![[Comment] Offline: President Trump—It Is Not Too Late](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/33edb6cd7db39a81ba27a2ec060c6b15.webp)
[Comment] Offline: President Trump—It Is Not Too Late
The Physicians for Human Rights report warns that recent U.S. cuts to global health funding under the Trump administration have crippled HIV/AIDS programs in South Africa, Kenya and other high‑burden countries. Primary‑care testing, PrEP distribution, antiretroviral therapy and research labs have been severely reduced or halted, leaving thousands without life‑saving services. The report argues that the funding gap threatens both public health outcomes and the return on decades of American investment in vaccine and drug development. It calls on Congress, multilateral agencies and non‑U.S. donors to restore support before the crisis deepens.

Data Security Is a Question of Trust
UK Biobank discovered that de‑identified health data from its 500,000‑volunteer cohort appeared on a Chinese Alibaba auction site, prompting a rapid joint response from the biobank, Alibaba and UK and Chinese authorities. The leak was traced to three accredited researchers...

If We Can Fund a $200 Billion War, Why Not Fund Food Security at Home?
The Pentagon is seeking over $200 billion to expand a war with Iran, a sum that could instead fund a generational push for food security in the United States. Poor diet contributes to the disqualification of most young Americans from military...

‘Searching For Drug Peace’ Trailer: Hot Docs Film Explores “Daring Reform Activist” Waging War On The War On Drugs
The documentary *Searching for Drug Peace* will debut at Hot Docs in Toronto on April 28, 2026, chronicling activist Dana Larsen’s battle against the North American War on Drugs. The film highlights the staggering overdose toll—nearly 80,000 deaths in the U.S. in...

While SF Sees Fewer Fatal Overdoses, Death Rate Is Still Among the Country’s Worst
San Francisco reported 49 accidental drug overdose deaths in March, bringing the year‑to‑date total to 148. While the monthly and YTD figures represent a decline from previous years, the city’s overdose death rate still ranks among the nation’s highest. Officials...

Utah Medical Board’s Call To Halt AI Drug Pilot Triggers Med Practice Showdown
Utah’s medical board has ordered an immediate pause on the state’s pioneering AI‑driven prescription renewal pilot, the first of its kind in the nation. The pilot, launched by a consortium of health systems and a tech vendor, used machine‑learning algorithms...

CMS Proposes Electronic Prior Authorization for Drugs
The Centers for Medicare & Medicaid Services (CMS) has issued a proposed rule to extend electronic prior authorization (e‑PA) to pharmaceuticals covered under both medical and pharmacy benefits. The rule mandates the use of Fast Healthcare Interoperability Resources (FHIR) APIs...

‘We’re Not Prepared to Replace Them’: Immigration Changes Hit Nursing Homes Hard, Undermining Workforce Stability
Recent U.S. immigration policy shifts—most notably a $100,000 fee for new H‑1B petitions and tighter temporary protected status rules—are threatening the nursing‑home labor pipeline. Immigrants already represent roughly 40% of staff at Georgia‑based A.G. Rhodes and 21‑25% nationwide, especially in...
Why Cardiac Monitoring Belongs at the Foundation of Your Service Line Strategy: A Conversation with Amanda Maples, RN, BSN, MHA
Amanda Maples, senior manager of customer experience at iRhythm, explains how continuous cardiac monitoring can serve as a foundational tool for cardiovascular service lines. She highlights the technology’s role in shortening diagnostic timelines, especially for women’s heart disease and frequent‑flyer arrhythmia...

Patent Analysis Is Increasingly Shaping AI-Driven Target and Drug Candidate Selection
A new review in Nature Reviews Drug Discovery outlines how AI is expanding target identification by merging multi‑omics, knowledge graphs, and foundation models. The authors argue that patentability, commercial tractability, and competitor analysis should be evaluated alongside druggability and safety...

Synergy CEO Eyes Top Spot In Home Care, Prioritizing Growth Without Franchise Overload
Synergy HomeCare, now operating in 626 territories across 44 states, plans to add roughly 100 new franchise locations in 2026, matching its 2025 expansion pace. CEO Charlie Young stresses that rapid growth must not overwhelm franchisees, so the company selectively...

Bioprocessing Method Is a Critical Factor for IgM Oligomerization
Researchers compared fed‑batch and semi‑continuous perfusion bioprocesses for recombinant IgM production in CHO DG44 and CHO K1 cell lines, generating either pentameric (5IgM) or hexameric (6IgM) antibodies with or without a J‑chain. The perfusion approach, which continuously supplies fresh medium...

Journey to Joy: Leveraging National Burnout Data — Executive Insights and Innovations From Leading Health Systems
Physician well‑being is emerging as a core quality metric, prompting health systems to replace generic engagement surveys with targeted burnout assessments. The American Medical Association highlighted its Organizational Biopsy® tool, now deployed by over 130 organizations, which tracks EHR burden,...

First Phase of VA EHR Rollout Successful, Deputy Secretary Says
The Department of Veterans Affairs completed the first wave of its electronic health records (EHR) modernization in Michigan, activating systems at Ann Arbor, Battle Creek, Saginaw and Detroit in April. Deputy Secretary Paul Lawrence said the rollout revives a program...

How The US Oncology Network Is Taking a New Approach to Physician Burnout
The US Oncology Network is confronting a mounting physician burnout crisis that now affects nearly 60% of oncologists, threatening cancer care quality. By deploying AI‑driven ambient scribes, streamlining EHR workflows, and promoting self‑care practices, the network aims to alleviate administrative...

Utah Measles Outbreak Reaches More than 600 Cases
Utah's measles outbreak has surged to 602 confirmed cases, according to the state Department of Health and Human Services, making it the largest U.S. outbreak this year. The majority of infections—405—occur among individuals who are unvaccinated or only partially vaccinated,...

OIG Reiterates a Core Message: Stark Compliance and Fair Market Value Alone Do Not Shield Against Anti-Kickback Statute Risk
On April 23, 2026 the HHS Office of Inspector General added two FAQs clarifying that compliance with Stark law or reliance on a fair‑market‑value analysis does not automatically shield a transaction from the federal Anti‑Kickback Statute. FAQ #4 warns that even...
White House Announces MFN Agreement with Drug Manufacturer Regeneron
The White House unveiled a most‑favored‑nation (MFN) pricing agreement with Regeneron covering its blockbuster eye‑disease drug Eylea. The pact obligates Regeneron to extend to Medicare the lowest net price it offers any private payer, with annual price reviews built into...

Elevate Podcast: Dr. Shoshana Ungerleider, Founder, End Well Project
The latest Elevate podcast episode features Dr. Shoshana Ungerleider, founder of the End Well Project, discussing how movies, television and broader pop culture shape public perceptions of end‑of‑life care. The conversation explores the ways media influence patient and family expectations,...

Healthcare AI Policy Must Keep Humans at the Center
Florida Senator Gayle Harrell, recipient of the HIMSS26 Policy Influencer Changemaker Award, called for AI regulations that keep clinicians in the decision loop. She warned that unchecked AI could erode clinician authority and patient safety. The remarks came amid rapid...

FDA Fast-Tracks the Development of Psychedelic Medications Following President’s Executive Order
The FDA announced an accelerated regulatory pathway for psychedelic‑based therapies after the April 18 executive order targeting serious mental illness. It will prioritize serotonin‑2A agonists for treatment‑resistant depression, PTSD and substance‑use disorders, and issue national‑priority vouchers for psilocybin and methylone studies...

HIMSS Membership Is a 'Game-Changer' For Legislators
HIMSS membership is being touted as a "game‑changer" for lawmakers, according to Florida Senator Gayle Harrell, a HIMSS26 Policy Influencer Changemaker. By joining the Health Information and Management Systems Society, legislators gain a structured channel to understand the health issues...

Building Payer-Provider Partnerships for a Stronger Health Ecosystem
At NAACOS Spring 2026, health‑system leaders argued that adversarial payer‑provider dynamics are stalling value‑based care. Panels from InnovaCare Health, Astrana Health, and Millennium Physician Group shared concrete strategies for collaborative contracts across Medicare Advantage, Medicaid, and commercial lines. Key themes...

How One HIMSS Member's Policy Work Is Shaped by Experience
Gayle Harrell, a Florida state senator and 2026 HIMSS Changemaker, used her experience running a medical practice to shape health‑IT legislation. Her frontline insight led to new Florida bills that update HITECH compliance and mandate electronic prescribing. The policies aim...

FDA Grants Three Priority Vouchers to Psychedelics
The FDA has granted three Commissioner's National Priority Vouchers to psychedelic‑focused firms—Compass Pathways, Transcend Therapeutics and the Usona Institute—following President Trump’s executive order to accelerate mental‑health treatments. All three companies already hold Breakthrough Therapy Designations, and the vouchers promise faster...

The Hidden Cost of Waiting: 5 Stats On What Gets Lost By Delaying Mobile Refresh
Home health agencies spend an average of $288 per caregiver on mobile devices, yet many postpone refresh cycles to save costs. Delaying upgrades drives higher replacement frequency, unplanned purchases, excess data usage, and forfeits residual device value. The practice also...

ALONE-AF: Stopping OAC After Ablation Doesn’t Reduce Cognitive Function
A substudy of the ALONE‑AF trial found that stopping oral anticoagulation (OAC) after successful catheter ablation did not lead to cognitive decline over two years. Montreal Cognitive Assessment (MoCA) scores rose modestly in both the OAC and no‑OAC groups, with...

Tortugas Neuroscience Nabs $106M
Tortugas Neuroscience announced a $106 million seed and Series A financing round, one of the larger early‑stage biotech raises this year. The capital, led by Cure Ventures and co‑led by The Column Group and AN Venture Partners, will fund the completion of...

AHA Urges Delay on TEFCA Individual Access SOP over Patient Privacy Concerns
The American Hospital Association (AHA) has urged the Sequoia Project to postpone the rollout of version 3.0 of the Trusted Exchange Framework and Common Agreement (TEFCA) Individual Access Services (IAS) Standard Operating Procedures, originally slated for August 2027. The AHA argues that...
A Common Stomach Bug Has Become Harder to Treat
The CDC’s latest report shows that extensively drug‑resistant (XDR) shigella now accounts for 8.5% of U.S. infections, up from zero cases in 2011. These XDR strains are unresponsive to the five antibiotics traditionally used for severe cases. While only about...

AI Learns to Predict Breast Cancer Risk From How Single Cells Respond to Pressure
Researchers at City of Hope and UC Berkeley unveiled a microfluidic platform, mechano‑NPS, that squeezes individual breast epithelial cells to gauge their mechanical response. By training a machine‑learning classifier called MechanoAge, they derived a "mechanical age" metric that correlates with...

The BioPharm Brief: Innovation, Gene Therapy Momentum, and Next-Gen ADCs Lead Today’s Headlines
The latest Ron Lanton Report shows biopharma innovation moving beyond Boston and San Francisco into emerging regions powered by academic collaborations, venture capital, and niche talent pools. The European Medicines Agency issued a positive opinion on Novartis’ intrathecal onasemnogene abeparvovec, expanding...

Chapters Health Unveils New Pediatric Hospice Program
Chapters Health System, one of the nation’s largest nonprofit hospice networks, has launched Chapters Kids, a pediatric hospice and palliative‑care program serving more than 300 seriously ill children across eight states. The model provides concurrent hospice, letting patients continue curative...

HFMA’s P&P Board Comments on GASB’s Proposed Implementation Guide, Financial Reporting Model Improvements-Subsidies
On April 24, 2026, the Healthcare Financial Management Association’s Principles and Practices Board submitted formal comments on the Governmental Accounting Standards Board’s Proposed Implementation Guide for Financial Reporting Model Improvements‑Subsidies. The board’s letter tackles five key areas: the proposed effective...

Advisory Details Shifting Tactics of Chinese Cyber Actors Using Covert Networks for Malicious Activity
A joint advisory from U.S. and international cyber agencies warns that China‑aligned threat actors have shifted to large‑scale covert networks of compromised routers, IoT devices, and other edge hardware. These hidden botnets let attackers mask origins, evade defenses, and target...

The Slow March of Clinical Biomarkers to Become Surrogate Endpoints
Clinicians are urging the FDA to recognize more clinical biomarkers as surrogate endpoints, hoping to accelerate drug approvals and reduce trial durations. Regulators remain cautious, approving only six biomarkers through the 2007 Biomarker Qualification Programme and often demanding definitive clinical...
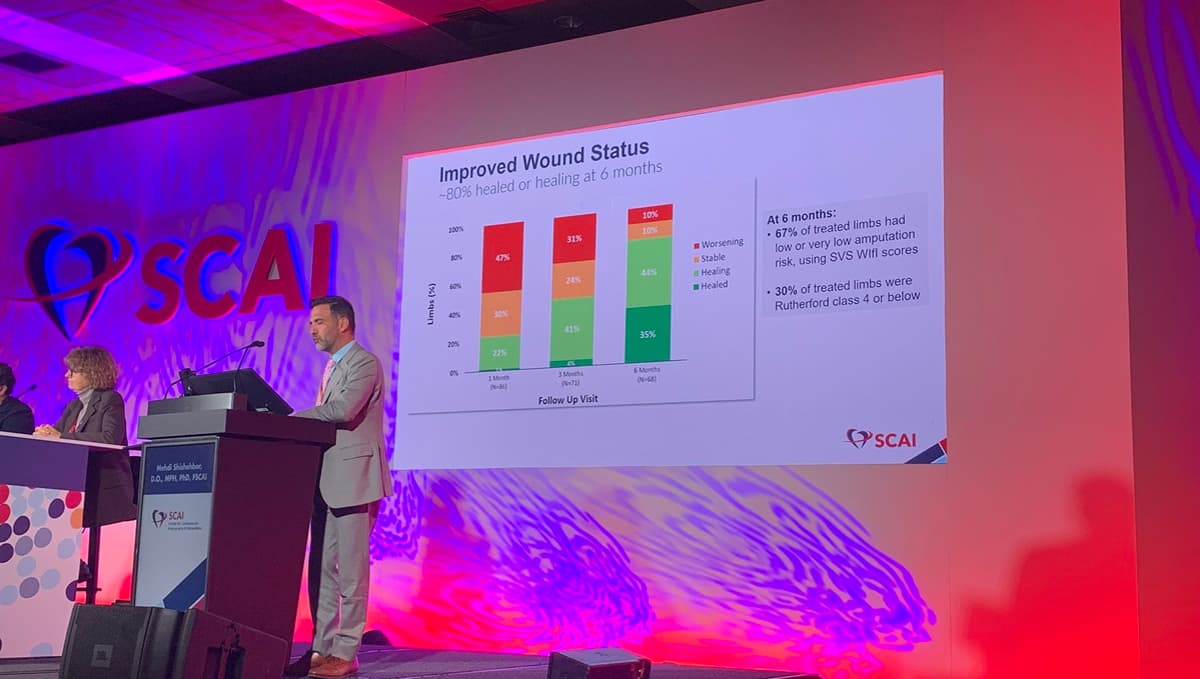
No-Option CLTI Patients Report Better QoL After Blood Flow Diversion Therapy: PROMISE III
The PROMISE III trial shows that transcatheter arterialization of the deep veins (TADV) using the LimFlow device markedly improves quality of life for no‑option chronic limb‑threatening ischemia (CLTI) patients. At six months, 80% of participants had healed or healing foot wounds...
Alphamab Oncology to Present Phase 1 Data on Bispecific ADC JSKN016 in HER2-Negative Breast Cancer at ASCO 2026
Alphamab Oncology will unveil Phase 1 results for its bispecific antibody‑drug conjugate JSKN016 at ASCO 2026, focusing on HER2‑negative breast cancer. The first‑in‑human trial in China demonstrated antitumor activity and a manageable safety profile in heavily pretreated patients, including triple‑negative cases. JSKN016...

IHS Leaders Tie Cybersecurity Directly to Patient Care
At the 2026 Splunk GovSummit, Indian Health Service leaders declared cybersecurity a core component of patient care. Serving 2.7 million patients across 37 states, IHS ties security to clinical continuity, emphasizing real‑time monitoring and resilience in remote and urban facilities. The...
Semaglutide NAION Risk Higher than Risk with SGLT2 Inhibitors
A VA‑based study published in JAMA Ophthalmology found that patients with type 2 diabetes taking semaglutide experienced more than double the risk of nonarteritic anterior ischemic optic neuropathy (NAION) compared with those on SGLT2 inhibitors. The analysis covered 102,361 veterans, with...

Policy Watch: FDA Moves to Implement Trump Order on Psychedelic Drugs
President Trump’s recent executive order tasked the FDA and ARPA‑H with accelerating psychedelic research. In response, the FDA issued Commissioner’s National Priority Vouchers to three firms—Compass Pathways, Transcend Therapeutics and the Usona Institute—fast‑tracking review of psilocybin and methylone candidates for...

STAT+: FDA to Speed up Review of Three Psychedelics as Mental Health Treatments
The FDA announced it will grant priority‑review vouchers to accelerate the evaluation of three psychedelic therapies—Compass Pathways’ psilocybin for treatment‑resistant depression, Usona Institute’s psilocybin for major depressive disorder, and Transcend Therapeutics’ MDMA‑like compound for PTSD. The move is part of...

Increasing Home Care Capabilities to Decrease Hospitalizations
Hospitals are shifting more high‑acuity patients to the post‑acute and home‑care setting to expand capacity. At Becker’s 16th Annual Meeting, leaders from Tampa General, PocketRN and Homewatch CareGivers highlighted how home care, family support and technology can prevent readmissions. Tampa...

How Supplier Collaboration Drives Medical Device Innovation and Accelerates Time to Market
Velosity argues that early collaboration with contract manufacturers is essential for medical‑device OEMs to innovate and accelerate time‑to‑market. By involving manufacturers from design‑for‑manufacturability through scalable planning, companies avoid costly redesigns and gain expertise in quality, regulatory and supply‑chain risk. Velosity’s...

Canadian Life Sciences Is at a “Generational Moment,” But Experts Disagree on Its Future
Canadian life sciences, contributing roughly 2% of GDP, is at a pivotal juncture as federal and provincial governments roll out new support mechanisms. BIOTECanada highlighted a federal task force, a Life Sciences Fund and BDC Capital’s $150 million CAD (~$110 million USD)...

Genomic Tool Untangles How Microbes Spread—Even when They Look Almost Identical
Researchers unveiled TRACS, a new genomic algorithm that pinpoints how microbes spread by detecting minute genetic differences. Published in Nature Microbiology, the tool successfully mapped transmission of SARS‑CoV‑2, Streptococcus pneumoniae, and Plasmodium falciparum across diverse cohorts. By distinguishing recent direct...

‘We Want to Be Known as Your Best Partner’: UnitedHealthcare Leaders Talk Provider Tensions, Prior Auth Cuts
UnitedHealthcare's commercial unit announced further cuts to prior authorization requirements, building on a 20% reduction already achieved and targeting an additional 30% as part of its CMS‑aligned commitment. The company highlighted a 40% increase in gold‑carded provider groups, noting that...

Medicare Advantage Buyers Beware: The Rules for Selling Plans Are Changin...
The Centers for Medicare & Medicaid Services (CMS) is easing marketing rules for Medicare Advantage (MA) plans, allowing brokers and agents to promote private‑insurance alternatives with fewer disclosure constraints. The change comes as MA enrollment already covers roughly 51% of...
:max_bytes(150000):strip_icc():format(jpeg)/xanax-withdrawal-symptoms-timeline-and-treatment-4685921_V2-8fa2660abc484c10a4cab7c2fa3029a3.png)
How Long Does Xanax Withdrawal Last?
Xanax (alprazolam) is widely prescribed for anxiety and insomnia, but even short‑term use can create physical dependence. Withdrawal symptoms can begin within 8‑12 hours, peak between days three and five, and may persist for weeks, with a protracted phase lasting...

St. Jude CFO to Step Down for West Virginia Role
Pat Keel, the chief financial officer of St. Jude Children’s Research Hospital, announced she will leave the Memphis‑based nonprofit after more than a decade to assume a new executive position in West Virginia. Keel joined St. Jude in February 2016...