Pim1 Identified as Promising Therapeutic Target for Inflammatory Arthritis Treatment
Researchers have pinpointed the serine/threonine kinase Pim1 as a pivotal driver of abnormal Th17 cell differentiation in inflammatory arthritis. Elevated Pim1 levels were detected in CD4⁺ T cells from rheumatoid arthritis and ankylosing spondylitis patients, and mice lacking Pim1 in these cells displayed markedly reduced joint swelling, cartilage loss, and bone erosion. Mechanistic studies revealed that Pim1 fuels mitochondrial calcium influx and oxidative phosphorylation, supplying the metabolic energy needed for pathogenic Th17 development. Screening of FDA‑approved compounds identified Nilotinib as a potent Pim1 inhibitor that mitigates disease severity in mouse models, opening a new therapeutic avenue.
Gene-Based Therapies Could Transform Future Pancreatitis Treatment
Pancreatitis remains a largely untreatable inflammatory disease, with current care limited to symptom management. Gene‑based therapies—spanning augmentation, inhibition, and editing—are emerging to address the genetic drivers across lipid‑metabolism, trypsin regulation, ductal secretion, and ER‑stress pathways. Recent preclinical work shows AAV‑mediated...
Pharvaris to File FDA NDA for Oral Deucrictibant, First-in-Class HAE Therapy
Pharvaris announced it will submit an FDA New Drug Application for its immediate‑release oral deucrictibant by the end of June. The filing is backed by Phase 3 RAPIDe‑3 data showing symptom relief in a mean of 1.3 hours, far faster than the...

ASCO to Spotlight Pancreatic Cancer Innovation Beyond KRAS
The page is BioCentury’s cookie policy, outlining five categories of cookies used on its website. Strictly necessary cookies support authentication and navigation, while functional cookies enable personalization. Marketing, advertising, and analytics cookies are used to tailor offers, display ads, and...

Fool.com Analysts Flag Abivax and Viking Therapeutics as Potential Double‑digit Gainers by 2030
Fool.com analysts project that Abivax (ABVX) and Viking Therapeutics (VKTX) could see their share prices double by 2030, driven by late‑stage trial readouts and expanding market opportunities. The forecast hinges on successful regulatory milestones for Abivax’s ulcerative colitis drug and...
NIR Fluorescence Surgery Enhances Oral Cancer Removal
Near‑infrared (NIR) fluorescence imaging is being integrated into oral cancer surgery to highlight malignant tissue that standard visual inspection can miss. A multi‑center trial of 120 patients demonstrated a 30% reduction in positive surgical margins and shaved roughly 12 minutes...

New Study Could Improve Testing and Treatment for Rare Brain, Spinal Cord, and Eye Cancers
Researchers at Fudan University identified hepatitis A virus cellular receptor 1 (HAVCR1) as a fluid‑based biomarker that distinguishes primary central nervous system lymphoma (PCNSL) and its eye‑only variant, primary vitreoretinal lymphoma (PVRL), from non‑cancerous conditions. In a study of 199 lymphoma...
Inocras Announces ASCO 2026 Online Publication: Whole-Genome HRD Phenotyping as a Predictor of PARP Inhibitor Benefit in First-Line Maintenance High-Grade...
Inocras announced that whole‑genome sequencing‑based HRD phenotyping predicts benefit from PARP inhibitor maintenance in high‑grade serous ovarian cancer. In a real‑world study of 84 patients at Severance Hospital, HRD‑positive tumors had a median progression‑free survival of 27.5 months versus 12.0...
New Indicator for Response to Therapy in Pediatric Cancers Identified
Researchers at the University of Birmingham reported that a high aneuploidy score can predict which children with relapsed solid tumors respond to a combined low‑dose irinotecan and PARP‑inhibitor regimen. The Phase I/II eSMART arm enrolled 70 patients across the UK, France,...

From Decades to Years - AI Could Speed Search for Brain Drugs Hiding in Plain Sight
Scientists at the UK Dementia Research Institute are using artificial intelligence to sift through patient data, voice recordings, eye scans and lab‑grown brain cells, aiming to repurpose existing drugs for neurological conditions such as motor neurone disease (MND). By training...

Abivax Presents First Quarter 2026 Financial Results and Reports Three-Year Interim Data From Study 108, a Phase 2a/2b Open-Label Extension...
Abivax reported Q1 2026 results, highlighting €491.6 million (≈$540 million) in cash and a runway extending into Q4 2027. The company released three‑year interim data from Study 108, showing that 68% of 130 ulcerative colitis patients remained in clinical remission after 144 weeks of obefazimod...
CODX Leads Biotech Rally with 55% Surge on Ebola Assay Breakthrough
Co-Diagnostics (CODX) surged 55.23% to $3.71 after announcing completion of an assay for the Bundibugyo Ebola virus, sparking a broader market rally that saw Sunshine Biopharma (SBFM) rise on generic Amoxicillin approval, GOVX highlight its Ebola strategy, and RegeneRx (RGNX)...
Galderma Receives U.S. FDA Approval for Differin® Epiduo® Acne Gel Prescription-to-OTC Switch
Galderma announced that the U.S. FDA has approved Differin Epiduo® Acne Gel for over‑the‑counter sale to consumers aged 12 and older. The switch moves a prescription‑strength combination of adapalene 0.1% and benzoyl peroxide 2.5% into the retail market, leveraging more than...

FDA Approves Hepcludex, First Treatment for Chronic Hepatitis D
The U.S. Food and Drug Administration has granted approval to Hepcludex (bulevirtide‑gmod), marking the first FDA‑cleared therapy for chronic hepatitis D in adults without cirrhosis or with compensated cirrhosis. Approval follows the phase 3 MYR301 trial, which showed a 48% combined response...

FDA Clears Gilead's Hepatitis D Drug, Four Years After Prior Rejection
Gilead’s hepatitis D therapy Hepcludex (bulevirtide) has finally secured FDA approval after a prior rejection over manufacturing and distribution flaws. The drug becomes the first U.S.‑cleared treatment for chronic hepatitis D, a rare but aggressive liver infection affecting roughly 70,000 Americans. Phase 3...

Enzymes Involved in Cholesterol Transport May Point to New Cancer Therapies
Scientists at Sanford Burnham Prebys and the University of Illinois Chicago identified phosphatidylinositol 5‑phosphate 4‑kinases (PI5P4Ks) as critical for cholesterol trafficking in TP53‑mutant cancers. In mouse models, deleting PI5P4K α and β prevented tumor formation by causing lysosomal cholesterol mislocalization...

Skape Bio Unlocks Generalizable GPCR Drugs Using AI Protein Design
Skape Bio, founded by former UW protein‑design researcher Chris Norn, has unveiled an AI‑driven platform that creates miniprotein therapeutics for G‑protein‑coupled receptors (GPCRs). A recent Nature paper shows functional miniproteins targeting 11 diverse GPCRs, including agonists validated on three receptors....

Human Gut Organoids with Functional Nerves Developed that Can Be Mass Produced
Researchers at Cincinnati Children’s Hospital and Nantes Université have unveiled a 3D‑printed scaffold system that accelerates the growth of human gut organoids to transplantation maturity in 14 days—half the previous timeline. The confined culture system yields centimeter‑scale intestinal, colon and stomach...
Avanzanite Bioscience’s Partner Agios Announces PYRUKYND® (Mitapivat) Approval in the European Union for Adults with Thalassaemia
Avanzanite Bioscience announced that the European Commission has granted marketing authorisation for PYRUKYND® (mitapivat) to treat anaemia in adults with transfusion‑dependent or non‑transfusion‑dependent alpha‑ or beta‑thalassaemia. The approval follows positive CHMP opinion and data from the Phase 3 ENERGIZE and ENERGIZE‑T...
PharmaEssentia to Present New Clinical Data Supporting the Use of Ropeginterferon Alfa-2b in Essential Thrombocythemia at ASCO and EHA 2026
PharmaEssentia will unveil new Phase 3 SURPASS‑ET data at ASCO and EHA 2026, highlighting that early initiation of ropeginterferon alfa‑2b yields markedly higher two‑year progression‑free survival (76.9% vs 43.1%) in high‑risk essential thrombocythemia (ET). Integrated analyses of SURPASS‑ET and Phase 2b EXCEED‑ET,...
Experimental mRNA Vaccine May Protect Against Multiple Ebola Viruses
Researchers have created an experimental mRNA vaccine that protects rodents from three orthoebolavirus strains, including the Bundibugyo virus driving the current outbreak in the Democratic Republic of the Congo and Uganda. The study shows the vaccine elicits robust antibody responses...
Expanded Label for Efgartigimod Offers Fast-Acting Treatment for gMG: James F. Howard, MD
On May 11, 2026, the FDA broadened the indication for efgartigimod, approving it for all adult patients with generalized myasthenia gravis, including AChR‑positive, MuSK‑positive, LRP4‑positive, and triple‑seronegative subtypes. The decision follows the phase‑3 ADAPT SERON trial, which enrolled 119 patients...
Team Finds Markers of Inflammatory Breast Cancer in Blood
University of Texas researchers have discovered blood‑based RNA biomarkers that reliably differentiate inflammatory breast cancer (IBC) from other breast cancer subtypes. Using the Thermostable Group II Intron Reverse Transcriptase (TGIRT) sequencing platform, they captured complex and fragmented RNAs missed by...

BioMarin's Voxzogo Shows Surprising Growth Boost in Hypochondroplasia Trial
BioMarin announced that its achondroplasia drug Voxzogo increased annualized growth velocity by 2.33 cm per year in a Phase 3 study of children with hypochondroplasia. The result exceeds performance seen in the approved indication and positions the company to file for a...

Researchers Successfully 3D Print Living Cornea
Researchers have successfully 3D‑printed a living cornea using decellularized donor tissue as a scaffold and stem cells to repopulate it. By applying extrusion shear forces, they aligned collagen fibers to replicate the natural architecture, achieving 90% cell viability and observable...

OSE Immunotherapeutics Reports Positive Phase 2 Data for Tedopi/Keytruda in Recurrent Ovarian Cancer
OSE Immunotherapeutics SA announced that its Phase 2 TEDOVA trial met its primary endpoint, showing the Tedopi‑Keytruda combination extended median progression‑free survival to 4.1 months versus 2.8 months with best supportive care in platinum‑sensitive recurrent ovarian cancer. The addition of Keytruda...

Semaglutide Drives Significant Fat Loss in Bariatric Non‑responders
For patients who did not respond well to bariatric surgery, semaglutide shifted weight loss far beyond placebo over 68 weeks. Most of that loss came from body fat rather than muscle. obesity
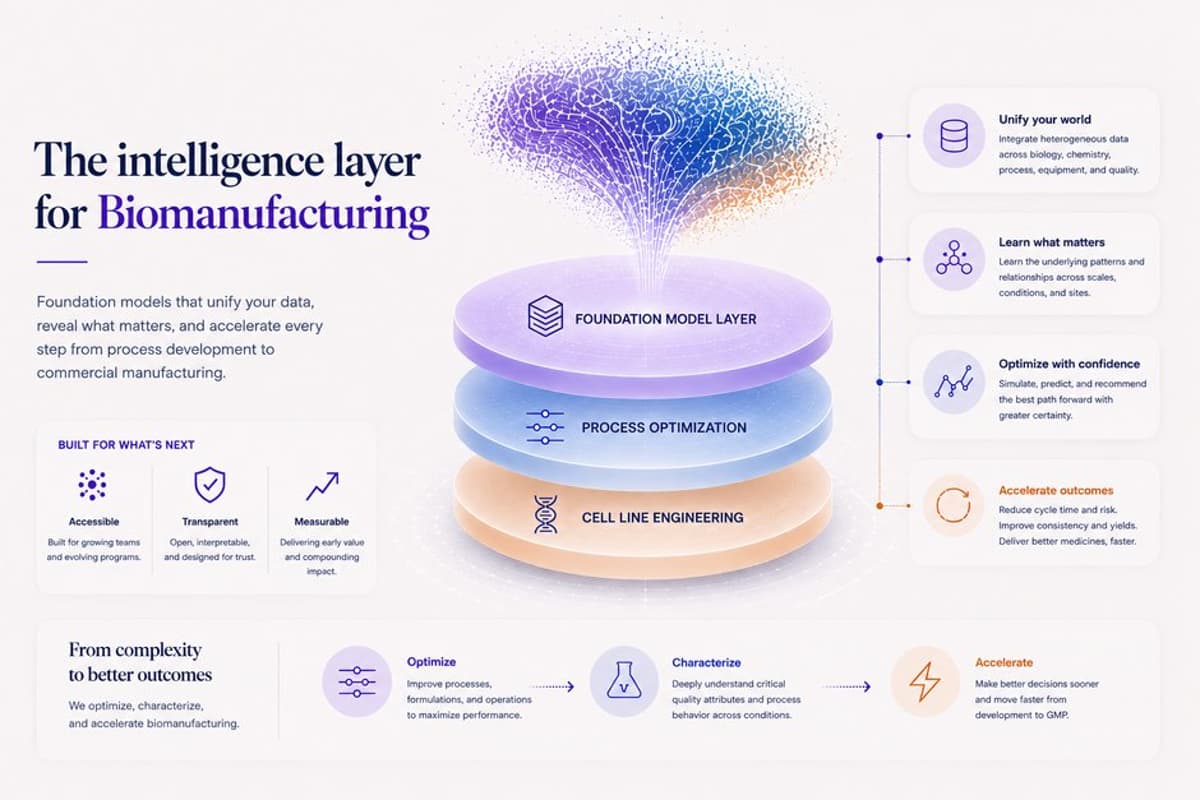
AI Models Slash Biomanufacturing Delays, Save $100M Monthly
Abinitio Bio is building the foundation models for biomanufacturing, turning 6-18 month process decisions into hours of compute and saving pharma $100M+ per month of delay on blockbusters. Congrats on the launch, @DanielMukasa1! https://t.co/r5nz5gVKPM https://t.co/HgPPRDs7Ew

First Generic Drug Approvals
The FDA’s Center for Drug Evaluation and Research approved 19 first‑generic drug applications between January and March 2026, spanning therapeutic areas such as oncology, infectious disease, psychiatry, renal care, and pain management. Notable entries include the first generic bortezomib injection...
ASCO26: 5 Data Snapshots Ahead of the Year’s Biggest Cancer Drug Meeting
Ahead of the ASCO Annual Meeting, several late‑stage oncology trials were previewed in newly released abstracts. Merck’s antibody‑drug conjugate sacituzumab tirumotecan (sac‑TMT) combined with Keytruda cut disease‑progression risk by 65% and achieved a 70% response rate in first‑line non‑small cell...

OSE’s Tedopi-Keytruda Combo Clears Phase II Ovarian Cancer Hurdle
French biotech OSE Immunotherapeutics announced positive Phase II data for its cancer vaccine Tedopi combined with Merck's Keytruda in platinum‑sensitive recurrent ovarian cancer. The combination improved median progression‑free survival to 4.1 months versus 2.8 months with best supportive care, cutting the risk of...

Focus on Biosimilar Adoption: Driving Uptake Through Strategy
Teva’s U.S. Value & Access team has moved beyond simple payer formulary placement, adopting a holistic ecosystem approach that coordinates payers, providers, pharmacies, and health systems to accelerate biosimilar uptake. By redesigning contracts to include step‑therapy, lower patient cost‑sharing, and...
What Is Immunotherapy and How Does It Treat Cancer and Other Conditions?
Immunotherapy research has surged, with clinical trials climbing from 1,257 between 2006‑2016 to 4,591 in the past decade. Cancer treatments dominate the field, featuring checkpoint inhibitors, CAR‑T cells, and mRNA‑based vaccines now approved for more than 30 tumor types. Researchers...

Cognitive Effects Vary by Therapy for Advanced Prostate Cancer
A phase‑2 ARACOG trial presented at ASCO showed that men with advanced prostate cancer receiving darolutamide experienced significantly less cognitive decline over 24 weeks than those on enzalutamide. The study enrolled 111 patients (median age 71) and evaluated five computer‑based neurocognitive tests,...

Texas A&M Nasal Spray Reverses Brain Aging Markers in Mice
Texas A&M scientists unveiled a nasal spray packed with extracellular vesicles that, in mouse trials, erased neuroinflammation and restored mitochondrial function, leading to measurable cognitive gains. The two‑dose regimen produced effects within weeks that lasted months, positioning the therapy as...
FDA Clears FoundationOne CDx as Companion Diagnostic for Tepotinib in MET‑Exon‑14 NSCLC
The U.S. FDA approved Foundation Medicine’s FoundationOne CDx test as a companion diagnostic for tepotinib (TEPMETKO) in MET exon‑14‑skipping non‑small cell lung cancer, enabling broader patient identification for the targeted therapy. The clearance leverages real‑world data from over 150,000 patients...

Quality Control: The Key to Stunning Plots
1/ You want beautiful volcano plots. Striking heatmaps. Big discoveries. But before the analysis, comes one boring step you can't skip: QC. It’ll save your science. 🧵 https://t.co/PBnehqINrR

Black Diamond Shares Tumble 6% Despite 15‑month PFS for Silevertinib in 1L NSCLC
Black Diamond Therapeutics saw its shares drop 6.2% to $3.33 after announcing that its Phase 2 trial of Silevertinib delivered a median progression‑free survival of 15.2 months in first‑line EGFR‑non‑classical NSCLC. The market reaction highlights investors’ skepticism that the data will...
NervGen Secures $60 Million in Share‑Warrant Offering to Push NVG‑291 Forward
NervGen Pharma Corp priced a public offering of 24 million common shares and matching warrants at $2.50 per unit, generating roughly $60 million in gross proceeds. The cash will be used to advance the company’s lead candidate NVG‑291 for spinal‑cord injury, while...

The NMPA Grants Conditional Approval to Boehringer Ingelheim’s Hernexeos for 1L HER2-Mutant NSCLC
China's National Medical Products Administration granted conditional approval to Boehringer Ingelheim’s oral HER2‑mutant NSCLC drug Hernexeos (zongertinib). The decision is based on a Phase Ib trial of 74 treatment‑naïve patients that delivered a 75.7% overall response rate and a median...

LSX Nordic Congress 2026: What Came Out of the Biotech Partnering Event
At the LSX Nordic Congress 2026 in Copenhagen, life‑science leaders emphasized that Nordic biotech firms must look beyond their small domestic markets to achieve growth. Executives highlighted a scarcity of risk‑willing capital, especially for Series A and B rounds, prompting...

Biogen, Denali to Drop Drug in Non-Genetic Parkinson’s After Mid-Stage Study Flop
Biogen and Denali Therapeutics announced that their LRRK2 inhibitor BIIB122 failed to meet primary endpoints in a Phase 2b trial of early‑stage Parkinson’s disease. The mid‑stage study, which targeted patients without the LRRK2 genetic mutation, showed no statistically significant slowing...

AI‑Designed Miniproteins Toggle GPCRs, Matching Drugs in Mice
Scientists at the University of Washington’s Institute for Protein Design and biotech Skape Bio used artificial intelligence to engineer miniproteins that can activate or block G protein‑coupled receptors. In mouse experiments the engineered proteins performed on par with an approved...
Moderna’s Once-Rebuffed mRNA Flu Shot to Face Scrutiny From FDA Adcomm
The FDA will hold its Vaccines and Related Biological Products Advisory Committee on June 18 to evaluate Moderna’s investigational mRNA flu vaccine, MFLUSIVA, aimed at adults 50 and older. Earlier this year the agency issued a refusal‑to‑file letter, arguing the...

Eli Lilly’s Retatrutide Delivers up to 28% Weight Loss in Phase 3 Trial
Eli Lilly announced that its experimental triple‑agonist retatrutide achieved up to 28.3% weight loss (about 70 lb) over 80 weeks in a pivotal Phase 3 trial, with 45% of participants shedding 30% or more. The data positions the drug as a potential game‑changer...

Tempus AI Launches ArteraAI Prostate Test for Metastatic Prostate Cancer
Tempus AI announced the clinical launch of the ArteraAI Prostate Test, a CLIA‑certified and CAP‑accredited prognostic assay for metastatic hormone‑sensitive prostate cancer (mHSPC). The test combines a digital pathology AI algorithm with Tempus’ next‑generation sequencing data to generate personalized risk...
Biogen, Denali Pull BIIB122 After Phase 2b LUMA Fails to Slow Parkinson's
Biogen and Denali Therapeutics announced they are halting development of BIIB122, a LRRK2 inhibitor for Parkinson's disease, after the Phase 2b LUMA trial failed to meet its primary and secondary endpoints. The setback removes a key candidate from both companies'...
Cyclarity Therapeutics Reports Safety Data for 7-Ketocholesterol Clearance
Cyclarity Therapeutics reported first clinical evidence that its AI‑engineered cyclodextrin drug, UDP‑003, can safely bind and promote urinary excretion of 7‑ketocholesterol, a toxic oxysterol linked to atherosclerosis. The Phase 1 safety trial demonstrated favorable pharmacokinetics and pharmacodynamics, with no serious adverse...

Particle‑by‑Particle Tracking Uncovers Heterogeneous Drug Release in PLGA Nanocarriers
A research team led by the Institute of Materials Science of Barcelona used single‑particle super‑resolution microscopy to follow drug release from PLGA nanocarriers over 30 days. The study showed that some particles dump their cargo within hours while others retain...
Akari Therapeutics Shares Double on KRAS Synergy Data, Raises $5.5M for ADC Trial
Akari Therapeutics' shares surged more than 100% in pre‑market trading after the company disclosed preclinical data that its lead ADC, AKTX-101, works synergistically with KRAS inhibitor Adagrasib in pancreatic cancer models. The biotech also priced a $5.5 million private placement to...