Know What's Happening in Healthcare
Know What's Happening in Healthcare

FDA approves KRESLADI, first CIRM‑backed gene‑editing therapy
The FDA cleared KRESLADI, a gene‑editing treatment from Rocket Pharmaceuticals for severe leukocyte adhesion deficiency‑I in children, eliminating the need for a bone‑marrow donor. It is the first product directly funded by California’s Institute for Regenerative Medicine to receive approval.
Also developing:
By the numbers: Syneron Bio raises $150M Series B
Gene therapy for sickle cell disease has moved from experimental promise to an actionable clinical reality, with early CRISPR‑based treatments already cutting crises and hospital stays. A recent Sanius Health survey of 94 patients shows strong interest—71% want more information—yet nearly half lack basic awareness and cite safety and cost as major concerns. The article argues that the next growth phase hinges on robust real‑world data, longitudinal monitoring, and transparent patient‑centric ecosystems. Industry leaders must pair scientific breakthroughs with new reimbursement models and infrastructure to sustain long‑term value.

An EMT recounts a harrowing transport of a stroke‑survivor, Molly, highlighting how standard stair‑chair equipment failed to accommodate her size and mobility limitations, leading to injury and indignity. The author uses the incident to illustrate structural violence and the marginalization...

Shine Technologies announced a $240 million financing round led by Patrick Soon‑Shiong’s NantWorks, bringing its total capital raised to over $1 billion. The infusion will accelerate the company’s commercial fusion initiatives and expand its Lu‑177 isotope production, a cornerstone of targeted cancer...

The UK Department of Health and Social Care (DHSC) has launched an industry‑readiness survey on Value Based Procurement (VBP), targeting MedTech and health‑technology firms. The ten‑minute questionnaire seeks a single consolidated response per company and must be submitted by 27 March....

The UK Health Data Research Service (HDRS) is set to launch later this year as a secure, single gateway for national health and care datasets, aiming to unlock the country’s extensive longitudinal records. The government announced Melanie Ivarsson, a former...

Researchers identified that the aging gut microbiome harbors increased Klebsiella aerogenes, which elevates histamine production and compromises intestinal barrier integrity. The excess histamine suppresses Nlrp6 expression, disrupting LC3‑mediated autophagy and intensifying inflammation in septic models. Experiments showed that lowering histamine...
Segmed, a leader in real‑world medical imaging, announced a partnership with Verily to make its de‑identified, diagnostic‑grade imaging datasets available on Verily’s AI‑native Pre Exchange and Workbench platforms. The initial offering is a longitudinal breast‑cancer cohort featuring digital breast tomosynthesis...

Real‑world data (RWD) is reshaping natural history studies by delivering longitudinal, patient‑level information that was previously only obtainable through costly prospective trials. Advances in artificial intelligence and natural language processing now allow rapid extraction of structured data from unstructured sources...
A new donor‑derived cell‑free DNA (dd‑cfDNA) blood test is transforming post‑transplant surveillance by providing real‑time, noninvasive insight into graft health. The assay quantifies donor DNA fragments released when a transplanted organ is injured, offering a high negative predictive value that...

The U.S. FDA granted accelerated approval to Ascendis’ Yuviwel (navepegritide) for increasing linear growth in children aged two years and older with achondroplasia and open epiphyses. Approval rests on three placebo‑controlled trials and up to three years of open‑label extension...

Employers often rely on isolated claims or EAP call counts, which dramatically understate the true scale of behavioral health issues in the workforce. By aggregating medical, pharmacy, and EAP data—and even website analytics—companies can capture both prevalence and cost, revealing...

An independent NHS evaluation across seven Suffolk care homes using Nobi Smart Lights reported up to a 75% reduction in falls‑related hospital admissions and up to a 65% drop in ambulance call‑outs. The study compared six months of baseline data...

Cell therapy manufacturing is vulnerable to contamination because living cells cannot be terminally sterilized. Regulatory bodies such as EU GMP Annex 1 and the FDA now require risk‑based contamination control strategies that prioritize closed, sterile, and automated processes. Closing open steps...

The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) issued a positive opinion recommending Dupixent (dupilumab) for paediatric chronic spontaneous urticaria (CSU) in children aged 2‑11. The recommendation is based on robust Phase III data from the LIBERTY‑CUPID...

Synergy Spine Solutions received FDA pre‑market approval for its Synergy Disc, a cervical artificial disc designed to preserve motion and correct spinal alignment. Clinical data showed an 87.1% composite success rate at 24 months, with significantly lower neck‑pain and arm‑pain...

Opus Genetics presented early Phase I/II data for its OPGx‑BEST1 gene therapy targeting best vitelliform macular dystrophy and autosomal‑recessive bestrophinopathy. In a sentinel 63‑year‑old participant, the treatment was well tolerated and delivered a 12‑letter gain in best‑corrected visual acuity after...

BioMarin’s enzyme substitution therapy Palynziq received FDA approval for use in adolescents aged 12 and older with phenylketonuria (PKU). The decision was based on Phase III PEGASUS trial results showing 44.4% of participants lowered blood phenylalanine below guideline levels, with a...

A Drexel‑led study demonstrated that low‑melt polyaryletherketone (LM PAEK) can be fused‑filament‑fabricated into porous knee‑revision cones that outperform traditional PEEK in shear strength and stiffness. Using a Taguchi L8 design, the researchers printed 64 cylindrical surrogates with gyroid and diamond TPMS...

Gifthealth’s co‑founder John Romano outlines how AI‑driven pricing tools and automated patient‑support platforms can lower out‑of‑pocket costs and improve medication adherence for the 60 million Americans living with gastrointestinal diseases. By integrating real‑time claims adjudication, discount automation and personalized education, the...

The article profiles 18 UK biotech firms spanning oncology, immunology, regenerative medicine and AI‑driven drug design, highlighting recent milestones such as Amphista’s $30 million upfront deal with BMS, Autolus’ FDA and EU approvals for obe‑cel, and Isomorphic Labs’ $600 million raise to...

Kardi Ai, a Czech med‑tech firm, has secured EU MDR Class IIa certification and is expanding its long‑term ECG monitoring platform into the DACH region. The solution records heart rhythm for up to 12 months, uses AI to deliver physician‑ready reports within...

Lewis A. Grossman's book "Choose Your Medicine" traced historic battles over alternative‑medicine freedom, a pattern echoed today as Health Secretary Robert F. Kennedy Jr. pushes to loosen FDA oversight of unapproved stem‑cell and peptide treatments. The interview highlights recurring public resistance...

Motion control underpins modern medical devices such as surgical robots, imaging systems, and laboratory instruments, yet it remains a complex engineering hurdle. Dave Beckstoffer of Portescap highlights how early, collaborative partnerships with motion‑control specialists can streamline development and boost reliability....

Researchers have developed a monolithic 3D nanoelectrode array (HD‑NEA) that integrates 26,400 vertical nanowire electrodes directly onto commercial CMOS chips. The low‑temperature wafer‑scale post‑fabrication process maintains circuit functionality while delivering uniform, high‑yield electrode performance across 4‑inch wafers. In vitro recordings...

The Patient‑Centered Health Technology Initiative (PHTI) released a Clinical AI report built from a workshop with senior leaders across health systems, insurers, tech firms, and federal agencies. Participants identified policy, reimbursement, and evidence gaps as primary barriers to scaling AI...

X4 Pharmaceuticals’ Xolremdi (mavorixafor) received a positive opinion from the European CHMP, recommending approval under exceptional circumstances with an EC decision expected in Q2 2026. The recommendation is based on the global Phase III 4WHIM trial involving 31 patients aged 12 and...

European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) issued a positive opinion recommending conditional approval of Ipsen’s Ojemda (tovorafenib) as monotherapy for pediatric low‑grade glioma with BRAF fusions, rearrangements or V600 mutations. The recommendation is based on...

North American insurers are confronting a surge in claim severity as medical inflation, social inflation, and AI‑generated fraud drive costs upward. The Gallagher Bassett 2026 Carrier Report shows 64% of carriers see more complex claims, with 56% pinpointing rising medical expenses...

Life‑science firms face heightened D&O liability as stock volatility spikes around clinical trial readouts and regulatory decisions. Shareholder lawsuits often begin with books‑and‑records demands that probe whether public statements matched internal knowledge. The article stresses disciplined, consistent disclosure and early...

Healthcare revenue cycle leaders are shifting from rule‑based automation to AI‑driven predictive analytics. Machine‑learning models now scan claim data, documentation and payer behavior to flag denial risks before submission. Early corrections improve first‑pass acceptance, shorten cash cycles and reduce revenue...

Otsuka Medical Devices and Otsuka Pharmaceutical announced that Japan’s National Health Insurance will cover the Paradise™ Ultrasound Renal Denervation (uRDN) system starting March 1, with commercial sales beginning March 2. The device, developed by Recor Medical, targets resistant‑hypertension patients who remain uncontrolled...

The episode examines the rapid rise of tattoos in the U.S., now covering roughly a third of adults, and details the health hazards associated with modern ink, including toxic heavy metals, chronic inflammation of lymph nodes, and an elevated risk...

Quanterix (QTRX) reported FY 2025 revenue of $16.1 million, down $11 million from the prior year, driven by steep declines in collaboration and contract‑manufacturing income. R&D spending fell modestly to $140.7 million, while SG&A rose to $65.5 million due to higher professional fees for...

Cerus Corp reported record total revenue of $233.8 million in 2025, a 16% increase year‑over‑year, driven by strong product sales and robust growth in EMEA. Product revenue rose 14% for both the quarter and full year, with IFC kit sales...

Researchers integrated inflammatory cytokines, kynurenine pathway balance, and amygdala reactivity with the IDEA‑RS sociodemographic risk score to predict adolescent depression over three years. The composite model (IDEA‑BIO‑RS) raised predictive accuracy from a 0.715 AUC to 0.889, correctly classifying 82.2% of...

Xeris Biopharma reported record Q3 2025 product revenue of $74.1 million, a 40% year‑over‑year increase driven primarily by Recorlev’s 109% revenue jump. The company posted its first quarterly net income and an adjusted EBITDA of $17.4 million, reflecting strong operating leverage and...

The researchers conducted a crowdsourced megastudy that tested 12 digital single‑session interventions (SSIs) for depression among a large U.S. adult sample recruited online. Using a preregistered design, three of the SSIs produced statistically significant reductions in depressive symptoms, with effect...

UniQure reported a regulatory setback as the FDA now deems the Phase 1/2 external‑control data for AMT‑130 insufficient for a Biologics License Application, creating uncertainty around the U.S. filing timeline. Despite this, the high‑dose AMT‑130 achieved a statistically significant 75% slowdown...

UroGen Pharma reported over $127 million in cash at the end of Q4 2025, affirming sufficient liquidity to reach profitability if current plans hold. The company highlighted steady month‑over‑month growth in Zasturi enrollment, with community treatment share now around 35‑40% and institutional...

Senseonics reported a 60% jump in full‑year revenue to $35.3 million and lifted gross margins above 50% after a year of selling the Eversense 365 implantable CGM. The company completed a full transition of commercial operations from Ascensia, eliminating revenue‑sharing and gaining...

Surgery Partners reported Q4 2025 net revenue of $821.5 million, a 6.6% year‑over‑year increase, and adjusted EBITDA of $136.4 million, up 6.1% with a 16.6% margin. The company revised full‑year revenue guidance to $3.275‑$3.30 billion and adjusted EBITDA to $535‑$540 million, citing delayed capital...

Japan’s healthcare system is rapidly expanding multilingual services as inbound tourism surges, with 2,500 hospitals and clinics now listed as foreign‑patient friendly—a 1.6‑fold rise over six years. The Hakuba International Clinic exemplifies the trend, handling 40‑50 walk‑ins daily, 80% of...

According to ITOA, NAD declines with age, causing epigenetic changes & disease susceptibility In 2010, we showed NAD-dependent enzyme, SIRT3, protects the heart 🐁 New study says NAD precursor NMN protects the heart via SIRT3 in mice fed a Western diet https://www.sciencedirect.com/science/article/pii/S0022316626000842
But also it’s not only measles, pertussis (whooping cough) is also returning, and based on latest ACIP (or what used to be ACIP) stunts, we’ll see rotavirus, meningococcal meningitis, HBV liver cancer, return after that. A public health disaster built...

Medical students Jay Pendyala and Jonathan Berg compare chess strategy to clinical reasoning, highlighting how assessing a patient mirrors evaluating a board position. They argue that diagnostic algorithms serve as openings, but flexibility is required when cases deviate from textbook...

The Department of Justice is appealing a ruling that blocked a subpoena targeting a queer‑owned clinic providing gender‑affirming care, arguing that the clinic’s patient‑education materials should be treated as drug labeling under the Federal Food, Drug, and Cosmetic Act. By...

Compliance failures are now costing MedTech manufacturers millions, as regulators require deeper traceability, faster reporting, and unified post‑market oversight. Gaps in ERP and QMS integration lead to delayed approvals, recalls, and costly litigation, turning compliance into a direct revenue driver....

Dr. Charan Teja Bobba recounts treating a patient with meth‑associated dental disease in a MassHealth safety‑net clinic. Methamphetamine caused extensive enamel loss, xerostomia, and rapid decay, demanding complex restorative procedures over several visits. By involving the patient in every step and building...
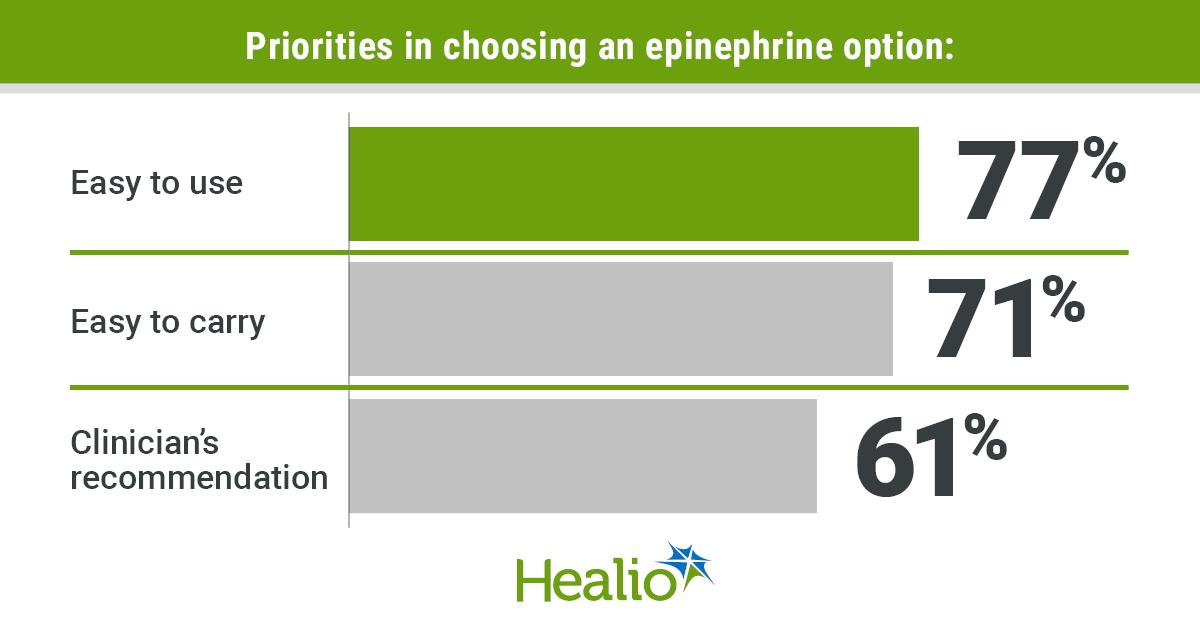
A cross‑sectional survey of 210 anaphylaxis patients and caregivers revealed that 90% prefer needle‑free epinephrine delivery, while 89% value devices that are small and easy to carry. Respondents also expressed strong concerns about temperature effects on epinephrine stability. The study...

Lymphangioleiomyomatosis (LAM), a rare estrogen‑sensitive lung disease, highlights the absence of standardized reproductive care for women with rare conditions. While FDA‑approved therapies have extended patients' lives, guidance on pregnancy, contraception, and menopause remains fragmented. The author’s personal journey—freezing embryos and...