
Is Longevity a $1.2 Quadrillion Opportunity?
Peter Diamandis released the 2026 Longevity Metatrend Report, a free 200‑page analysis of the rapidly advancing health‑span sector. The report highlights breakthroughs such as human trials of partial epigenetic reprogramming, AI‑engineered proteins achieving 50‑fold efficacy gains, and the first pig‑organ transplants into patients. It also notes wearable health platforms surpassing $10 billion valuations and a surge of capital, with companies raising between $280 million and $1 billion. ARK Invest estimates the total addressable market for longevity interventions at $1.2 quadrillion.

Genomic Evidence Confirms Natural Evolution (Variance) of Andes Hantavirus
A Swiss passenger infected on the MV Hondius cruise ship was found to carry an Andes hantavirus strain that is a direct descendant of a 2018 Argentine case. Whole‑genome sequencing revealed 98.7‑99% identity to the 2018 isolate and a mutation...

CEL-SCI Enters Strategic Agreement with Amarox for the Registration, Commercialization, and Distribution of Multikine in Saudi Arabia
CEL‑SCI Corp. has signed a strategic partnership with Saudi firm Amarox to register, commercialize, and distribute its immunotherapy Multikine in Saudi Arabia, with a 50/50 revenue‑sharing model. Amarox will act as the local regulatory representative, seeking the Saudi Food and...

OM1 Supports 650,000 Patient Real-World Regulatory Submission for FDA Approval of Hologic’s Aptima HPV Assay
OM1’s AI‑driven real‑world evidence platform underpinned a landmark FDA submission that cleared Hologic’s Aptima HPV assay for primary cervical cancer screening. The study aggregated data from more than 650,000 women across U.S. health systems, automating extraction from electronic health records...
Avenacy Announces Launch of Ready-to-Use Tranexamic Acid in 0.7% Sodium Chloride Injection in the U.S. Market
Avenacy, a specialty injectable drug company, has launched a ready‑to‑use Tranexamic Acid 0.7% Sodium Chloride Injection in the United States. The product mirrors the active ingredient of Pfizer’s CYKLOKAPRON® and is indicated for short‑term use in hemophilia patients undergoing tooth...
HistoSonics Moves to Advance Additional Histotripsy Applications Announcing FDA Submission for Kidney Tumors
HistoSonics has filed a De Novo request with the FDA to expand its Edison® Histotripsy System from liver to kidney tumor treatment. The submission is backed by the ongoing HOPE4KIDNEY trial, which has enrolled 67 patients and shows precise, non‑invasive tumor...
Therorna to Showcase Clinical-Ready Circular RNA in Vivo CAR-T and CircRNA Pipeline at the 2026 American Society of Gene &...
Therorna Inc. will present three posters at the 2026 ASGCT meeting, highlighting its lead in‑vivo CAR‑T candidate TI‑0032, a circRNA‑encoded CD19×CD3 T‑cell engager, and the HPV16 therapeutic vaccine TI‑0093. TI‑0032 has just entered a first‑in‑human investigator‑initiated trial for refractory autoimmune...
Researchers Develop Body-Compatible Dermal Electrode
Researchers at POSTECH have created a dermal bioelectrode that inserts like a microneedle but becomes soft in the dermis, eliminating immune response. The electrode’s effervescent sacrificial layer enables rapid penetration and then transforms to a flexible structure, delivering stable biosignal...

Late Line RCC: Where Darlifarnib Fits and Why LITESPARK-012 Matters
At the International Kidney Cancer Symposium, Kura presented phase 1 data showing its next‑generation farnesyl transferase inhibitor darlifarnib combined with cabozantinib achieved a 44% objective response rate in clear cell renal cell carcinoma patients previously treated with cabozantinib. The cohort was...

Noncovalent Fragments vs WRN
Researchers at Merck and Proteros reported a noncovalent fragment‑based campaign against the Werner syndrome helicase (WRN), a synthetic‑lethal cancer target. Using a 1,020‑compound fluorine‑fragment library screened by 19F‑NMR and a separate 500‑compound SPR screen, they identified seven primary hits, three...
Evidence for Sleep Apnea to Accelerate Vascular Aging via Increased Cellular Senescence
Researchers modeled obstructive sleep apnea by exposing C57BL/6J mice to intermittent hypoxia. The exposure rapidly increased epigenetic age acceleration and p16‑positive senescent cells in vascular tissue. Mice developed higher systolic and diastolic pressure and endothelial dysfunction. Systemic removal of p16‑expressing...

ISS Expedition 74 Crew Conducts DNA Nano-Therapy and Space Agriculture Research
On Thursday, Expedition 74 crew members performed a suite of high‑impact experiments aboard the ISS. NASA engineer Jessica Meir used a spectrophotometer to study DNA‑like nanomaterials, data that could accelerate cancer‑targeting nano‑therapies. ESA’s Sophie Adenot tended alfalfa in the Veggie unit, probing...

Seeds of Power: China Turns to Genetic Engineering to Become Global Superpower
China is intensifying control over seed genetics, using hybrid breeding and GM technologies to narrow yield gaps with the United States and cut import dependence on corn and soybeans. The global seed market is dominated by Bayer, Corteva, Syngenta and...

When Superbugs Threaten Vulnerable Children: Can AI Help Solve Antibiotic Resistance?
A wave of drug‑resistant bloodstream infections is killing newborns in Southeast Asia, highlighting the accelerating global antibiotic resistance crisis. The World Health Organization warns that the pipeline for new antibiotics is dangerously thin, leaving clinicians with few treatment options. MIT...

The Adiponectin Paradox: Fat’s Secret Longevity Signal or a Bio-Marker of Decline?
Adiponectin, a hormone secreted by fat cells, is celebrated for its anti‑inflammatory and insulin‑sensitizing effects, yet epidemiological data reveal a paradox: while centenarians exhibit high levels, elevated adiponectin in most older adults correlates with higher mortality and frailty. Researchers attribute...

Gene Editing Has the Science Figured Out and Now Needs an Entire Stack of New Business Models, Reimbursement Mechanics, and...
Gene editing has moved from scientific proof‑of‑concept to commercial reality, highlighted by Vertex's CASGEVY therapy earning $43 million in Q1 2026 and treating over 500 patients at a $2.2 million list price. While the science is solid, the therapy’s multi‑step delivery—spanning screening, cell...

Is Novo Nordisk Turning the Page on CagriSema?
Novo Nordisk says its launch timeline for the dual‑agonist CagriSema remains unchanged despite scrapping a single‑chamber delivery device. The Phase 3 trial showed a 23% average weight loss over 84 weeks, impressive but still below Lilly’s tirzepatide at 25.5%. Meanwhile, Novo...

Weekly Reads: Lab-Grown Sperm, Stem Cell Pills, Intranasal EVs, EMF Cell Paper Doubts, Makary Toast?
A Wired report says Paterna Biosciences has produced human sperm in the lab using stem‑cell techniques, suggesting in vitro gametogenesis could soon enable fully lab‑derived embryos. At the same time, a South Korean study claiming magnetic control of genes is under...

Drug Development Funnel: What I Learnt Building One From Scratch
A new D3.js funnel visualizes drug‑development attrition, showing that the headline billions‑dollar cost reflects cumulative failures rather than a single success. The plot reveals that pre‑clinical spending (~$430 M per approved drug) rivals the cost of late‑stage trials, and that only...

TriSalus (TLSI) Initiates Patient Enrollment for PREDICTT Liver Tumor Clinical Trial
TriSalus Life Sciences announced on May 4 that patient enrollment has begun for the PREDICTT trial, a prospective study of its Pressure‑Enabled Drug Delivery (PEDD) platform in liver tumors. The investigator‑led trial at MD Anderson Cancer Center will enroll about 20...
Here Is Why Ovid Therapeutics (OVID) Is One of the Best Fast Growing Penny Stocks
Ovid Therapeutics saw its price target raised by H.C. Wainwright to $4 from $2, reflecting confidence in its advancing pipeline. The company reported clean safety data for a 7 mg dose of its GABA‑aminotransferase inhibitor OV329, with no serious adverse events....
Lexicon (LXRX) Reports $21.1M Q1 Revenue Driven by Novo Nordisk Milestones
Lexicon Pharmaceuticals reported Q1 2026 revenue of $21.1 million, up sharply from $1.3 million a year earlier, largely due to $20 million in development milestones from its LX9851 partnership with Novo Nordisk. Net loss narrowed dramatically to $1.0 million from $25.3 million in Q1 2025, aided...
AtaiBeckley (ATAI) Reports Positive Phase 2a Results for Social Anxiety Treatment EMP-01
AtaiBeckley announced expanded Phase 2a results for EMP‑01, an oral R‑MDMA formulation targeting Social Anxiety Disorder. The study showed a 38% reduction in patient‑reported symptoms and a 32% drop in real‑world avoidance behaviors by Day 43, with 49% of participants classified as...
Glucose Nanoparticles Help CBD Cross the Blood-Brain Barrier
Researchers have engineered glucose‑coated polymer nanoparticles that dramatically improve cannabidiol (CBD) delivery across the blood‑brain barrier. The particles use a PEG‑PHB core to solubilize CBD and a surface glucose layer to hijack GLUT‑1 transport, while reactive‑oxygen‑species triggers release in inflamed...

17-Alpha Estradiol - Another Top Anti-Aging Drug
The post details efforts to source 17α‑estradiol (alfatradiol) for anti‑aging and renal‑protective research, listing vendors and pricing—including Octagonchem’s $1,200 for 100 g and Shanghai Jizhi’s roughly $80 for 200 mg (≈¥568). It proposes a group‑buy workflow that starts with small‑scale sampling, purity...

The $1 Trillion Cardiovascular Reset
The blog highlights Eli Lilly's $1.3 billion acquisition of Verve Therapeutics, targeting a one‑shot PCSK9 gene‑editing therapy that could replace daily statins. It underscores the massive adherence problem—up to 75% of statin users stop within two years—and the untapped 1.4 billion‑person market for...

Pharmaceutical Executive Daily: FDA Extends Review of Leqembi's sBLA
The FDA has extended the priority review of Eli Lilly's subcutaneous Leqembi by three months, delaying the agency's action date while requesting additional information. The once‑weekly injection is designed to replace the current infusion regimen for patients with early Alzheimer’s...
Electromagnetic Field Activation of Gene Therapy as an Approach to Reprogramming
Researchers have engineered an electromagnetic‑field (EMF)‑responsive DNA element that remotely activates partial cellular reprogramming genes in mice. By cycling EMF exposure, the system triggers the Oct4‑Sox2‑Klf4 cassette without permanent gene integration, extending median lifespan to 108 weeks—about 70 human years....

Kanvas Biosciences Receives New Funding to Develop First-In-Category Microbiome Therapeutic for Improving Maternal, Newborn and Child Health
Kanvas Biosciences announced a new investment from the Gates Foundation to develop the world’s first fully synthetic microbiome therapeutic targeting maternal environmental enteric dysfunction (EED). The product will use a complex, multi‑strain consortium designed with AI‑driven discovery and spatial microbiome...
Vistagen Announces Completion of Last Patient Visit in PALISADE-4 Phase 3 Public Speaking Challenge Study of Fasedienol for Acute Treatment...
Vistagen announced that the final participant has finished the randomized, double‑blind portion of its Phase 3 PALISADE‑4 trial evaluating fasedienol nasal spray for acute social anxiety disorder. The company expects to release topline efficacy and safety data in the second quarter...
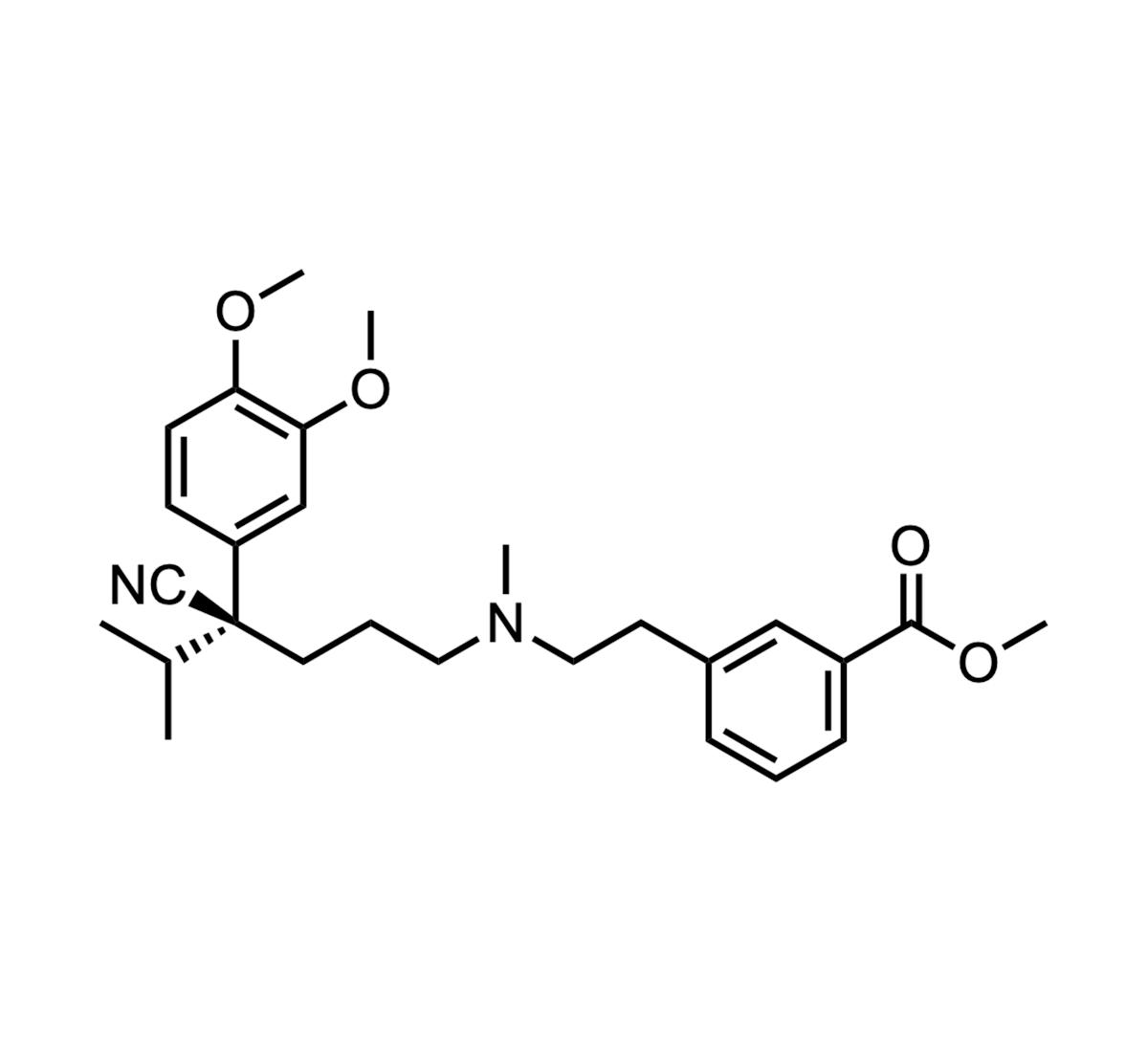
Etripamil
Etripamil (Cardamyst®) received FDA approval as a rapid‑acting, intranasal L‑type calcium channel blocker for converting acute paroxysmal supraventricular tachycardia (PSVT) episodes to sinus rhythm in adults. The drug leverages an ester‑sensitive phenylalkylamine scaffold to achieve fast onset and a short...

Bristol Myers Squibb Receives European Commission Approval of Sotyktu (Deucravacitinib) for the Treatment of Active Psoriatic Arthritis in Adults
Bristol Myers Squibb announced that the European Commission has approved Sotyktu (deucravacitinib) for adults with active psoriatic arthritis, either as monotherapy or with methotrexate. The approval follows positive Phase 3 POETYK PsA‑1 and POETYK PsA‑2 trials, which demonstrated statistically significant improvements in ACR20...

Friday Hope: Eriodictyol: Found in Citrus Fruits, This Flavonoid Downregulates ACE2, TMPRSS2 and TGF-Β
The post highlights eriodictyol, a citrus‑derived flavanone, as a multi‑target therapeutic candidate. Recent studies show it can down‑regulate ACE2 and TMPRSS2, the key entry proteins for SARS‑CoV‑2, and suppress TGF‑β‑driven fibrosis in animal models. Additional research links eriodictyol to neuroprotective...

FDA Approves the First PROTAC in History, a $1B siRNA Wave Hits Cardiometabolic Disease, and Cytokinetics Cracks Non-Obstructive HCM –...
The FDA granted its first-ever approval for a PROTAC drug, Arvinas’ Veppanu, targeting ESR1‑mutated metastatic breast cancer, marking a regulatory milestone for targeted protein degradation. In parallel, precision‑medicine siRNA deals surged: Madrigal paid $25 million upfront (up to $975 million in milestones)...

Endometriosis Imaging Study Highlights 99mTc-Maraciclatide as Diagnostic and Monitoring Tool
Serac Healthcare and Oxford’s Nuffield Department have published Phase 2 results of the DETECT study, showing that the gamma‑emitting radiotracer 99mTc‑maraciclatide can non‑invasively locate endometriotic lesions, including superficial peritoneal disease. Imaging findings matched laparoscopy in 16 of 19 participants and identified disease...

A Light at the End of the Tunnel for Huntington’s Disease Treatment
Researchers at Florida Atlantic University have uncovered a cellular pathway that enables mutant huntingtin protein (mHTT) to travel between neurons via tunneling nanotubes (TNTs). Using LC‑MS/MS, they identified the intracellular pH sensor Slc4a7 as a critical membrane partner of the...

Webinar Q&A Follow Up: Immunoassay Signal Amplification: Bold New Solutions for Existing ELISAs
Cavidi’s principal scientist Peter Stenlund explained how the BOLD signal‑amplification platform boosts ELISA sensitivity by lowering the lower limit of quantification while modestly reducing the upper limit. The technology relies on click‑chemistry conjugation of stable DBCO‑modified oligos, offering precise stoichiometry...

Aplantex Closes $5 Million Bridge Round to Accelerate Shift to Production-Scale Green Biotechnology
Aplantex announced the close of a $5 million bridge financing round in 2025, exceeding its original target. The round was led by board member François Ravenelle with Investissement Québec’s Impulsion Fund as a principal backer. The capital will fund the company’s transition from...

Cera and uMed Announce New Partnership
Cera, Europe’s largest HealthTech provider, has partnered with uMed’s automated living registry platform to bring clinical trial recruitment into patients’ homes. The collaboration leverages Cera’s network of over 2.5 million monthly home‑care visits and uMed’s regulated digital consent platform to reach...

FDA’s Expedited Drug Reviews Are Hailed in some Quarters but Other Approval Practices Are Problematic
In July 2025 the FDA launched the Commissioner’s National Priority Voucher (CNPV) pilot to slash drug review times from six‑to‑eight months down to one‑or‑two months, issuing 17 vouchers for therapies ranging from cancer to cholesterol drugs. Commissioner Marty Makary later...

Why Not Do Random Testing in Randomized Trials Designed to Measure Risk of Infection?
The post argues that randomized trials measuring infection risk, such as the recent Moderna flu study, rely on symptom‑driven testing rather than random testing of all participants. This selective approach inflates reported efficacy by omitting mild or asymptomatic cases and...
Targeting Ischemic Disease with DiaMedica CEO Rick Pauls — Episode 254
DiaMedica Therapeutics, led by CEO Rick Pauls, is advancing a recombinant KLK1 protein to treat ischemia‑driven diseases such as preeclampsia, fetal growth restriction, and acute ischemic stroke. The podcast episode highlights the company’s focus on restoring vascular blood flow and...

Asembia AXS26: How Drug Innovation Could Strain Access Systems
In a follow‑up interview at the Asembia AXS26 summit, Aradigm Health CEO Will Shrank warned that emerging therapies priced above $1 million per patient could overwhelm existing market‑access and financing frameworks. He highlighted upcoming treatments for type 1 diabetes, wet macular degeneration...

Opentrons Debuts Simulation and Visualization for AI-Generated Lab Workflows
Opentrons Labworks introduced Protocol Visualization for Flex, a new simulation and visualization layer built into Opentrons App version 9.0 and slated for release in April 2026. The tool lets scientists preview AI‑generated, Python‑API, or Protocol Designer workflows in a dynamic virtual deck,...

Parallel 3D Bioprinting Builds Tissue Model Arrays in Minutes
Researchers have introduced a slippery‑liquid‑infused porous surface (SLIPS) droplet microarray that enables parallel digital light processing (DLP) bioprinting of hydrogel tissue models. By removing physical walls and using hydrophilic spots on a superhydrophobic background, the system prints dozens to hundreds...

The FDA's One-Day Inspection Pilot Is Already Running
The FDA launched a one‑day inspection pilot announced by Commissioner Marty Makary, with roughly 46 screening assessments completed since April across food, biologics, medical devices and clinical research sites. Most assessments resulted in a No Action Indicated outcome, though a...

Simple Protein Redesign Produces the Most Active Designed Enzyme Ever
Researchers at UCSF combined crystallographic fragment screening with directed evolution to repurpose a simple designed protein, ABLE, into two distinct functional proteins. One of the new proteins, KABLE, is a Kemp eliminase that exhibits ten‑fold higher activity than any previously...

Reviewing the Role of Advanced Glycation Endproducts in Aging and Age-Related Disease
Advanced glycation endproducts (AGEs) are protein‑sugar adducts that accumulate with age, altering protein structure and activating the RAGE receptor to drive chronic inflammation. The review highlights how AGEs cross‑link collagen, stiffening the extracellular matrix and contributing to diabetes, cardiovascular disease,...

The Goalposts Shifted in Berlin, Your Trial Timeline Didn’t
The European Society for Medical Oncology (ESMO) Breast 2026 conference in Berlin showcased a wave of late‑stage breast cancer data that is reshaping therapeutic benchmarks. Several trials reported higher pathologic complete response rates and novel biomarker‑driven combinations, tightening the competitive...
More Evidence for Muscle Stem Cell Activity to Be Inhibited by the Aged Tissue Environment
Researchers discovered that the extracellular matrix (ECM) of aged mice suppresses the growth of both young and rejuvenated muscle stem cells (MuSCs). Elevated collagen levels in the aged ECM create a non‑autonomous barrier that limits stem‑cell proliferation, even when intrinsic...