
COSCIENS Posts $10.8M Q1 Profit, Exits AvenActive Program After German Unit Deconsolidation
COSCIENS Biopharma posted a $10.8 million net income for the quarter ended March 31, 2026, largely thanks to a $10.9 million gain from the deconsolidation of its German subsidiaries. The company also said it will discontinue the Phase 2a AvenActive program after the study failed to show statistically significant results.

AstraZeneca Reports the P-III (VOLGA) Trial Data on Imfinzi + Enfortumab Vedotin (EV) for Muscle-Invasive Bladder Cancer
AstraZeneca presented Phase III VOLGA data showing that the combination of Imfinzi (durvalumab) and Enfortumab Vedotin improves event‑free survival and overall survival in muscle‑invasive bladder cancer patients who cannot receive cisplatin. The trial enrolled 695 patients undergoing radical cystectomy and compared two...

Why AI Fails in Drug Development and How to Build Tools That Actually Deliver Real Value
Artificial intelligence can add real value to drug development only when it is tightly woven into existing expert‑led workflows and respects real‑world constraints. The authors, drawing on their experience at Intrepid Labs and Quotient Sciences, illustrate how AI tools that...

BeOne Wins Mantle Cell Lymphoma Approval, Opening New Therapy Class
The FDA granted accelerated approval to BeOne Medicines’ BCL2 inhibitor sonrotoclax, marketed as Beqalzi, for patients with relapsed or refractory mantle‑cell lymphoma who have failed at least two prior therapies, including a BTK inhibitor. The drug is the first BCL2...

Alembic Pharma Gets USFDA Tentative Approval for Generic Prostate Cancer Drug
Alembic Pharmaceuticals received tentative USFDA approval for its 300 mg generic Darolutamide tablets, deemed therapeutically equivalent to Bayer’s Nubeqa. The approval applies to an Abbreviated New Drug Application, clearing a major regulatory hurdle for the Indian firm. Darolutamide targets prostate cancer...

STAT+: Biogen’s Tau-Targeting Alzheimer’s Drug Posts Mixed Results in Mid-Stage Study
Biogen’s Phase 2 trial of the tau‑targeting Alzheimer’s drug diranersen (BIIB080) showed that the compound lowered tau protein in cerebrospinal fluid and brain tissue and was linked to a modest slowing of cognitive decline. The study evaluated three escalating dose levels,...
FDA Hands BeOne, Taiho Approvals for Blood Cancers
BeOne Medicines’ BCL‑2 inhibitor Beqalzi (sonrotoclax) earned FDA accelerated approval as a third‑line treatment for mantle‑cell lymphoma, marking the first U.S. approval of a BCL‑2 drug for this indication. The approval is based on a phase 1/2 trial showing a 52%...

Regulatory Round-Up: A Month of Key FDA Approvals for Cancer Drugs
The FDA approved four oncology drugs in May 2026, marking a focused regulatory push in cancer therapeutics. Taiho Oncology received clearance for the oral Inqovi‑venetoclax regimen for elderly acute myeloid leukemia patients, while BeOne Medicines earned accelerated approval for Beqalzi...

Imfinzi Set to Become First Immunotherapy for Stomach Cancer Patients on NHS
AstraZeneca’s immunotherapy Imfinzi (durvalumab) has received NICE approval, becoming the first immunotherapy available on the UK NHS for patients with resectable gastric and gastro‑oesophageal junction cancers. The approval follows the Phase III MATTERHORN trial, which showed that adding Imfinzi to standard...

Investigating the Causes of Rheumatoid Arthritis Pain
Researchers at Karolinska Institutet used AMSBIO's adeno‑associated virus (AAV) vectors to conditionally knock out the Ifnar1 gene in sensory neurons of mouse models of rheumatoid arthritis (RA). By pharmacologically blocking the IFN1/MNK‑eIF4E signaling cascade, they achieved significant relief of joint...

Precigen Narrows Q1 Loss as PAPZIMEOS Launch Fuels $21.6M Revenue
Precigen (PGEN) reported a first‑quarter net loss of $7.9 million, a sharp improvement from $54.2 million a year earlier, driven by $21.6 million in product revenue from its newly launched PAPZIMEOS therapy. The company said broad U.S. payer coverage and a permanent J‑code...

METiS TechBio Raises $270M in Record HKEX AI‑Biotech IPO
METiS TechBio listed on the Hong Kong Stock Exchange, selling 201.23 million H‑shares for HK$2.11 billion ($270 million). The offering, underwritten by Jefferies, Deutsche Bank Securities Asia and CITIC Securities, was oversubscribed more than 6,900 times, setting a new benchmark for AI‑driven biotech...

BeOne’s Next-Gen BCL2 Inhibitor Wins FDA Approval, Taking Aim at Venclexta
BeOne Medicines received accelerated FDA approval for sonrotoclax, its next‑generation BCL‑2 inhibitor, for mantle cell lymphoma. The decision rests on a single‑arm trial that delivered a 68% overall response rate in heavily pre‑treated patients. Sonrotoclax is positioned to compete directly...

Physical Fitness Does Not Strongly Influence Mainstream Epigenetic Clocks
Epigenetic clocks, built from DNA‑methylation patterns in blood, are widely used to estimate chronological age and mortality risk. A new systematic review and meta‑analysis of 44 studies involving 145,465 participants examined whether physical fitness influences these clocks. The analysis found...

METiS TechBio's $270 M AI‑Drug Delivery IPO Sets HKEX Record
METiS TechBio listed on the Hong Kong Stock Exchange, raising HK$2.11 bn ($270 m) and becoming the world’s first publicly traded AI‑powered drug‑delivery company. The offering was oversubscribed more than 6,900 times, drawing $148 m from 18 cornerstone investors including BlackRock. The IPO...
Molecular Grappling Hooks Boost Cancer Drug Retention and Tumor Shrinkage
Scientists led by Michael Evans and Charles Craik reported a nanodevice that physically tethers anticancer agents to tumor cell membranes. In mouse models the device cut tumor size more sharply than the drug alone while sparing healthy tissue. The breakthrough...

Propanc Biopharma Executes 1‑for‑25 Reverse Split to Retain Nasdaq Listing
Propanc Biopharma, Inc. (PPCB) approved a 1‑for‑25 reverse stock split that will take effect on May 18, 2026, reducing its outstanding shares from roughly 57 million to 2.28 million. The move is designed to bring the company back into compliance with Nasdaq's...

The Case for Smarter Neuroinflammation Clinical Trials
INmune Bio, led by CEO David Moss, is developing non‑immunosuppressive therapies that target innate immune pathways, including the XPro1595 program for neurodegenerative disease and the CORDStrom mesenchymal stromal cell platform for recessive dystrophic epidermolysis bullosa (RDEB). The company argues that...
AZ Cues up Broader Use of Imfinzi in Bladder Cancer
AstraZeneca’s immunotherapy Imfinzi (durvalumab) demonstrated a significant survival benefit in the phase‑3 VOLGA trial for muscle‑invasive bladder cancer (MIBC) patients who cannot receive platinum chemotherapy. The study combined Imfinzi with Pfizer‑Astellas’ antibody‑drug conjugate Padcev before surgery, extending both event‑free and...
Osel and Partners Launch Phase IIb FLIP-2 Trial of Lactin-V
Osel, Imperial College London and the March of Dimes have launched the Phase IIb FLIP‑2 trial to evaluate Lactin‑V, a live‑biotherapeutic containing Lactobacillus crispatus, for preventing preterm birth in high‑risk pregnancies. The study will enroll 360 women across four UK maternity...

Pfizer Receives EC Approval for Hympavzi to Treat Haemophilia
Pfizer secured European Commission marketing authorisation for Hympavzi (marstacimab) to treat hemophilia A or B patients with factor inhibitors aged 12 and older who weigh at least 35 kg. The once‑weekly subcutaneous therapy eliminates routine lab monitoring and delivered a 93%...

Pfizer Receives EC Approval for Hympavzi to Treat Haemophilia
Pfizer secured European Commission marketing authorisation for Hympavzi (marstacimab) to treat hemophilia A or B with inhibitors in patients aged 12 years and older weighing at least 35 kg. The once‑weekly subcutaneous therapy showed a 93% reduction in mean annualised bleeding rate versus on‑demand...

Taiho Oncology (Part of Taiho Pharmaceutical) Reports the US FDA Approval of Inqovi + Venetoclax for Newly Diagnosed AML
The U.S. Food and Drug Administration approved Inqovi (decitabine and cedazuridine) in combination with venetoclax for newly diagnosed acute myeloid leukemia patients who are 75 years or older or otherwise ineligible for intensive induction chemotherapy. The decision is based on...

Cell and Gene Therapy Catapult Invests in Lir Therapeutics to Accelerate Development of Its AI-Driven AAV Design Platform
The UK’s Cell and Gene Therapy Catapult is investing in Lir Therapeutics through Innovate UK’s Cross‑Catapult Investment Pilot to fast‑track Lir’s AI‑driven AAV capsid design platform, nAAVigator. The funding will support biological validation, benchmarking, and data generation to prove the platform’s...
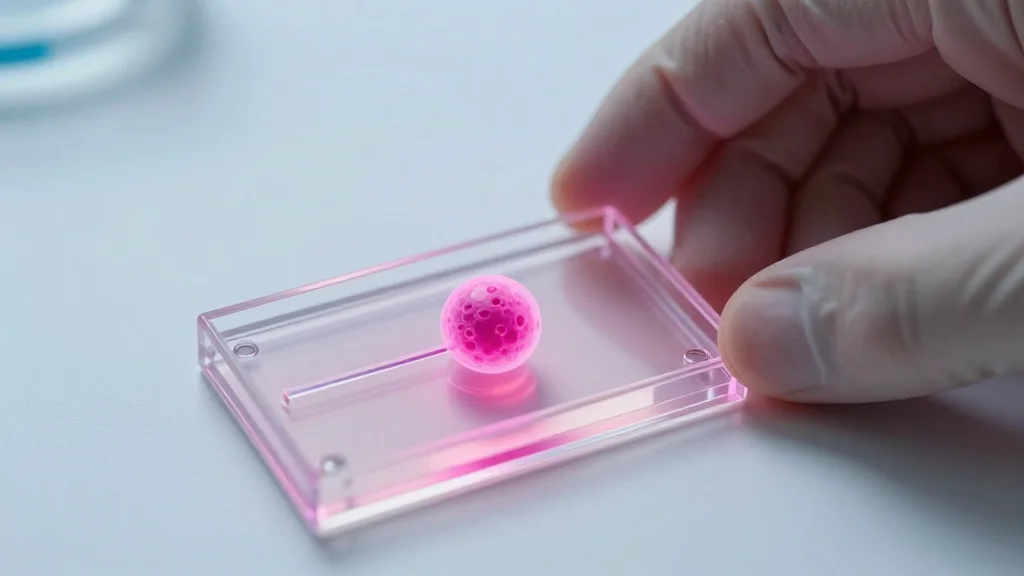
Living ‘Tumour on a Chip’ Could Offer New Brain Cancer Insights
Researchers at Nottingham Trent University, in partnership with biotech firm Kirkstall Ltd, have built a living tumour‑on‑chip that mimics the human blood‑brain barrier to study glioblastoma. The microfluidic device cultivates patient‑derived brain‑cancer cells alongside a synthetic barrier, enabling real‑time monitoring...

BeOne Medicines Receives US FDA Accelerated Approval for Beqalzi (Sonrotoclax) to Treat R/R Mantle Cell Lymphoma
BeOne Medicines received FDA accelerated approval for its BCL‑2 inhibitor Beqalzi (sonrotoclax) to treat adults with relapsed or refractory mantle‑cell lymphoma after at least two prior lines, including a BTK inhibitor. The decision was based on a global Phase I/II...
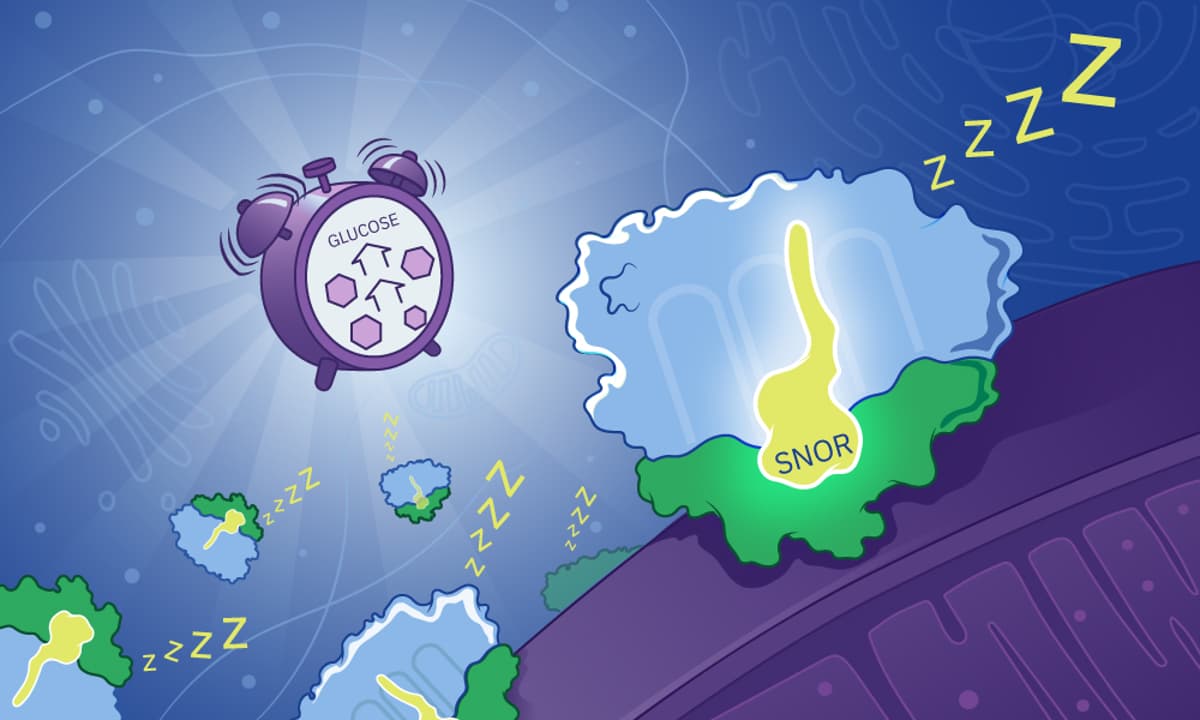
Providing a Cellular ‘All-Clear’ Signal to Resume Protein Synthesis
Scientists at EMBL and the University of Virginia identified a previously unknown protein, SNOR, that binds to ribosomes in glucose‑starved yeast and induces a dormant, low‑translation state. Using in‑situ cryo‑electron tomography and visual proteomics, they mapped SNOR at the ribosomal...
Argenica Therapeutics Establishes Expert Committee to Advance Phase 2b Stroke Trial
Argenica Therapeutics has created a Clinical Advisory Committee to steer the design of its Phase 2b trial of the neuroprotective agent ARG‑007 for acute ischemic stroke. The committee, featuring leading stroke neurologists such as Professors Geoffrey Donnan and Jeffrey Saver, will...
BMS Signs $15.2 B Deal with China's Hengrui on 13 Drug Programs
Bristol Myers Squibb has agreed to pay $600 million up front and commit up to $15.2 billion in milestones to partner with China’s Hengrui Pharma on 13 early‑stage oncology and immunology candidates. The deal splits rights by geography and therapeutic area, giving...
FDA Clears Oral Decitabine/Cedazuridine + Venetoclax for Elderly AML Patients
The FDA approved the oral combination of decitabine/cedazuridine (Inqovi) and venetoclax (Venclexta) for newly diagnosed acute myeloid leukemia patients who are 75 years or older or otherwise ineligible for intensive chemotherapy. The decision rests on phase‑2 ASCERTAIN‑V data showing a...

Atrium Therapeutics Inc (RNA) Q1 2026 Earnings Call Transcript
Sarepta Therapeutics reported Q1 2026 revenue of $731 million, a modest 2% decline, while posting GAAP operating profit of $358 million and ending the quarter with $748 million in cash. The company reiterated full‑year revenue guidance of $1.2‑$1.4 billion and highlighted non‑cash collaboration income...

Benitec Biopharma Inc (BNTC) Q3 2026 Earnings Call Transcript
Bio-Techne reported Q3 2026 revenue of $311.4 million, a 2% decline year‑over‑year, while adjusted operating margin improved to 34.2% after a 310‑basis‑point sequential gain. The company highlighted strong cash flow, $86.7 million operating cash, and a solid balance sheet with...

Navigating the Clinical Progress of Antibody-Drug Conjugates: Emerging Opportunities and Remaining Challenges
Antibody‑drug conjugates (ADCs) have become one of the fastest‑growing anticancer modalities, reaching 19 FDA‑approved products worldwide by the third quarter of 2025. The portfolio has expanded from hematologic indications to a broad array of solid tumors, driven by successful HER2‑targeted...

Sutro Biopharma Inc (STRO) Q1 2026 Earnings Call Transcript
Sutro Biopharma reported Q1 2026 total revenue of $83.2 million, driven by $64.7 million in collaboration and license fees and $18.5 million in U.S. Iptrozy product sales, which grew 18% quarter‑over‑quarter. For the first time, more than half of new Iptrozy patients were...

Sangamo Therapeutics Inc (SGMO) Q1 2026 Earnings Call Transcript
Sangamo Therapeutics announced that the FDA reaffirmed the use of eGFR slope as an endpoint for an accelerated Biologics License Application for its Fabry gene therapy ST-920, positioning a filing as early as Q1 2026. The STAR study showed a mean...

Recurrent Strokes Less Severe on Asundexian: OCEANIC-STROKE
The OCEANIC‑STROKE trial of 12,327 patients showed that the factor XIa inhibitor asundexian reduced recurrent ischemic strokes by 26% versus placebo in secondary prevention. Additional analyses revealed that strokes occurring on asundexian were less severe, with fewer NIHSS scores ≥8 and...

FDA Approves Rocket's Kresladi, First Gene Therapy for Ultra‑Rare LAD‑1
Rocket Pharmaceuticals announced that the U.S. Food and Drug Administration has approved Kresladi, its inaugural gene‑therapy product, to treat leukocyte‑adhesion deficiency type 1 (LAD‑1). The approval marks the first gene‑editing treatment for this ultra‑rare pediatric immunodeficiency and signals a new commercial...
New Drug Candidate that Reprograms the Immune System Shows Promise as a Brain Cancer Treatment
McMaster University researchers have engineered a uPAR‑targeted CAR‑T cell that reprograms the immune system to attack glioblastoma. In preclinical mouse studies, a single infusion of 1 × 10⁶ engineered T cells eradicated established brain tumors and prevented recurrence, outperforming unmodified T cells....
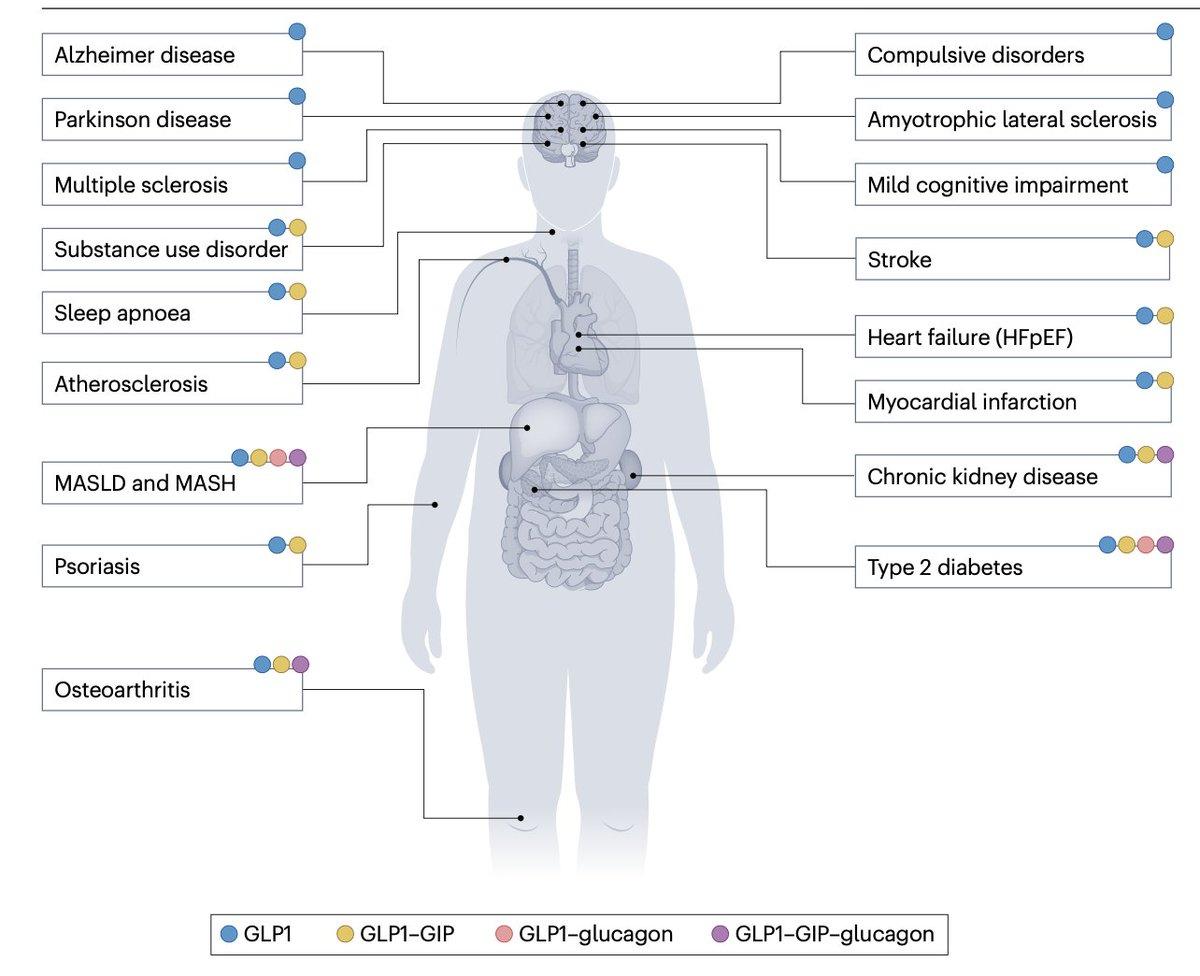
GLP‑1/GIP/Glucagon Drugs Target New
Just a few conditions being pursued for secondary prevention or treatment by GLP-1/GIP/Glucagon drugs in a new and comprehensive review @NatRevDrugDisc https://t.co/pjfc2jkAGU

Drug Trials Snapshots: Forzinity
Stealth Biotherapeutics’ mitochondrial cardiolipin binder FORZINITY was granted FDA accelerated approval on September 19 2025 for treating muscle weakness in Barth syndrome patients weighing at least 30 kg. The pivotal SPIRA‑201 study enrolled 12 male patients in a randomized, double‑blind, placebo‑controlled crossover followed...

How Biologic Developers Are Using Optimized Platforms and Operations to Reach First-in-Human Trials
Catalent’s Emily Schirmer explains that early chemistry, manufacturing and controls (CMC) decisions and early engagement with a capable CDMO can dramatically shorten the path to first‑in‑human (FIH) trials. By evaluating the full supply chain—drug substance, product, fill‑finish and distribution—developers avoid...

Teaching Your Body To Make Designer Antibodies
A study published in Science demonstrates that a handful of gene‑edited blood‑forming stem cells can turn the body into a self‑sustaining factory for designer antibodies. In mice, as few as 7,000 edited cells produced therapeutic antibody levels that were boosted...

FDA Approves Grifols Malaria Blood Screening Assay
The U.S. Food and Drug Administration has cleared Grifols’ Procleix Plasmodium assay for malaria screening of blood donors. The nucleic‑acid test uses magnetic target capture, transcription‑mediated amplification and chemiluminescence to identify ribosomal RNA from five human‑infecting Plasmodium species. It runs on...

Is Argenx SE (ARGX) Among the Most Promising Biotech Stocks to Buy?
Argenx SE (NASDAQ:ARGX) announced that the FDA has broadened Vyvgart and Vyvgart Hytrulo to treat all serotypes of adult generalized myasthenia gravis, including anti‑AChR, anti‑MuSK, anti‑LRP4 and triple‑seronegative patients. In its Q1 2026 report, the company posted $1.3 billion in global product...

Development of Local Anesthetic Drug Products With Prolonged Duration of Effect
The FDA released a draft Level 2 guidance (Docket FDA‑2023‑D‑0608) on developing local anesthetic products with prolonged duration of effect. The document outlines recommended development pathways, trial designs, and data requirements for future new‑drug applications. It aims to help sponsors generate evidence...

Cellares and ProTgen Automate Manufacturing of Progenitor T-Cell Therapy for Blood Cancer
Cellares, an integrated development and manufacturing organization, has partnered with ProTgen to automate the production and quality control of ProT-096, a personalized progenitor T‑cell therapy for refractory leukemia and other blood cancers. Cellares will deploy its Cell Shuttle and Cell...

Lilly Highlights Maintenance Strategies for GLP‑1 Weight Loss
Lilly data point to ‘maintenance’ strategies for GLP-1 weight loss https://t.co/LGyx82AJKf by Kristin Jensen $LLY $NVO

Current Clinical Trials of Alzheimer’s Drugs
The 2026 annual report on Alzheimer’s drug development lists 158 therapies across 192 active clinical trials, enrolling 54,728 participants. Over the past decade the number of trials and tested agents grew by roughly 35% and 40%, respectively, with 59 new...

FDA Clears ArteraAI Breast, First AI Tool to Guide Chemotherapy for Early‑Stage Breast Cancer
Artera announced FDA clearance of its ArteraAI Breast digital pathology risk‑stratification tool, the first AI‑driven test approved to guide chemotherapy decisions for early‑stage hormone‑receptor‑positive, HER2‑negative breast cancer. The multimodal model, trained on data from more than 8,500 patients, provides oncologists...

Adopting Creative Chemistry to Optimize Bioprocessing Workflow
Professor Sunny Zhou of Northeastern University argues that creative chemistry can mitigate unique bioprocessing challenges of antibody‑drug conjugates (ADCs). He highlights two major vulnerabilities: light‑sensitive payloads that cause aggregation and linker cleavage by host‑cell enzymes, both of which can compromise...