
Pharma Pulse: GLP-1 Momentum Builds While Lilly Expands Genetic Medicine Manufacturing
Eli Lilly opened its first dedicated genetic‑medicine manufacturing plant in Lebanon, Indiana, expanding U.S. capacity for advanced gene‑editing and RNA therapies. GLP‑1 drugs accounted for eight percent of all prescriptions filled in March 2026, highlighting their growing role beyond diabetes. Bayer agreed to acquire ophthalmology‑focused Perfuse Therapeutics, while Madrigal licensed ARO‑PNPLA3 for MASH treatment. The FDA also authorized fruit‑flavored electronic nicotine delivery systems, signaling a notable regulatory shift.

The Vaccine Safety Signal the Media Still Won’t Read
A peer‑reviewed study published in Vaccine (Sept 2022) re‑analyzed Pfizer and Moderna mRNA COVID‑19 trial data and found excess serious adverse events—10.1 per 10,000 for Pfizer and 15.1 per 10,000 for Moderna—yielding harm‑to‑benefit ratios of roughly 4.4 : 1 and 2.4 : 1 respectively. The...

Immortal Dragons: The Quest to ‘Make Death Optional’
Immortal Dragons, a Singapore‑based longevity fund founded by 34‑year‑old CEO Boyang Wang, secured $40 million—$4 million from friends and family and $36 million of Wang’s own money—to back moonshot biotech projects. The fund’s portfolio includes Frontier Bio, which is developing 3D bio‑fabrication techniques...

The Latest News in Vaccine Obstruction
Large-scale safety studies of COVID‑19 and shingles vaccines, analyzing millions of records, found rare serious side effects, but the FDA blocked their publication citing unsupported conclusions. The agency also refused to file Moderna’s mRNA flu‑COVID combo vaccine, despite European approval...

Metabolic Stability in Peptide Therapeutics
Peptide therapeutics are gaining traction but remain hampered by poor metabolic stability, limited permeability, and rapid clearance. The article outlines four primary metabolic pathways—hydrolysis, oxidation, reduction, and conjugation—and examines the hurdles of oral delivery, in‑vitro tools, and experimental workflows used...

AstraZeneca’s Camizestrant Hit by FDA Advisory Committee Vote While Truqap Moves Ahead
FDA’s Oncologic Drugs Advisory Committee issued a split verdict on AstraZeneca’s oncology pipeline this week. The committee voted 3‑to‑6 against the benefit‑risk profile of camizestrant combined with a CDK4/6 inhibitor for ESR1‑mutated advanced breast cancer, despite a 56% progression‑free survival...

Pharmaceutical Executive Daily: Zentalis Doses First Patient with Azenosertib in Phase III Trial
Zentalis Pharmaceuticals announced the first patient dosing in the Phase III Aspenova trial of Azenosertib, an oral WEE1 inhibitor for Cyclin E1‑positive, platinum‑resistant ovarian cancer. The study is run with the GOG Foundation, the European Network of Gynecological Oncology Trials (EN‑GOT), and...

Assessing Candidate IGF-1 Receptor Inhibitors for the Ability to Modestly Slow Aging in Mice
The study tested two small‑molecule IGF‑1 receptor inhibitors, picropodophyllin (PPP) and NVP‑ADW742, in 13‑month‑old C57BL/6 mice to assess healthspan and survival. Both drugs improved memory, blood pressure, glucose tolerance and frailty metrics, with NVP‑ADW742 extending healthspan by about 93 days....

PRP Therapy Protocols Lack Expert Consensus
Platelet‑rich plasma (PRP) therapy lacks a unified peri‑procedural protocol, with leading experts disagreeing on NSAID washout periods, supplement restrictions, cryotherapy, and rehabilitation timing. The article highlights that ten top clinicians offered divergent recommendations on pre‑procedure NSAID use, corticosteroid washout, and...

S-Mitochonic Acid 5. Increases ATP, NAD+ and SIRTUINS
Researchers have synthesized the S‑enantiomer of Mitochonic Acid‑5 (MA‑5) with 99 % enantiomeric purity. The compound strengthens the mitochondrial protein Mitofilin, preserving crista junction geometry and boosting ATP synthase efficiency. It also acts as a direct NAMPT agonist, raising intracellular NAD⁺,...

Eli Lilly Opens First Dedicated Genetic Medicine Facility
Eli Lilly inaugurated Lilly Lebanon Advanced Therapies, its first dedicated genetic‑medicine manufacturing site in Lebanon, Indiana, alongside a $4.5 billion capital infusion. The investment lifts Lilly’s Indiana spending to over $21 billion since 2020 and its U.S. commitments to more than $50 billion. The...
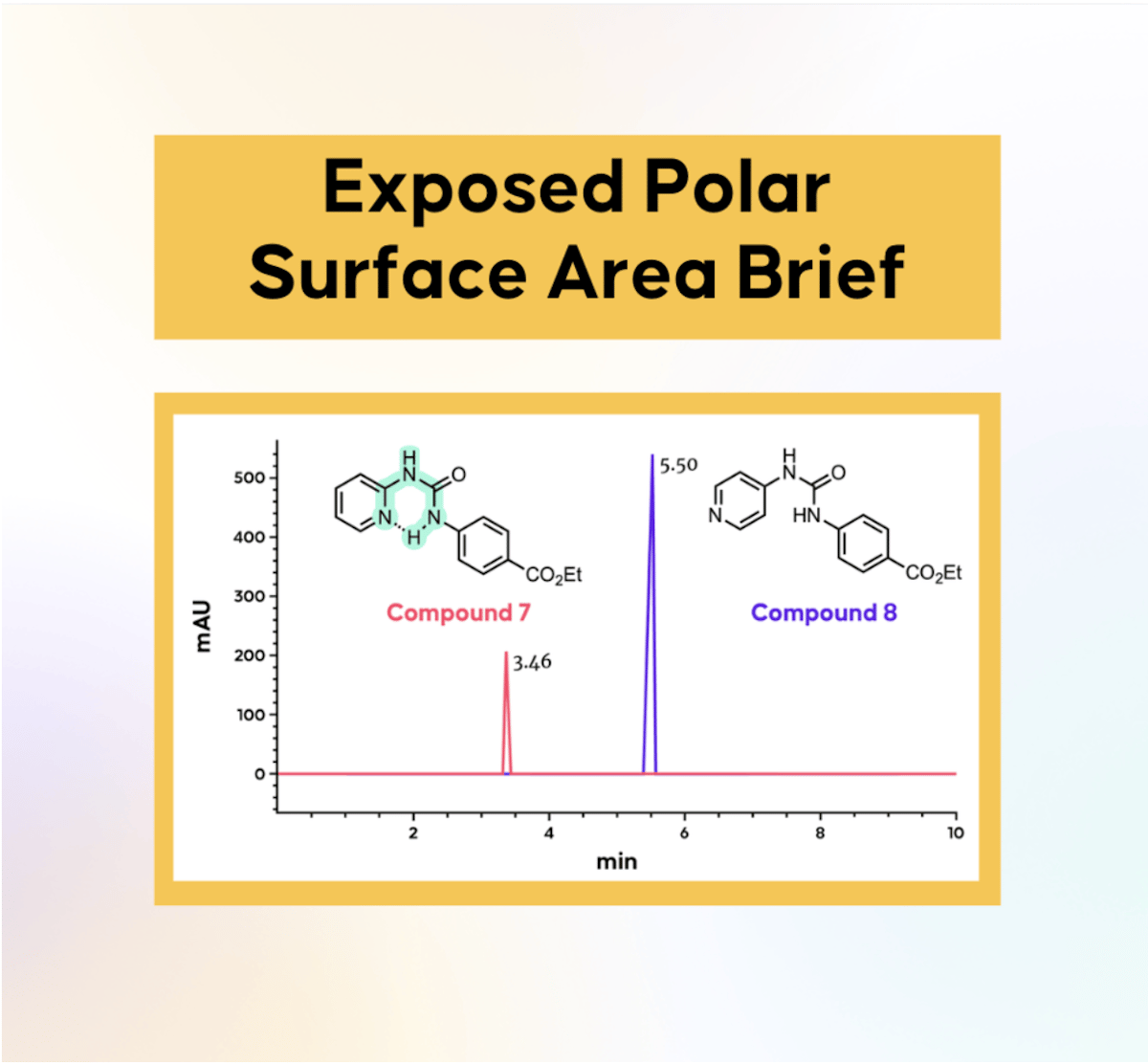
EPSA: A Useful Metric Across Chemical Space
The article highlights EPSA (Experimental Polarity Surface Area) as a robust metric for assessing molecular polarity across broad chemical space. Unlike traditional PSA, EPSA is derived from supercritical fluid chromatography, offering experimental insight into a compound’s three‑dimensional polarity profile. The...

Waiv Enters Collaboration with Daiichi Sankyo to Deliver AI-Derived Biomarkers for ADC Program
Waiv, the Paris‑based AI precision‑testing firm formerly known as Owkin Dx, has partnered with Daiichi Sankyo to lead digital pathology biomarker discovery for an antibody‑drug conjugate (ADC) program. The collaboration will apply Waiv’s end‑to‑end computational pathology platform to early‑phase trial data,...

Lucent Diagnostics Announces Collaboration with Tempus to Integrate Blood-Based Alzheimer’s Biomarker Testing Into Clinical Workflows
Lucent Diagnostics, a Quanterix brand, partnered with Tempus AI to embed its LucentAD® Complete blood‑based Alzheimer’s biomarker panel into Tempus’ clinical ordering platform. The collaboration creates a Tempus Next care‑gap program that automatically identifies patients who meet guideline criteria for...

Simulations Plus and NVIDIA Collaborate to Scale GPU-Accelerated, AI-Assisted Modeling Workflows
Simulations Plus announced a technical collaboration with NVIDIA to embed GPU‑accelerated and AI‑assisted modeling into drug‑development workflows. The partnership will re‑engineer Simulations Plus’ PBPK, PK/PD and QSP engines for NVIDIA GPUs, delivering up to a 75% reduction in simulation runtimes...

Most Oncology Brands Lose Before Launch Day — New ZoomRx Analysis of 40+ Drugs Quantifies the Awareness Gap That Predicts...
ZoomRx’s 2026 "Laggards and Leaders" analysis of 44 oncology brands shows that pre‑launch physician awareness drives commercial success. Brands entering the market with about 75% aided awareness outperform those starting at 55%, a 25‑point gap that persists through four years....

Capturing True Single-Cell Resolution with Your Spatial Data
Spatial biology has transformed life‑science research, yet imaging and sequencing platforms still grapple with cell‑boundary segmentation and grid‑based spot limitations. Linda Orzolek of OMAPiX explains how Takara Bio’s Trekker technology delivers true single‑cell spatial resolution by isolating nuclei and pairing...

Can Mammals Regrow Lost Limbs? This New Treatment Could Be the First Step
Researchers at Texas A&M have demonstrated that a two‑step treatment using growth factors FGF2 and BMP2 can trigger partial digit regeneration in mice. The protocol first applies FGF2 to create a blastema‑like cell mass, then adds BMP2 to drive bone...

Understanding Potential Ocular Side Effects of Injectable GLP-1 Medications
Recent research suggests a rare but serious link between the GLP-1 receptor agonist semaglutide (Ozempic, Wegovy) and non-arteritic anterior ischemic optic neuropathy (NAION), a form of eye stroke that can cause permanent vision loss. A 2026 JAMA Network Open study...

“Sounds Great. Do You Want FDA Approval?” A Regulatory Analysis of Psychedelics
On April 18, 2026 President Trump signed an executive order compelling the FDA to prioritize review of psychedelic drugs, issuing priority‑review vouchers for three compounds already holding Breakthrough Therapy designation. The order also creates a Right‑to‑Try pathway for ibogaine, directs...

Piezoelectric MXene Scaffold Promotes Cartilage Repair While Limiting Vessel Growth
Researchers unveiled an origami‑folded PLLA/MXene scaffold that converts joint motion into piezoelectric signals and, when exposed to near‑infrared light, generates mild heat. The dual‑modality design doubles electrical output versus pure PLLA and reaches ~41 °C, a temperature that suppresses VEGF‑driven angiogenesis...

When the Data Favor Motion Preservation, How Long Does It Take for Surgeon Culture to Catch Up?
Recent IDE trial data on the Total Posterior Spine (TOPS) System suggest that motion‑preserving implants can match or exceed outcomes of traditional fusion for grade I degenerative spondylolisthesis at L4‑5. The study showed comparable pain relief, functional scores, and lower rates...

Pharmaceutical Executive Daily: BioNTech Reports 2026 First Quarter Results
Cellenkos secured FDA clearance to start a Phase 1b/2a trial of its off‑the‑shelf allogeneic T‑reg therapy CK0802 for steroid‑refractory graft‑versus‑host disease, with enrollment slated for late 2026. BioNTech reported first‑quarter 2026 revenue of $138 million, a 35% drop from the prior year,...

FDA Clears Investigational New Drug Application for Phase Ib/IIa Trial of CK0802 in Steroid-Refractory Graft-Versus-Host Disease
The U.S. Food and Drug Administration has cleared Cellenkos' investigational new drug application for CK0802, paving the way for a Phase Ib/IIa trial in patients with steroid‑refractory graft‑versus‑host disease (GVHD). The mid‑stage study will evaluate safety, tolerability and early efficacy, using...

Austin Russian: How Fragmentation Delays Rare Disease Therapy Access
Austin Russian, SVP of Program Excellence at PANTHERx Rare, warned that fragmentation across prescribers, insurers, pharmacies and manufacturers slows patient access to orphan drugs. As more rare‑disease therapies reach the market, the lack of a single coordinating entity creates miscommunication...

Partial Reprogramming Concern Altos Labs Is Becoming Less Stealthy
Altos Labs, launched in 2022 with roughly $3 billion in private funding, is intensifying its public profile as it pursues partial cellular reprogramming to reverse organ aging. The company is racing alongside rivals such as Life Biosciences, which has just begun...

PharmaLogic Expands Radiopharmaceutical Footprint With New Atlanta Facility
PharmaLogic announced the opening of a new PET radiopharmaceutical manufacturing facility in Atlanta, Georgia, as part of its $250 million US expansion. The state‑of‑the‑art plant will increase production capacity, accelerate delivery to Southeast hospitals, and serve as an innovation hub for...

Bracco Launches BubbleGen™ Early Access Program for Microbubble-Based Cell Selection and Activation at ISCT
Bracco Imaging announced an Early Access Program for its new BubbleGen™ technology, which uses buoyant microbubbles to isolate and activate specific cell subtypes. The platform offers a one‑step, magnetic‑residue‑free alternative to traditional bead‑based cell separation, initially demonstrated with CD3⁺ T‑cell selection...

NImmune Biopharma Announces Presentations at Digestive Disease Week 2026 Supporting a Differentiated Profile and Superior Efficacy of Oral, Once-Daily NIM-1324...
NImmune Biopharma presented Phase 1 data for its oral LANCL2 drug NIM‑1324 at Digestive Disease Week, showing safety, tolerability, target engagement and superior efficacy versus existing IBD therapies. The study met all primary and secondary endpoints with no dose‑limiting toxicities and...
Hepta Reveals Blood-Based Epigenetic Signatures of GLP-1 Response, Enabling Precision Medicine in Obesity and MASH
Hepta unveiled a blood‑based cfDNA methylation assay at Digestive Disease Week 2026 that can identify patients who will lose at least 10% of body weight on semaglutide before the first dose. The SAMARA trial showed baseline epigenetic signatures distinguished responders...
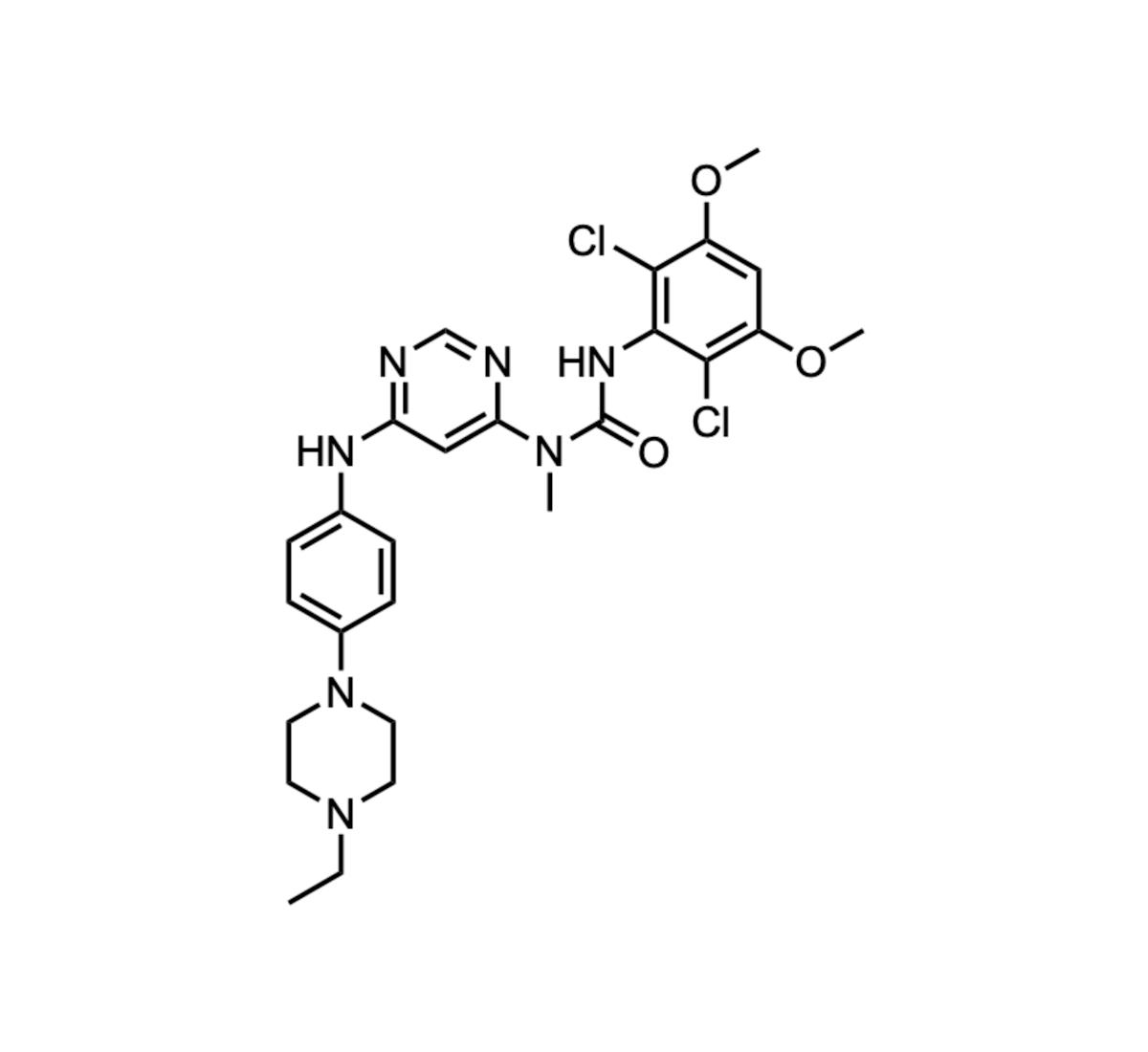
Infigratinib
Infigratinib, a pan‑FGFR inhibitor previously approved for cholangiocarcinoma, is being repurposed to treat achondroplasia. After its FDA accelerated approval was rescinded in 2024 due to enrollment challenges, BridgeBio reported that the Phase 3 PROPEL 3 trial met its primary endpoint in February 2026....

Connected by Design: How AI and Automation Are Transforming Drug Discovery at BMS
Bristol Myers Squibb (BMS) is shifting from isolated AI tools to an integrated, learning‑driven ecosystem that connects data, models, and automation across discovery and development. The company highlights its shared data backbone, AI co‑scientists, and lab‑in‑the‑loop automation as foundational layers,...

From PhD to Regional Medical Director
Jill, a PhD‑trained scientist, now serves as Regional Medical Director (Medical Science Liaison) in Denver, overseeing scientific communication, clinical strategy, and KOL engagement for cancer therapies. Her path moved from postdoc to scientific project manager, then to MSL before attaining...

Odyssey Therapeutics (ODTX) IPO Deck
Odyssey Therapeutics, a clinical‑stage biopharma specializing in precision medicines for autoimmune and inflammatory disorders, unveiled its initial public offering deck in May 2026. The company aims to raise capital to advance its Phase 2‑tested pipeline, which targets conditions such as rheumatoid...

Pointed Ironies: SERD Wars, ADC Hype, and What Really Works in Breast Cancer
The FDA rejected camizestrant at the ODAC meeting, while approving vepdegestrant a day later. AstraZeneca’s vepdegestrant leverages ctDNA to detect ESR1 mutations early, allowing a treatment switch while patients remain on a CDK4/6 inhibitor backbone. In contrast, Arvinas pursued a...

The Technology Behind the Story: 3D Skin Grafts and the World of Scarpetta
The article highlights how 3D‑printed skin grafts are transitioning from laboratory experiments to clinical tools for complex wounds. Researchers at Columbia University have demonstrated patient‑specific, three‑dimensional grafts that fit irregular body parts like a glove, reducing surgery time and improving...
NHS to Roll Out ‘1-Minute’ Immunotherapy Jab to Tens of Thousands with Cancer
The NHS is introducing a sub‑cutaneous form of pembrolizumab (Keytruda) that can be administered in just one minute, replacing the traditional two‑hour IV infusion. The rapid jab is approved for 14 cancer types, including lung, breast, head‑and‑neck, and cervical cancers,...
Can Hydra Biology Inform Strategies to Extend Life in Other Species?
Hydra vulgaris displays negligible senescence thanks to a perpetual pool of stem cells that continuously replace its tissues. Researchers suggest that inserting Hydra‑like gene expression patterns into the short‑lived rotifer Brachionus manjavacas could extend the rotifer's healthspan and lower age‑related...

Inhibiting ID1 and ID3 Reduces Lung Fibrosis
Researchers identified that the transcription factors ID1 and ID3 are markedly elevated in fibroblasts from idiopathic pulmonary fibrosis (IPF) patients. Using both small‑molecule inhibitors and gene‑silencing techniques, they suppressed these proteins in mouse models, which halted fibroblast activation and triggered...

RESEARCH: HBOT in CANCER (Hyperbaric Oxygen Therapy) - 2025 Review Paper From Sichuan University, Chengdu, China
A 2025 review paper from Sichuan University evaluates hyperbaric oxygen therapy (HBOT) as an adjunct in cancer treatment. The authors synthesize pre‑clinical data showing HBOT reverses tumor hypoxia and enhances the potency of radiotherapy and certain chemotherapies. They also examine...

Cibus Transfers Gene-Edited Herbicide-Tolerance Traits in Rice to Interoc Ahead of Latin American Launch
Cibus Inc. has transferred gene‑edited rice material that confers herbicide tolerance to its Latin American partner Interoc, meeting a scheduled development milestone. Interoc will begin testing and scaling production to launch Cibus‑enhanced seed varieties across the region. The transfer leverages...
Can Ivermectin and Mebendazole Treat Cancer?
The Wellness Company posted a self‑reported study of its ivermectin 25 mg/mebendazole 250 mg capsule, claiming an 84% clinical‑benefit ratio and that 33% of participants showed no evidence of disease. The analysis drew on 197 baseline surveys and 122 six‑month follow‑ups, but relied solely...

Canagliflozin - Another Top Longevity Drug
The class of sodium‑glucose cotransporter‑2 (SGLT2) inhibitors has moved from glucose‑lowering pills to a cornerstone of cardiometabolic care. Large‑scale trials and real‑world registries consistently show that agents such as canagliflozin, dapagliflozin and empagliflozin cut heart‑failure admissions, slow chronic kidney disease...

Loyal Raises $100 Million: Dog Longevity Drugs Targeting IGF-1 and PPAR Pathways
Loyal, a biotech startup focused on canine longevity, raised $100 million in Series A funding to advance its anti‑aging pill that targets the IGF‑1 and PPAR pathways. The drug received a favorable “likely effective” assessment from the FDA, positioning it for market...

The FDA Wants to Make Many Popular Prescription Drugs OTC—A Great Idea. Here’s Why It’s Unlikely to Happen
FDA Commissioner Marty Makary argues that most prescription drugs should be reclassified as over‑the‑counter unless safety concerns exist. The article highlights how prescription status inflates prices by routing purchases through insurance, citing lower cash prices for OTC versions of Claritin,...

Telomeres: History, Health and Hallmarks of Aging
Bill Andrews, a co‑discoverer of human telomerase, argues that telomere shortening is the primary limiter of human lifespan and that systemic activation of telomerase can reverse biological aging. He promotes small‑molecule activators such as TAM‑818 and botanical blends like Telo‑Vital,...

Molecular Hydrogen May Reduce Fatigue and Support Physical Function in People with Long COVID
A single‑blind, 14‑day pilot trial published in *Nutrients* examined hydrogen‑rich water versus regular water in 32 adults with long‑COVID. Participants drinking the hydrogen‑infused water reported statistically significant reductions in fatigue and showed measurable gains in six‑minute walk distance (42‑62 m), chair‑stand...

Ubiquitin Rides Again
A new Nature paper introduces a high‑throughput assay that reveals ubiquitination of non‑protein substrates, most notably glycogen. The study shows that ubiquitinated glycogen is routed to lysosomes for degradation, a pathway that intensifies during fasting and is disrupted in glycogen...

FDA Clears Custom Titanium Cranial Implant for Skull Repair
The U.S. FDA granted 510(k) clearance to CGBIO’s EASYMADE TI, a patient‑specific titanium cranial and craniofacial implant made via laser powder‑bed fusion. The device is designed from each patient’s CT scan, manufactured in South Korea and shipped to U.S. hospitals within...

Pharmaceutical Executive Daily: UCB to Acquire Candid Therapeutics
UCB signed a definitive agreement to acquire privately held Candid Therapeutics for $2 billion upfront with up to $200 million in milestones. The deal, slated to close by late Q2 or early Q3 2026 pending antitrust clearance, adds a suite of T‑cell engager...