside transparent plastic cups at its Dallas lab. The company touts the invention as a step toward resurrecting extinct birds such as the dodo and giant...
Stanford Researchers Block Aging Enzyme to Regrow Knee Cartilage, Launch Oral Trials
Stanford University scientists led by Helen Blau and Nidhi Bhutani have demonstrated that an injection blocking the aging enzyme 15-PGDH can regenerate knee cartilage in aged mice and human tissue samples. The breakthrough, which also boosted muscle mass and endurance,...

IRC Warns Ebola Outbreak Could Become Deadliest on Record as Cases Top 900
The International Rescue Committee warned Tuesday that the Ebola outbreak in the Democratic Republic of Congo and Uganda could become the deadliest on record, citing more than 900 suspected cases and 223 deaths as the virus spreads to major cities....
Convergent Mitochondrial Impairment and Apoptosis Driven by Simultaneous Down-Regulation of Multiple Genes at 11p11.2 in Alzheimer’s Disease
Researchers identified that simultaneous down‑regulation of several genes within the 11p11.2 risk locus occurs in Alzheimer’s disease brains. Integrated transcriptomic and functional analyses revealed that this coordinated gene suppression disrupts mitochondrial complex I, elevates reactive oxygen species, and activates caspase‑7–mediated...

Gene Therapies to Fix Failing Hearts Gain Steam After Years in the Doldrums
Gene‑therapy researchers have launched the first human trial aimed at regenerating heart muscle by silencing the SAV1 gene, a brake on cardiomyocyte division. Pre‑clinical work in pigs showed a 14% boost in ejection fraction, prompting U.S. regulators to green‑light the...
Harvard Scientists Win Breakthrough Prize for Gene Therapy Triumphs
Five Harvard-affiliated scientists received the 2026 Breakthrough Prize in Life Sciences for pioneering discoveries in gene therapy, sickle cell disease, ALS, dementia, and inherited blindness treatments. Their work helped advance groundbreaking therapies including the first FDA-approved treatment for a genetic disease,...
Post-IPO, Kailera Looks Beyond Obesity to MASH
Kailera Therapeutics completed the largest biotech IPO on NASDAQ, raising $718.8 million after the greenshoe. In its first post‑IPO update, the company disclosed Phase I data for KAI‑4729, a triple GLP‑1R/GIPR/GCGR agonist developed with Jiangsu Hengrui. The early trial in healthy volunteers...
Science Spotlight: Silencing Seizures by Fixing Gene Mutations
Two independent research teams reported in Science Translational Medicine that in‑vivo gene editing can repair disease‑causing SCN1A mutations in mouse models of severe epilepsy. The University of Zurich used AAV‑delivered prime editing to correct the K1270T Nav1.1 mutation in neonatal...

DNA 'Nicks' Make for Safer, More Precise Genetic Analysis
Cornell researchers have upgraded the CRISPR‑based MAGIC technique by swapping double‑strand cuts for single‑strand DNA nicks. Using Cas9‑derived nickases, they demonstrated that a lone nick can still drive mitotic recombination in fruit‑fly tissues, dramatically lowering cellular toxicity. The study, published...

Gilbane Breaks Ground on $450M Philadelphia Cancer Lab
Gilbane Building Co. broke ground on TerraPower Isotopes' Bellwether Laboratory in Philadelphia, a 250,000‑square‑foot, $450 million facility dedicated to producing radioactive molecules for cancer treatment. The project, backed by Bill Gates‑owned TerraPower, will eventually employ more than 225 workers. Gilbane will...

A New Generation of Drugs Could Lower Heart Attack Risk for Millions
The episode spotlights a new class of genetic‑silencing drugs targeting elevated lipoprotein(a) (Lp(a)), a hereditary risk factor for heart attacks and strokes, with late‑stage trials from Novartis, Amgen and Eli Lilly poised to reveal efficacy data. Reporters explain how these therapies...

Sidewinder DNA Synthesis Cuts Errors to 1 in 10 Million, Accelerates AI‑Designed Genomes
Scientists have unveiled Sidewinder, a DNA‑synthesis technique that can assemble dozens of sequences in a single tube with an error rate of one mistake per 10 million joins. The method uses inexpensive raw materials and promises to make AI‑designed genomes affordable...

Outlook Therapeutics Secures FDA Appeal Win for nAMD Therapy, Stock Jumps
Outlook Therapeutics Inc. (OTLK) announced that the FDA granted its appeal for the ONS-5010/LYTENAVA neovascular age‑related macular degeneration therapy, prompting a rise in its share price. The decision follows positive data from the pivotal NORSE TWO trial and confirmatory evidence...
BioRestorative Completes Phase 2 Dosing of BRTX-100, Shares Jump 20%
BioRestorative Therapies Inc. finished dosing its Phase 2 trial of BRTX‑100 for chronic lumbar disc disease, prompting a 20% surge in its stock. The autologous stem‑cell therapy, already granted FDA Fast Track status, was administered to 99 patients over a 52‑week...

Merck and Mastercard Are Seeing Real Agentic AI Results. Both Say the Plumbing Came First.
Merck is leveraging AI agents to accelerate drug discovery and marketing, cutting research cycles by a third and delivering compliant marketing drafts up to 80% faster. The gains stem from a "plumbing‑first" strategy that now supports 2,500 AWS accounts, multiple...
FDA Accepts BridgeBio’s Application for Potential First Limb-Girdle Muscular Dystrophy Drug
BridgeBio’s oral therapy BBP‑418 has received FDA priority review, with a target action date of Nov. 27, 2025, positioning it for a potential launch in late 2026 or early 2027. The Phase 3 FORTIFY trial met all primary and secondary endpoints, showing...

Neurovalens Secures FDA De Novo Clearance for Modius Spero PTSD Device
Neurovalens announced FDA de novo clearance for its Modius Spero platform, a non‑invasive neuromodulation device targeting post‑traumatic stress disorder. The approval paves the way for prescription use among U.S. veterans starting July 2026, expanding the company’s portfolio of bioelectronic therapies.
Dyne Therapeutics Files BLA for Z‑Rostudirsen, Seeks Priority Review for DMD Treatment
Dyne Therapeutics announced the filing of a Biologics License Application with the FDA for Z‑rostudirsen, an exon 51‑skipping therapy for Duchenne muscular dystrophy, and has asked for priority review. The move follows a Phase 1/2 DELIVER trial that met its...
Hansa Biopharma’s Idefirix Hits 90% Graft Survival in EU Post‑Authorization Study
Hansa Biopharma announced that its gene‑therapy desensitization product Idefirix met the primary endpoint of a European post‑authorization study, delivering 90% one‑year graft failure‑free survival in highly sensitized kidney‑transplant patients. The results also showed 92% graft survival and 98% patient survival,...
Embryos Made without Sperm or Eggs Reveal Why Many Pregnancies Fail
Scientists in Vienna have created embryo organoids, called blastoids, entirely from stem cells without sperm or eggs. These models replicate the structure and early gene activity of a natural blastocyst, allowing researchers to observe implantation and other first‑week events in...
Antiviral Valganciclovir Extends GBM Survival to 5 Years
🧠 GBM patients on continuous valganciclovir survived 56.4 months — nearly 5 years — vs. the typical 12–15. Years ago, Thomas Seyfried told me that CMV damages mitochondria and drives tumor initiation and growth. This NEJM study validated that insight in a...
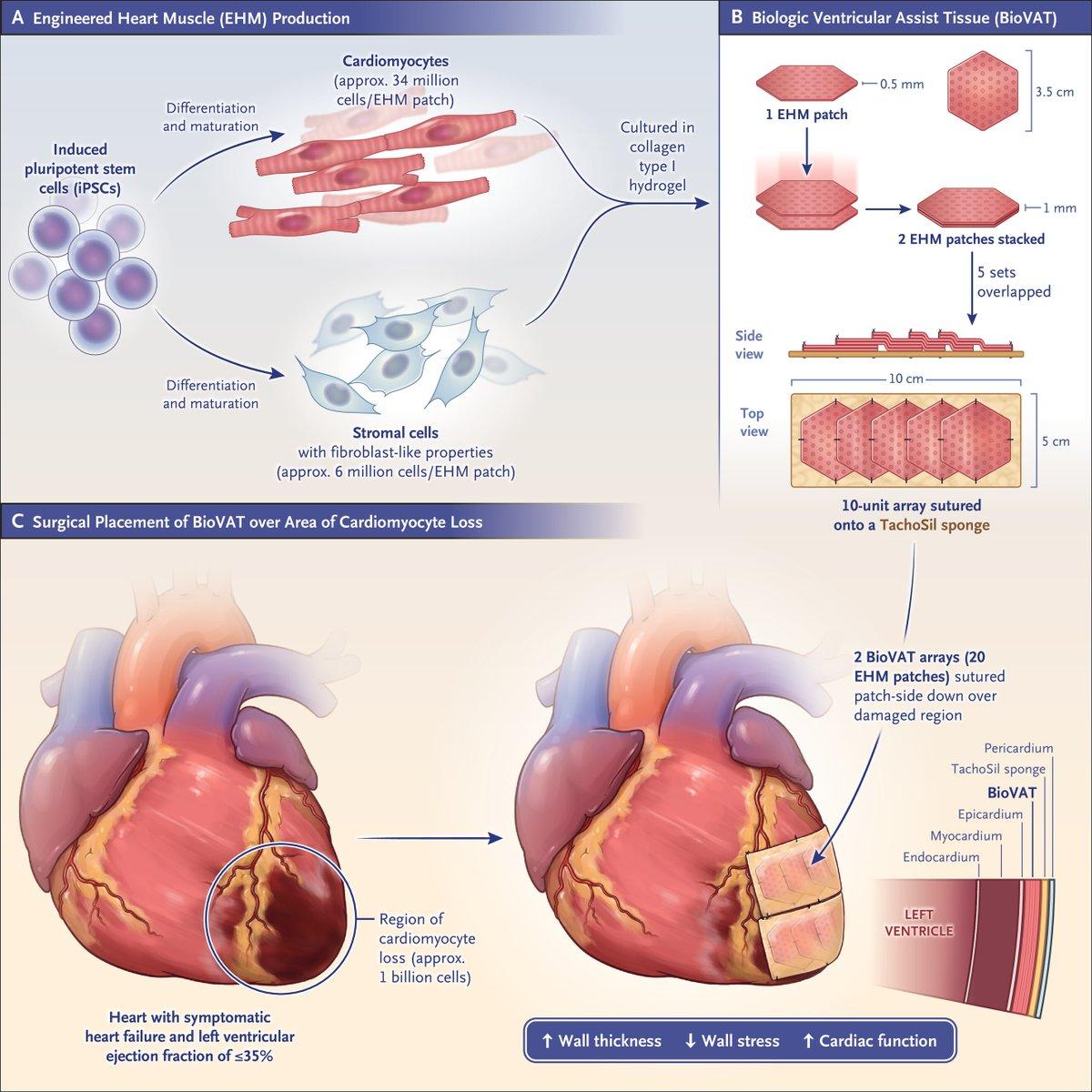
Stem‑cell Heart Patches Improve Outcomes in 12 of 20 Patients
Cardiac "remuscularization" for treating severe heart failure with patched heart muscle derived from stem cells (a biological ventricular assist device) successful in 12 of 20 patients @NEJM https://t.co/mAwgj2rAwE https://t.co/3ogxaJFDE0 https://t.co/J1ozMsS4Oh

Jade Biosciences Initiates First-in-Human Phase 1 Trial of BAFF-R Antibody JADE201
Jade Biosciences has dosed the first participant in a first‑in‑human Phase 1 trial of JADE201, an investigational half‑life‑extended afucosylated anti‑BAFF‑R monoclonal antibody. The randomized, placebo‑controlled study evaluates single ascending doses in rheumatoid arthritis patients, focusing on safety, tolerability, pharmacokinetics and pharmacodynamics....

New Stem Cell Subtype Remembers Inflammation, Impacts Prognosis
New @nature. Discovery of a new subtype of blood stem cells—HSC-iM—which have memory to previous inflammation, such as with Covid or aging, and prognostic significance https://t.co/hRfp5xnJGj

ClearNote Health Secures $52 Million in Series D Financing and Strengthens Leadership Team to Accelerate Growth in Early Cancer Detection
ClearNote Health announced a $52 million Series D financing round, bringing its total capital to over $185 million. The company also appointed Kevin Keegan, a veteran of Illumina, BD and Hologic, as President and COO. Proceeds will fund commercial expansion, clinical studies, and further...

Junevity to Present Breakthrough Research on siRNA Therapeutics at American Aging Association’s 2026 Annual Meeting
Junevity will present pioneering in‑vivo data showing an siRNA therapeutic can restore global gene networks to a healthier state. Co‑founder Dr. Janine Sengstack will share results from diabetic mouse studies and safety data in rats and non‑human primates, marking the...

STAT+: Kailera’s Own ‘Triple-G’ Drug Also Looks Very Powerful
The FDA postponed its decision on AstraZeneca’s experimental breast‑cancer therapy camizestrant after advisers criticized the SERENA‑6 trial design, giving the company extra time for additional analyses. Meanwhile, Blackstone Life Sciences pledged up to $1.3 billion to Apogee Therapeutics to fund Phase 3...

Chinese Scientists Use Supercomputer to Cut New Drug Screening Time From Years to Seconds
Chinese researchers have launched GalaxyVS, an AI‑driven drug‑discovery platform that leverages the Tianhe supercomputer to screen up to 100 billion chemical compounds in seconds. The system achieves a daily throughput of 16 trillion molecular dockings, a million‑fold speed increase over the previous...

Olezarsen Cuts Pancreatitis Events in Severe Hypertriglyceridemia Analysis
Swedish Orphan Biovitrum (Sobi) presented a pooled analysis of its phase 3 CORE and CORE2 trials showing that the RNA‑targeted drug olezarsen reduced acute pancreatitis events by 85% and lowered triglycerides up to 66% in patients with severe hypertriglyceridemia (baseline ≥880 mg/dL)....

Zuckerberg, Chan's Biohub Launches Protein 'World Model'
The Chan Zuckerberg Biohub has unveiled an AI‑driven "world model" for protein biology, built on an atlas of 6.8 billion proteins and 1.1 billion structures. The open‑source platform combines the ESMC language model with the ESMFold2 design engine to predict structures, design...

Roy Maute on Macrophage Checkpoint Inhibition: Pheast’s Vision for Advancing PHST001 in Solid Tumors
Pheast Therapeutics presented early Phase 1a data on PHST001, an anti‑CD24 macrophage checkpoint inhibitor, at AACR 2026. The drug demonstrated favorable tolerability, dose‑linear pharmacokinetics, robust CD24 receptor occupancy and signs of immune activation. Preliminary anti‑tumor activity was seen across heavily pretreated...

Father Celebrates NHS Approval of Duchenne Drug Givinostat for Son
Alex, whose 12‑year‑old son Ben has Duchenne muscular dystrophy, said he is "incredibly happy" after the National Institute for Health and Care Excellence approved Givinostat for nationwide NHS use. The approval ends a 14‑month campaign to secure the drug beyond...

FDA Extends Review of AstraZeneca’s Camizestrant NDA for ESR1‑Mutated Advanced Breast Cancer
The U.S. Food and Drug Administration has extended the PDUFA review date for AstraZeneca’s New Drug Application for camizestrant, an oral selective estrogen receptor degrader combined with a CDK4/6 inhibitor, after requesting more data to support its use in hormone‑receptor‑positive,...

Eli Lilly to Acquire Curevo for up to $1.5 Billion, Boosting Shingles Vaccine Portfolio
Eli Lilly announced a definitive agreement to acquire Curevo Vaccine for up to $1.5 billion in cash. The deal gives Lilly access to Amezosvatein, a Phase‑3‑ready shingles vaccine that matched the standard of care in Phase 2 while halving side‑effects. The transaction...