
Testosterone Treatment Found to Improve Sexual and Physical Function for Men After Prostate Cancer Surgery
A randomized SPIRIT trial led by Shalender Bhasin showed that three months of testosterone replacement therapy (TRT) markedly improved sexual activity, desire, physical function, and aerobic performance in men who had undergone radical prostatectomy. The study enrolled 136 men with low testosterone and undetectable PSA levels, assigning them to weekly testosterone injections or placebo for 12 weeks, with an additional 12‑week observation period. No participants experienced biochemical cancer recurrence, and quality‑of‑life scores rose across multiple domains, though erectile function remained unchanged. Results were published in JAMA Internal Medicine, suggesting TRT can be safe and beneficial for prostate‑cancer survivors with hypogonadism.

News Wrap: Supreme Court Temporarily Extends Access to Mifepristone
The U.S. Supreme Court issued an administrative stay on Friday, extending access to the abortion pill mifepristone through at least Thursday. The stay preserves telemedicine prescribing and mail delivery while justices consider an emergency request to block a lower‑court ruling...

The BioPharm Brief: Precision Medicine Expansion Accelerates Autoimmune and Targeted Oncology Development
The FDA broadened Vyvgart’s label to cover all adult patients with generalized myasthenia gravis, removing the previous antibody‑status restriction. Zai Lab received fast‑track designation for its DLL3‑targeting antibody‑drug conjugate aimed at extrapulmonary neuroendocrine carcinomas, a disease with limited options. The...
FDA Seeks Input On Chronic Diseases To Prioritize For Drug Repurposing
The FDA announced a public‑comment period to identify chronic diseases that should be prioritized for drug‑repurposing. The agency seeks stakeholder input on which conditions lack commercial interest and which approved drugs could be redirected to new therapeutic uses. This effort...

Supreme Court Continues Access to Abortion Pill by Mail, for a Few Days
The U.S. Supreme Court, through Justice Samuel A. Alito Jr., extended a temporary stay that keeps the abortion pill mifepristone available by mail nationwide until 5 p.m. on May 14. The pause follows an emergency petition by the drug’s manufacturers to block...

DOJ, DEA Reschedule Medical Cannabis Products
The U.S. Justice Department and DEA have reclassified FDA‑approved and state‑licensed medical cannabis products from Schedule I to Schedule III under the Controlled Substances Act. The change follows a 2025 Trump executive order and a 2024 DEA proposal that attracted 43,000 public...

Spotlight On: Biosimilar Litigations - May 2026
The May 2026 Venable LLP update delineates which disputes qualify as biosimilar litigations. It includes lawsuits between biosimilar applicants or manufacturers and reference‑product sponsors, as well as conflicts among biosimilar firms themselves. It expressly excludes disputes solely between reference‑product sponsors, university‑sponsor...
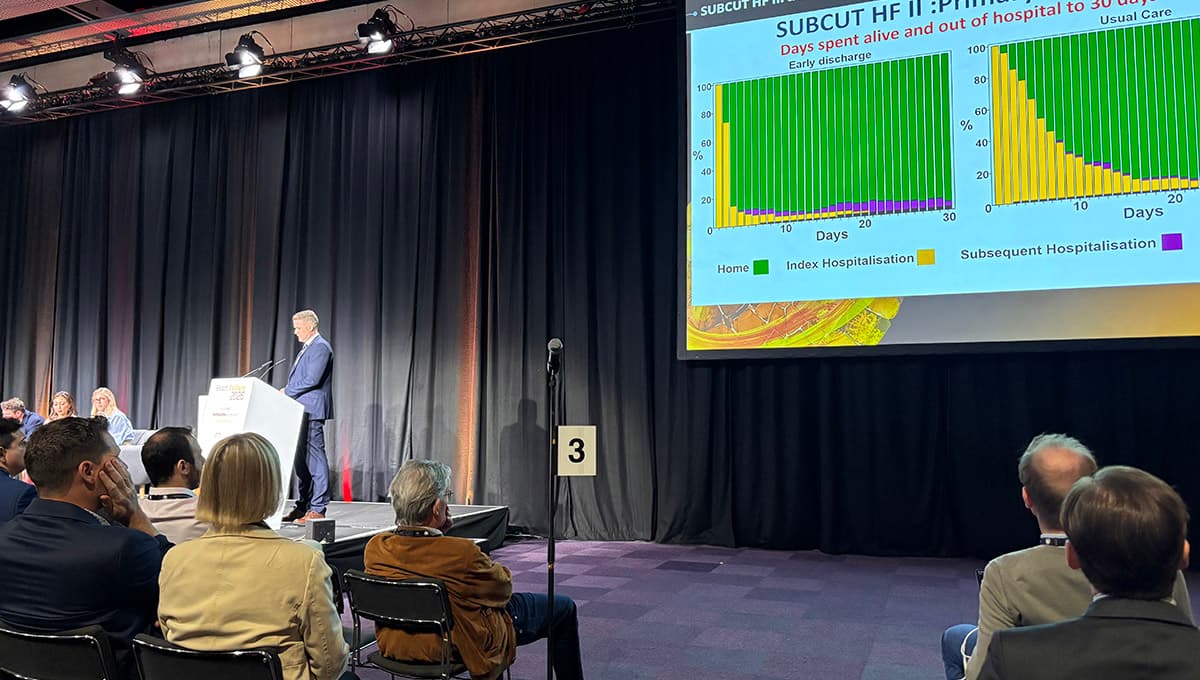
Subcutaneous Furosemide Aids With Earlier Discharge: SUBCUT II HF
A randomized SUBCUT II HF trial in 22 UK hospitals showed that subcutaneous furosemide delivered via a minipump enables safe early discharge for heart‑failure patients. The early‑discharge arm reduced average hospital stay from 11.0 to 5.6 days and added four additional days...

BIOTECanada Statement on Canada–Mexico Life Sciences Collaboration
BIOTECanada signed two memoranda of understanding with Mexico’s Asociación Mexicana de Laboratorios Farmacéuticos (AMILAF) and La Cámara Nacional de la Industria Farmacéutica (CANIFARMA), marking a new phase of life‑sciences collaboration. The MoUs were part of Mexico’s trade mission to Canada,...

A New Hantavirus Vaccine Is in the Works
Moderna announced that it is co‑developing an mRNA‑based hantavirus vaccine with Korea University’s Vaccine Innovation Center, a partnership that began in 2023. The effort follows a deadly outbreak on a Dutch cruise ship that killed three passengers and highlighted the...

FDA Alerts Health Care Providers and Patients About Increased Risk of New Blood Cancers with Tazverik (Tazemetostat) Use; Sponsor to...
The FDA has announced a voluntary market withdrawal of Tazverik (tazemetostat) after a Phase 1b/3 SYMPHONY‑1 trial revealed a 5.7% incidence of hematologic second primary malignancies (SPMs) in treated patients, compared with none in the control arm. The drug, approved in...

Advancing Aseptic Precision: How Modern Filling Lines Elevate Sterility, Flexibility, and Throughput
Modern pharmaceutical filling lines are adopting isolator‑based, fully automated systems that integrate washing, sterilization, filling, sealing, and environmental control. Automation and AI‑driven analytics are boosting sterility assurance, reducing human intervention, and meeting stricter EU GMP Annex 1 and FDA requirements. These...

Advancing Generic Drug Development: Bioequivalence Challenges for Patient-Centric Oral Formulations - 06/11/2026
The FDA’s Center for Drug Evaluation and Research hosted a two‑day workshop on June 11, 2026 to address bioequivalence challenges in patient‑centric oral generic drugs, including pediatric formulations. Experts discussed development hurdles for chewable tablets, orally disintegrating tablets, granules, suspensions, and sublingual...
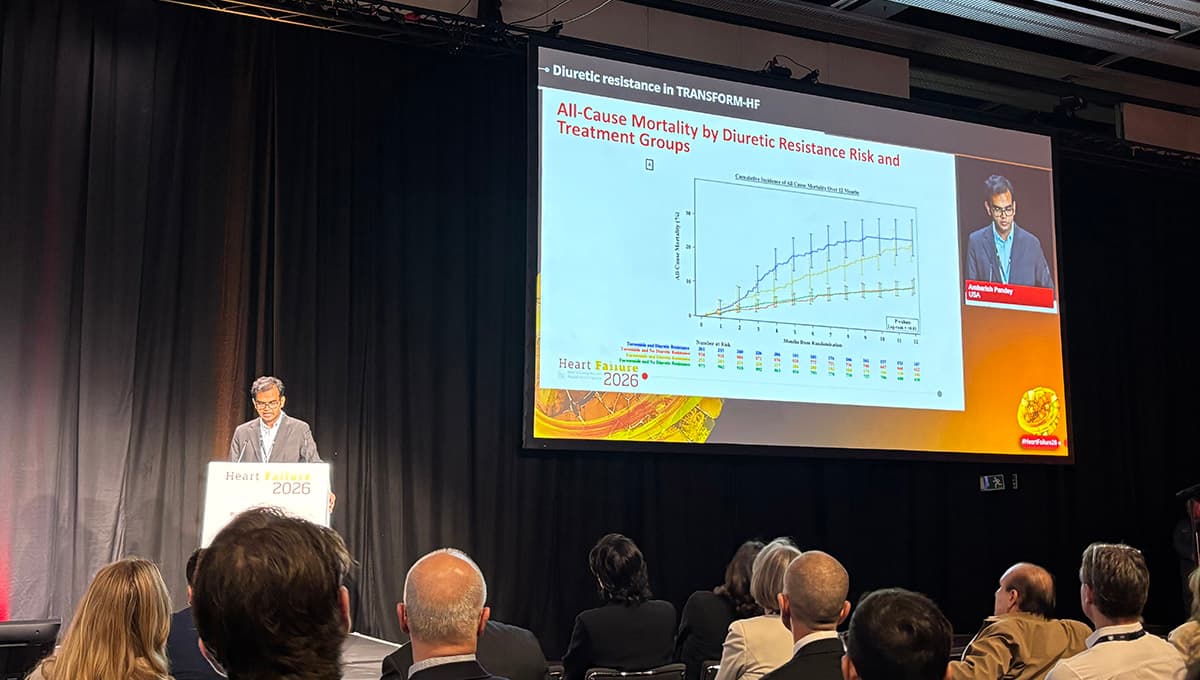
Diuretic Resistance Score Confirms TRANSFORM-HF’s Message
A post‑hoc analysis of the TRANSFORM‑HF trial applied the BAN‑ADHF risk score to identify patients at high risk of diuretic resistance. About 21% of the 2,421 participants scored above 12, indicating a markedly worse prognosis with 79% higher mortality and...

In the Clinic for May 11, 2026
BioWorld’s May 11, 2026 “In the Clinic” roundup aggregates the day’s most relevant biopharma and med‑tech snapshots, special reports, and infographics. The page links to data‑driven analyses covering topics from mRNA vaccine research and China’s GLP‑1 landscape to pulsed‑field ablation for atrial...

New Home for Novo's Parkinson's Cell Therapy; GSK's Deal to Sell Drug in China
Novo Nordisk has transferred its early‑stage Parkinson's disease cell therapy to a specialized biotech partner, allowing the pharma giant to offload development risk while retaining royalty rights. GSK secured a distribution agreement to launch its flagship drug in China, opening...

Inhibrx Says Combo Therapy Shrank More Tumors than Merck's Keytruda Alone
San Diego‑based biotech Inhibrx reported that its experimental antibody INBRX‑106, when paired with Merck’s immunotherapy Keytruda, produced a higher rate of tumor shrinkage than Keytruda alone in patients with metastatic head and neck squamous cell carcinoma. The early‑stage study enrolled...
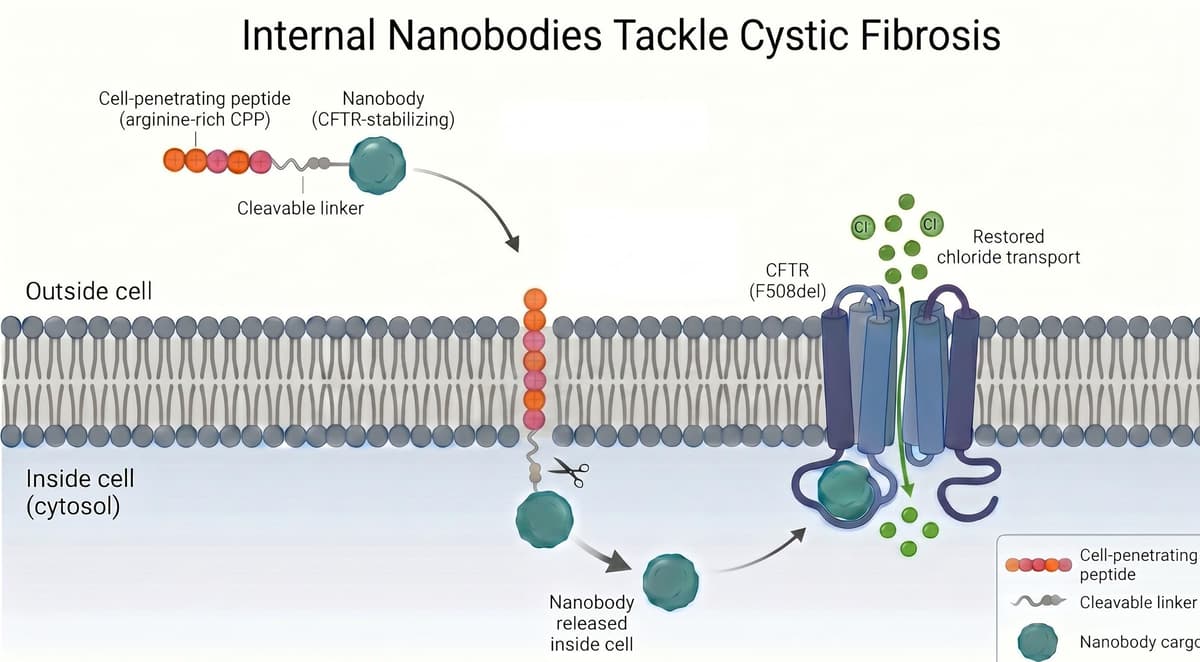
Internal Nanobodies Tackle Cystic Fibrosis
Researchers have engineered a cell‑penetrating nanobody that enters airway cells and stabilizes the misfolded CFTR protein responsible for cystic fibrosis. By fusing the nanobody to a ten‑arginine peptide, the hybrid molecule crosses the cell membrane and restores up to 90%...

Delos Capital and AP Biosciences Launch Collaboration to Create New Biotech Companies
Delos Capital and AP Biosciences have announced a strategic collaboration to launch and incubate new biotechnology companies focused on next‑generation antibodies. Through its Delos Foundry, Delos will lead therapeutic ideation, financing, and strategic development, while APBio supplies its proprietary antibody...
Ocrevus Slows Disability Progression in Advanced PPMS, Trial Finds
A Phase 3 ORATORIO‑HAND trial involving more than 1,000 adults with primary progressive multiple sclerosis (PPMS) showed that Ocrevus (ocrelizumab) significantly slows disability progression. Over a median follow‑up of nearly three years, the drug reduced the risk of confirmed disability...
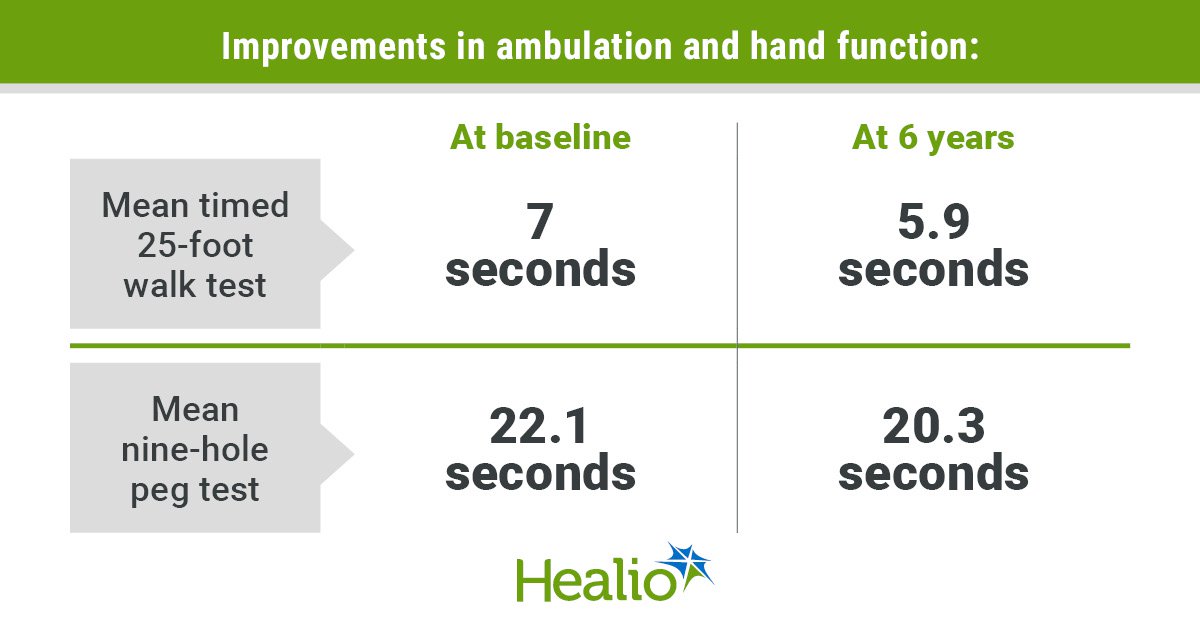
Ocrelizumab Preserves Ambulation, Hand Function in MS
A six‑year analysis of the ENSEMBLE trial shows that early‑stage relapsing‑remitting multiple sclerosis patients treated with ocrelizumab largely maintained functional ability. 86.1% preserved normal ambulation and 93% kept normal hand dexterity throughout the study, while 34% of those with baseline...

Braveheart Bio's Hengrui-Licensed Cardiac Drug Scores Second Clinical Win
Braveheart Bio announced that its heart‑muscle therapy, licensed from China’s Jiangsu Hengrui Pharmaceuticals, achieved its primary endpoint in a mid‑stage (Phase 2) trial for heart failure with reduced ejection fraction. The study reported an 8% absolute improvement in left‑ventricular ejection fraction...

Drug Repurposing
The FDA has issued a request for public comment on drug repurposing, seeking input from patients, clinicians, and researchers on priority disease areas and candidate drugs. The initiative builds on existing programs such as Project Renewal, which updates oncology drug...

The Next Frontier for Hantavirus: Finding Vaccines and Treatments
A hantavirus outbreak on a cruise ship has killed three people and sickened several others, marking the first documented person‑to‑person transmission of the disease. The strain’s heightened transmissibility has revived interest in vaccine and therapeutic candidates that were previously shelved...

STAT+: Pharmalittle: We’re Reading About Medicare and Alzheimer’s Drugs, Estrogen Patch Shortages, and More
Medicare enrollment in the newly approved Alzheimer’s therapies Leqembi and Kisunla is dramatically lower than the federal government expected. The agency now projects minimal spending on the drugs through 2027, a sharp reversal from earlier forecasts of billions in annual...

QP Essentials: Reducing Risk and Delays in EU and UK Trials
Clinical trial sponsors often encounter delays when shipping supplies to the EU and UK because Qualified Persons must certify each batch. Catalent’s webinar outlines the core QP responsibilities, highlights differences between EU and UK regulations, and offers practical steps such...
FDA Grants Fast Track Designation to Zai Lab’s DLL3-Targeting ADC for epNECs
Zai Lab’s DLL3‑targeting antibody‑drug conjugate zocilurtatug pelitecan received FDA Fast Track designation for extrapulmonary neuroendocrine carcinomas (epNECs). Early‑stage data showed a 38.2% objective response rate in heavily pre‑treated patients, indicating meaningful antitumor activity. The designation promises more frequent FDA interactions,...
Study Compares Two Antibiotics in Treating Severe Hemorrhagic Bacterial Pneumonia
Researchers at Osaka Metropolitan University compared cefiderocol (CFDC) and levofloxacin (LVFX) in a mouse model of severe hemorrhagic pneumonia caused by multidrug‑resistant Stenotrophomonas maltophilia. Both drugs significantly increased survival and reduced bacterial loads in lungs and heart relative to untreated...

Inside CSL’s Strategy to Simplify a Biotech Giant
CSL Limited is entering a strategic simplification phase, announced in August 2025, that will see its vaccine arm Seqirus spun off as a separate ASX‑listed company by the end of 2026 and a restructuring program that could cut up to 15% of...
Novo Nordisk microRNA Drug Fluffs Its Lines in Heart Failure
Novo Nordisk’s microRNA‑targeting drug CDR132L failed to demonstrate a statistically significant improvement in left ventricular end‑systolic volume index in the phase 2 HF‑REVERT trial. The study enrolled 280 post‑myocardial‑infarction patients with an ejection fraction of 45% or lower and elevated NT‑proBNP...
FUJIFILM Biosciences and NextCell Launch New Commercial Platform Comprised of RUO Stromal Cells and Cell Culture Media
FUJIFILM Biosciences and NextCell Pharma have launched a global commercial platform that pairs FUJIFILM’s PRIME‑XV MSC Expansion XSFM medium with NextCell’s research‑use‑only umbilical‑cord mesenchymal stromal cell product, NextCell‑Cord RUO. The bundle offers a standardized, scalable solution for MSC research, addressing...

Novo Hands Over Parkinson’s Therapy to Zuckerberg-Backed Startup
Novo Nordisk agreed to transfer its experimental Parkinson’s disease therapy to Cellular Intelligence, an AI startup backed by Mark Zuckerberg, to accelerate development. Cellular Intelligence will assume responsibility for clinical‑trial execution, while Novo retains rights to milestone payments and royalties...
GSK Enlists Local Aid for Chinese Rollout of Hep B Drug
GlaxoSmithKline (GSK) has signed a five‑and‑a‑half‑year agreement with Sino Biopharm’s subsidiary Chia Tai Tianqing Pharma (CTTQ) to import, distribute and promote its investigational hepatitis B antisense drug bepirovirsen across more than 5,000 Chinese medical centres, pending NMPA approval. The partnership gives...

Makary’s Reported FDA Removal Could Be ‘Broad Positive’ for Biopharma
Industry sources report that President Donald Trump has approved a plan to remove FDA Commissioner Marty Makary, following the recent departure of CBER head Vinay Prasad. Makary’s tenure has been marked by calls for greater transparency but also criticism over...

Makary’s Reported FDA Removal Could Be 'Broad Positive' For Biopharma
President Donald Trump reportedly agreed to remove FDA Commissioner Marty Makary, a move confirmed by the Wall Street Journal and echoed by anonymous sources. The announcement follows the recent departure of CBER director Vinay Prasad and adds to a pattern of...

PulseSight Therapeutics Reports P-I (PST-611-CT1) Trial Data on PST-611 in Dry AMD/Geographic Atrophy
PulseSight Therapeutics presented Phase I (PST-611-CT1) trial data for PST-611 in dry age‑related macular degeneration (geographic atrophy) at ARVO 2026. The study enrolled six patients across two dose cohorts and demonstrated favorable safety, tolerability, stable best‑corrected visual acuity, and no...

Deal Finally Unlocks NHS Access to ITF's Duchenne Drug
ITF Pharma’s oral HDAC inhibitor Duvyzat has secured NICE endorsement for ambulatory Duchenne muscular dystrophy patients, unlocking access for more than 500 NHS England cases. The drug will be funded immediately through the Innovative Medicines Fund while NHS England arranges...

European Regulators Greenlight Fractyl Health's Clinical Test of GLP-1 Gene Therapy
European regulators have granted Fractyl Health permission to begin the first human trial of a GLP‑1 gene‑therapy candidate. The Phase 1 study will test a single‑dose AAV‑based vector designed to produce continuous GLP‑1, aiming to replace daily injections for type 2 diabetes...

Quality CDMO, Inc. - 665961 - 05/01/2026
The U.S. Food and Drug Administration issued a closeout letter to Quality CDMO, Inc., confirming that the firm’s corrective actions have addressed the violations cited in the September 11, 2024 warning letter. While the FDA acknowledges the remediation, it emphasizes that compliance...

CRISPRi Screening Identifies Fungal-Specific Drug Targets
Researchers adapted a pooled CRISPR interference (CRISPRi) platform for Candida albicans, allowing high‑throughput repression of 130 essential, fungal‑specific genes. The screen revealed dosage‑sensitive vulnerabilities across ten infection‑relevant stress conditions. Testing two drug‑resistant clinical isolates showed most hits are conserved, indicating...

FDA Locks Down Post-Approval Pregnancy Safety Data Framework
The FDA issued final guidance on post‑marketing safety data collection for drugs and biologics used in pregnancy. The framework details best practices for pregnancy registries, case‑control, electronic health record‑based and population‑level studies, stressing early enrollment and multi‑pronged recruitment. It requires...

A Single Infusion Could Suppress H.I.V. for Years, Study Suggests
Scientists presented early data showing that a single infusion of genetically engineered immune cells can drive HIV to undetectable levels in patients. In a small study, two participants received the therapy, with one maintaining viral suppression for almost two years....

FDA Cliffhanger: Will Marty Makary Stay or Go?
The FDA’s leadership is in flux as reports suggest President Trump may fire Commissioner Marty Makary, though the president has so far remained noncommittal. Makary, a Johns Hopkins physician, has overseen controversial moves on abortion pills, flavored vapes, and accelerated...

Precision in Motion: Decoding the Critical Operations Behind the Logistics of Next-Generation Cell and Gene Therapies
Cell and gene therapies (CGTs) are transforming personalized medicine but demand ultra‑precise logistics because they are patient‑specific, highly sensitive, and often viable for only minutes to hours. Traditional freight cannot meet the sub‑30‑minute delivery windows, cryogenic temperature requirements, and real‑time...

The Capacity Crunch in Biopharma Is a Location Problem
Biopharma manufacturers are hitting a capacity crunch as demand for cell and gene therapies outpaces the construction of new GMP‑ready plants, which can take years. Companies are therefore prioritizing locations where compliant facilities, utilities and cold‑chain logistics already exist. Middlesex...

Day One Concludes Patient Enrolment for Tovorafenib’s FIREFLY-2 Trial
Day One Biopharmaceuticals announced that the Phase III FIREFLY‑2 trial of tovorafenib has finished enrolling roughly 400 pediatric low‑grade glioma patients across 140 sites worldwide. The open‑label, randomized study pits oral tovorafenib against four standard chemotherapy regimens as a first‑line option...
STAT+: Medicare Is Spending Far Less than Expected on New Alzheimer’s Drugs
Medicare’s uptake of the newly approved Alzheimer’s therapies Leqembi and Kisunla is far lower than projected, prompting the agency to forecast minimal spending through 2026‑2027. Earlier estimates had anticipated billions in annual costs, but real‑world adoption has stalled. The drugs...

Henlius Receives US FDA IND Clearance for HLX05-N (Biosimilar, Erbitux)
Henlius announced that the U.S. Food and Drug Administration granted Investigational New Drug (IND) clearance for HLX05‑N, its biosimilar version of Erbitux (cetuximab), aimed at treating metastatic colorectal cancer. The IND follows extensive analytical and non‑clinical studies that demonstrated high...

Nuclera Launches Antibody Triage Service to Advance AI-Driven Antibody Discovery
Nuclera, a biotech firm specializing in protein production, announced a new antibody‑screening service designed to accelerate AI‑driven antibody discovery. The offering uses a 96‑plex cell‑free expression platform and surface plasmon resonance to triage large in‑silico‑generated libraries, delivering early binding data...

Bologna’s Cellply Secures €7.15 Million to Develop Tools for Faster Immunotherapy and Cell Therapy Development
Italian deep‑tech startup Cellply has closed a €7.15 million ($7.8 million) funding round led by ENEA Tech e Biomedical. The capital will fund global rollout of its VivaCyte® single‑cell analysis platform and development of next‑generation ATMP quality‑control tools. The round also saw...