
Entrada Crashes as Duchenne Therapy Comes in ‘Below Expectations’ in Early Study
Entrada Therapeutics reported that its investigational oligonucleotide ENTR‑601‑44 raised dystrophin levels by 2.36% in the first cohort of its Phase 1/2 ELEVATE‑44‑201 trial, far below the company’s double‑digit target. The modest protein increase triggered a 50% plunge in the Boston‑based biotech’s share price, trading around $8 per share. Analysts highlighted a more promising 0.08 improvement in time‑to‑rise velocity and a clean safety profile, which could support dose escalation to 12‑18 mg/kg. The next cohort will test a higher dose with results expected by year‑end.

SGLT2 Inhibitors Cut Cardiovascular Death by 14%
As a medical school professor: when I trained, "diabetes drugs" meant blood-sugar drugs. That ceiling has been broken. Meta-analysis in Am J Med (Jaiswal et al, U Chicago, Mar 26 2026) pooled 18 RCTs, n=95,913 patients with diabetes, heart failure, or...

Podcast: Autonomous Labs Redefine the Role of Biopharma Researchers
Autonomous laboratories, integrating robotic hardware with AI-driven decision making, are emerging as a transformative force in biopharma R&D. In a GlobalData Media podcast, Frankie Fattorini interviewed Jason Kelly, CEO of Ginkgo Bioworks, who described how these labs can conduct experiments with unprecedented precision...
Studying These Young Alzheimer's Patients Led to Breakthroughs. Trump Cut the Funding
The Dominantly Inherited Alzheimer Network (DIAN) has leveraged over 200 families with rare early‑onset Alzheimer’s gene mutations to uncover how the disease begins and to test amyloid‑targeting drugs that later reached the market. Its international registry, funded by the NIH...

STAT+: FDA Revisits a Rare Cancer Treatment It Rejected a Few Months Ago
The FDA has announced it will re‑evaluate a rare‑cancer therapy it dismissed just months earlier, citing new data submitted by the drug’s sponsor. The treatment, aimed at a subtype of metastatic sarcoma, originally failed to meet the agency’s efficacy benchmarks...

Amylyx Pharmaceuticals Q1 Earnings Call Highlights
Amylyx Pharmaceuticals reported that the Phase 3 LUCIDITY trial of its GLP‑1 antagonist avexitide completed dosing and is on track for a top‑line readout in Q3 2026. The company launched a U.S. expanded access program to treat up to 250 adults with...

10‑Marker Bioage Score Outperforms Chronological Age
As a medical school professor: chronological age is a tax bracket. Biological age is the actual bill. Aging Cell paper from MARK-AGE (Moreno-Villanueva, Burkle et al, U Konstanz, 2026) screened 362 biomarkers in ~3,300 adults across 8 European countries, then distilled...

FDA Launches Real‑Time Clinical Trials to Accelerate Drug Development
The U.S. Food and Drug Administration announced the start of two real‑time clinical trials—one for mantle‑cell lymphoma and another for small‑cell lung cancer—and issued a request for information on a broader pilot program slated for summer 2026. The move aims...

Atara, Pierre Fabre's Cell Therapy to Get Another Shot at FDA Approval
Atara Biotherapeutics and Pierre Fabre Pharmaceuticals are reviving a T‑cell therapy that was rejected twice by the FDA. Regulators have signaled willingness to base a new approval decision on data from a Phase 3 trial, a departure from the earlier requirement...
UK NHS Rolls Out One‑minute Injectable Cancer Therapy, Slashing Visits for 14,000 Patients
The UK National Health Service is replacing two‑hour intravenous pembrolizumab infusions with a one‑minute subcutaneous injection for roughly 14,000 cancer patients each year. The change cuts appointment time by up to 90 % and promises to free clinical capacity while improving...

Wolfe Research Says Market Reaction on Eli Lilly (LLY) Foundayo Report Appears Overdone
Wolfe Research says the market overreacted to an FDA adverse‑event report on Eli Lilly’s GLP‑1 drug Foundayo. Lilly determined the serious hepatic failure case was not reasonably related to the product. Shares fell about 3% in pre‑market trading, yet analysts kept...

‘People Should Be Talking About It’: Moves to Curtail Vaccine Information Obscures Important Science, Doctors Say
U.S. health agencies, including the FDA, CDC and NIH, have halted or censored several vaccine studies and presentations in 2026, citing methodological concerns or political language guidelines. A leaked Covid‑19 booster effectiveness study, pulled at the last minute, still shows...

Vertex Secures German Reimbursement for CASGEVY Gene Therapy
Vertex Pharmaceuticals announced a reimbursement agreement with Germany's GKV‑Spitzenverband, granting sustainable access to its CRISPR/Cas9 therapy CASGEVY for patients 12 years and older with sickle cell disease or transfusion‑dependent beta thalassemia. The deal makes Germany the latest market to fund...
High Hs‑CRP, Not LDL, Drives Statin Benefit
"But my cholesterol is normal." JUPITER trial (NEJM): 18,000 people, normal LDL, high hs-CRP. Statin → cardiac events ↓44%, heart attacks ↓54%, strokes ↓48%. 322,000-person UK Biobank: hs-CRP outranked LDL. Full episode 🎙 https://youtu.be/FBLB1CQGBPM

The Rise of Trispecific Antibodies: Biopharma’s Next Big Bet After Bispecifics
Trispecific antibodies are emerging as the next wave of multispecific therapeutics, extending the success of bispecifics by simultaneously engaging three targets. More than 100 candidates are now in clinical trials, with major players such as Pfizer, Sanofi, AbbVie and Johnson...

Treatment-Resistant IBD May Benefit From New Combo Antibody Therapy
Phase 2b DUET‑Crohn’s and DUET‑UC trials, funded by Johnson & Johnson, tested the fixed‑dose co‑antibody JNJ‑4804 (guselkumab + golimumab) in patients whose IBD had failed prior advanced therapies. In ulcerative colitis, JNJ‑4804 matched guselkumab’s efficacy and outperformed golimumab, while in Crohn’s disease the highest dose...

Longevity Genes Can Switch From Benefit to Harm
As a medical school professor, I tell students: be careful with "longevity gene." Prof. Nikolai Slavov (Northeastern) just argued that single "longevity genes" are oversimplified. The same gene's effect on lifespan can flip sign with age -- helpful early, harmful later....

Invasive Nanowire BCIs Needed, Non‑Invasive for Biomarkers
I love brain-to-computer interfaces and one of my first papers and granted patents was in AI/ML for BCI. And I was very much impressed with BrainCo's non-invasive BCIs and artificial limbs and sensing fingers (that they sell to Tesla btw)....

Bayer Reports P-III (REVEAL) Trial Data on Iodine 124 Evuzamitide to Diagnose Cardiac Amyloidosis
Bayer announced that its investigational PET/CT radiotracer I‑124 evuzamitide met the primary sensitivity and specificity endpoints in the Phase III REVEAL trial of 170 adults with suspected cardiac amyloidosis. The study compared the tracer to standard clinical diagnosis and achieved the...

Choosing a Reference Genome Stalls Bioinformatics Before Coding
Bioinformatics is hard before you even write a single line of code. Here's why. 1/ You haven’t started your DNA-seq analysis. You haven’t aligned a read. And yet you’ve already hit a wall. Which human genome to use? https://t.co/nAj5MOqEFm

Simple Protein Redesign Produces the Most Active Designed Enzyme Ever
Researchers at UCSF combined crystallographic fragment screening with directed evolution to repurpose a simple designed protein, ABLE, into two distinct functional proteins. One of the new proteins, KABLE, is a Kemp eliminase that exhibits ten‑fold higher activity than any previously...

Sarepta Says to ‘Exercise Prudence’ in Setting Expectations for Elevidys’ Recovery
Sarepta Therapeutics urged analysts to "exercise prudence" as it navigates a turbulent recovery after three patient deaths linked to its Duchenne muscular dystrophy gene therapy Elevidys. The FDA imposed a boxed warning and limited the drug to ambulatory patients aged...

Silence Therapeutics Highlights Recent Business Achievements and Reports First Quarter 2026 Financial Results
Silence Therapeutics reported Q1 2026 results, ending the quarter with $70.1 million in cash and a net loss of $15 million, a sharp improvement from the $28.5 million loss a year earlier. The company highlighted progress on its lead siRNA candidate divesiran, with Phase 2...

Unlocking Lithium’s Hidden Effects on Alzheimer’s Disease at the Cellular Level
A University of Eastern Finland team mapped lithium chloride’s cellular actions in Alzheimer’s models, showing it reduces Tau hyperphosphorylation at several key sites and reshapes kinase and Rho GTPase signaling. Phosphoproteomic analysis revealed lithium’s impact extends beyond the primary GSK‑3β...

RESEARCH: NITAZOXANIDE in Head and Neck Squamous Cell Carcinoma - 2025 Paper From China
A 2025 Chinese study published in Springer Nature demonstrates that nitazoxanide, an FDA‑approved antiparasitic, exhibits potent anti‑tumor activity against head and neck squamous cell carcinoma (HNSCC). Using integrated single‑cell RNA sequencing and spatial transcriptomics, researchers identified that the drug down‑regulates...

Six-Month Trial Confirms Safety of Previously Uncharacterized Probiotic Strain
A six‑month, double‑blind trial involving 152 healthy adults found that daily consumption of Lactiplantibacillus plantarum K014 (≥1 × 10⁹ CFU) was safe, with blood counts, glucose, lipid, liver and kidney markers remaining within normal ranges. No adverse events were reported, and exploratory analyses suggested...

Sanofi Pulls Teplizumab From FDA’s Makary Review Over Political Interference
Sanofi has formally requested that the FDA withdraw its type‑1 diabetes therapy teplizumab from Commissioner Marty Makary’s accelerated review pathway after an acting CDER director, a political appointee, stepped into the scientific review. The move follows a missed April 21 decision...

Mayo Clinic and Stanford Launch First Blood Test to Map Tumor Microenvironment
Mayo Clinic and Stanford Medicine announced a new blood test that maps the tumor microenvironment, enabling more accurate prediction of immunotherapy response across 17 cancer types. The test uses spatial transcriptomics and AI to identify nine cellular neighborhoods from cell‑free...

NIH-Funded Study Suggests that Testosterone Suppresses Brain Tumor Growth in Males
A NIH‑funded study by Cleveland Clinic researchers found that loss of male hormones, especially testosterone, accelerates glioblastoma growth in mouse models by triggering inflammation and the hypothalamus‑pituitary‑adrenal (HPA) stress axis. Supplemental testosterone was associated with a 38% lower risk of...

Magic Mushroom Compound Shows Promise Against Cocaine Addiction
A randomized, double‑blind trial of psilocybin in 40 cocaine‑dependent adults, published in JAMA Network Open, found that 30% of participants receiving a single dose were completely abstinent after 180 days, compared with none in the placebo arm, and remaining users...
STAT+: Next-Gen Duchenne Drug From Entrada Disappoints
Entrada Therapeutics reported that its next‑generation exon‑skipping drug for Duchenne muscular dystrophy failed to achieve its primary efficacy endpoints in an early‑stage trial. The study showed only a modest rise in dystrophin levels, far below the thresholds set by the...

Entrada Shares Sink After Duchenne Drug Falls Short of Expectations
Entrada Therapeutics reported that its experimental Duchenne drug ENTR-601-44 generated only a 2.36‑point increase in dystrophin, far below the 10‑11% rise analysts had modeled. The shortfall sent the company’s shares tumbling more than 50% in pre‑market trading. By contrast, Avidity...

Catalyst Poised as Florida’s First Multi‑Billion Biotech Exit
Is Catalyst (based in Coral Gables) the first Florida biotech to have a multi billion exit?
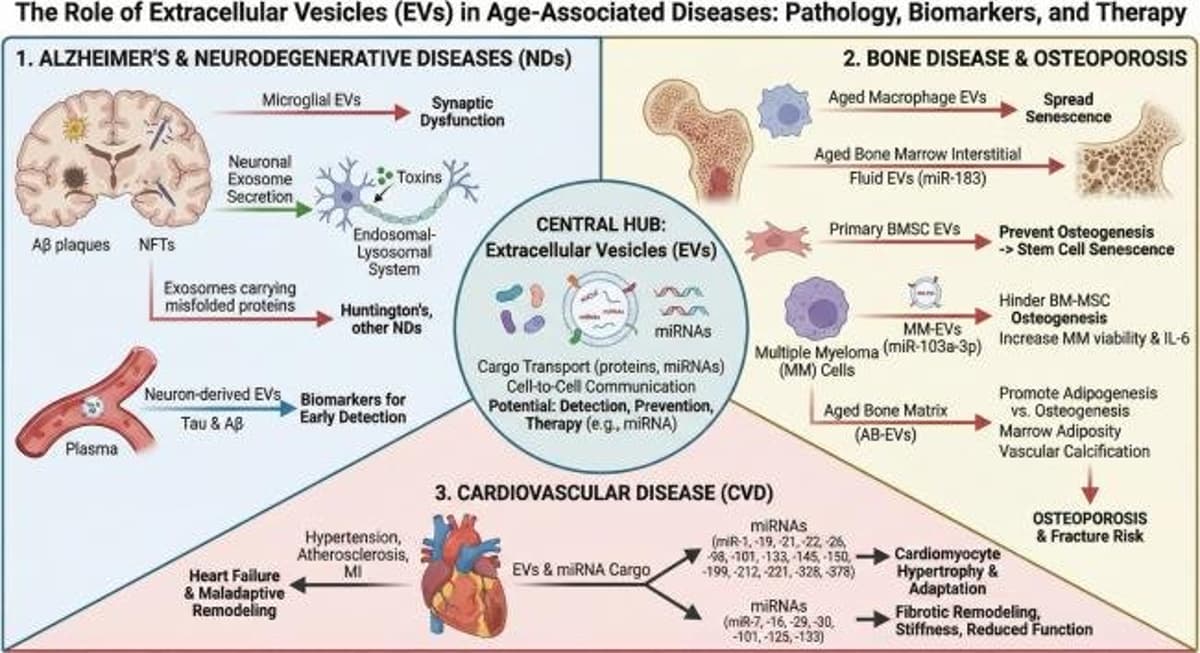
Extracellular Vesicles Deliver SASP, Fuel Aging, Offer Therapies
Extracellular Vesicles as Key SASP Carriers Driving Cellular Senescence, Inflammaging, and Therapeutic Opportunities in Aging and Age-Related Diseases https://t.co/SURS5ZbTD9 https://t.co/IXM2BkdurZ

Roche to Acquire PathAI for ~$1.05B
Roche has signed a definitive agreement to acquire AI‑driven diagnostics firm PathAI for an estimated $1.05 billion, comprising $750 million upfront and up to $300 million in milestones. The deal, slated to close in the second half of 2026, builds on a partnership...

Reviewing the Role of Advanced Glycation Endproducts in Aging and Age-Related Disease
Advanced glycation endproducts (AGEs) are protein‑sugar adducts that accumulate with age, altering protein structure and activating the RAGE receptor to drive chronic inflammation. The review highlights how AGEs cross‑link collagen, stiffening the extracellular matrix and contributing to diabetes, cardiovascular disease,...

Targeted Therapies Resurge; some Phase 3 Trials Risk Obsolescence
With a rapid fire raft of trial updates coming out of #esmobreast26, one thing stood out... targeted molecules are back in fashion and a few companies might be left holding the baby if their phase 3 trials have already started: https://t.co/jlKDLsecVZ...

The Goalposts Shifted in Berlin, Your Trial Timeline Didn’t
The European Society for Medical Oncology (ESMO) Breast 2026 conference in Berlin showcased a wave of late‑stage breast cancer data that is reshaping therapeutic benchmarks. Several trials reported higher pathologic complete response rates and novel biomarker‑driven combinations, tightening the competitive...

Scientists Discover Why Ozempic Works Better for some People
GLP‑1 drugs like Ozempic improve blood sugar and weight loss, but response varies. A Japanese study of 92 type‑2 diabetes patients found that those who overeat in response to external cues (appearance or smell) achieved greater weight loss and glucose...
More Evidence for Muscle Stem Cell Activity to Be Inhibited by the Aged Tissue Environment
Researchers discovered that the extracellular matrix (ECM) of aged mice suppresses the growth of both young and rejuvenated muscle stem cells (MuSCs). Elevated collagen levels in the aged ECM create a non‑autonomous barrier that limits stem‑cell proliferation, even when intrinsic...

Encoded Therapeutics Doses First Patient in Pivotal ETX101 Trial for Dravet Syndrome
Encoded Therapeutics has dosed the first patient in the pivotal ENDEAVOR Part 2 study of its AAV9‑based gene regulation therapy ETX101 for SCN1A‑positive Dravet syndrome, while also opening an expansion cohort for older children. The move advances the program into late‑stage...

Pharma Pulse: GLP-1 Momentum Builds While Lilly Expands Genetic Medicine Manufacturing
Eli Lilly opened its first dedicated genetic‑medicine manufacturing plant in Lebanon, Indiana, expanding U.S. capacity for advanced gene‑editing and RNA therapies. GLP‑1 drugs accounted for eight percent of all prescriptions filled in March 2026, highlighting their growing role beyond diabetes. Bayer...

The Vaccine Safety Signal the Media Still Won’t Read
A peer‑reviewed study published in Vaccine (Sept 2022) re‑analyzed Pfizer and Moderna mRNA COVID‑19 trial data and found excess serious adverse events—10.1 per 10,000 for Pfizer and 15.1 per 10,000 for Moderna—yielding harm‑to‑benefit ratios of roughly 4.4 : 1 and 2.4 : 1 respectively. The...

What’s Next for IVF
Advances in IVF are moving beyond traditional lab techniques toward AI, robotics, and novel embryo‑delivery devices. Researchers at the Carlos Simon Foundation have built a “Transfer Direct” system that injects embryos into the uterine lining, while AI platforms such as...

What Impact Could Protein Stabilisation Have for Cancer Patients?
In this episode of the DDW Podcast, CEO Anthony Johnson and CSO Matthew Fyfe discuss OutRun Therapeutics' novel protein‑stabilisation platform, which inhibits specific E3 ubiquitin ligases to prevent degradation of disease‑relevant proteins. Their lead program targets the E6AP ligase to...

Halozyme and Oruka Sign Agreement for Hypercon Technology
Halozyme’s Hypercon technology has entered an exclusive global licensing agreement with Oruka Therapeutics to develop ORKA-001, a biologic targeting the IL‑23p19 subunit for psoriasis and related inflammatory diseases. The deal provides Halozyme with an upfront cash payment, future milestone fees,...

Global Drugmakers Eye More China Biotech Deals After Record Year
Global pharmaceutical firms are intensifying their pursuit of Chinese biotech partners after 2025 set a record $135.7 billion in out‑licensing deals, nearly three times the 2024 total. Executives from Johnson & Johnson and Merck highlighted plans to expand on‑the‑ground resources and...

Polynucleotide Implants Promise Hair Regrowth for Complete Baldness
Looks like polynucleotide implants can save even a fully bald head by @IntEngineering #Tech #TechForGood #EmergingTech https://t.co/CtDKGSBbcb
Fibrosis, IPF and the Search for Better Therapies
The BioSpace "Denatured" podcast episode spotlights the persistent challenges of idiopathic pulmonary fibrosis (IPF) and broader fibrotic diseases. Host Jennifer C. Smith‑Parker interviews Georg Vo Beiske, CEO of Tribune Therapeutics, and Jonas Hallén, CMO of Calluna Pharma, to dissect current treatment gaps and...
Macrophage‐Inspired Nanomedicines: Harnessing Innate Biology for Precision Therapy
The review outlines macrophage‑inspired nanomedicines that exploit three delivery platforms—whole macrophage carriers, macrophage‑membrane‑coated nanoparticles, and macrophage‑derived exosomes. By leveraging the cells’ natural homing, barrier‑penetrating and immunomodulatory traits, these systems aim to improve targeting of cancers, atherosclerosis and other inflammatory diseases....