
Seaport Therapeutics (SPTX) IPO Deck
Seaport Therapeutics, a clinical‑stage biotech firm, is preparing an initial public offering to fund its oral drug program targeting major neuropsychiatric conditions such as depression and anxiety. The company’s IPO deck emphasizes a “proven path” in neuroscience, highlighting pre‑clinical data and a roadmap toward Phase 2 trials. Management leverages experience from large pharma to accelerate development and market entry. The offering seeks to raise roughly $150 million to expand its pipeline and commercial capabilities.

The Biotech Bi-Weekly: A Virtual Biology Initiative, a New Discovery Grant and a Protein Supplier to Watch in Cancer Research...
The biotech bi‑weekly highlights a wave of new funding and tools, starting with Biohub’s $500 million five‑year Virtual Biology Initiative to generate global multimodal datasets for predictive biology. Zymo Research launched the Fecal Microbiome Discovery Grant to support early‑stage researchers, while...
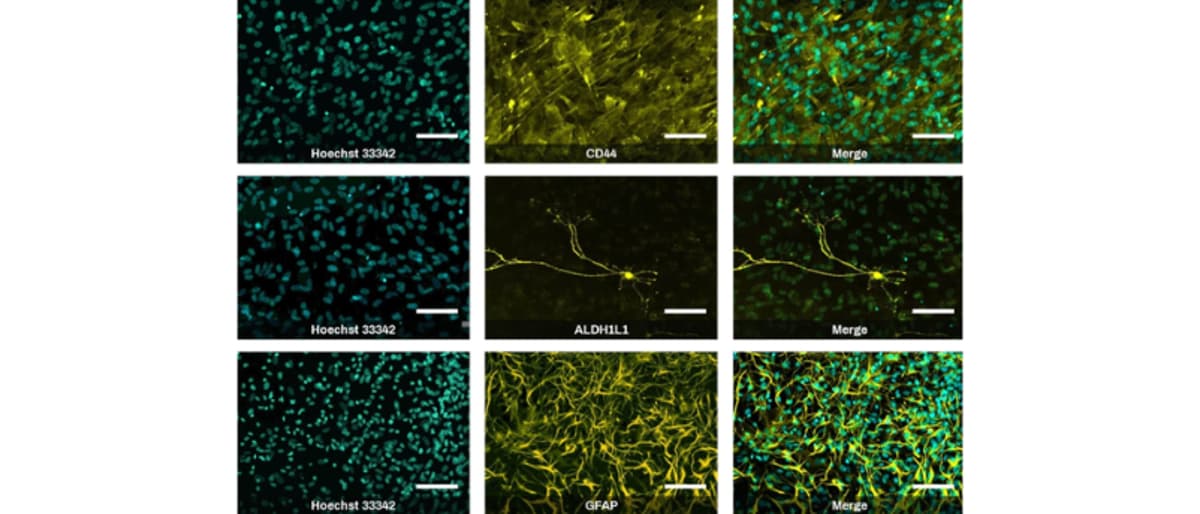
New iPSC Differentiation Kits for Neuroscience Research
AMSBIO introduced the Quick‑Glia™ product line, iPSC‑derived glial cell kits designed for neuroscience research. The kits convert human induced pluripotent stem cells into functional astrocytes or microglia in 1–2 weeks, delivering high‑purity, cryopreserved cells ready for disease modeling and drug...

XPRIZE Healthspan Names Top 100 Teams Advancing Healthy Aging
The XPRIZE Healthspan competition announced its top 100 teams, spotlighting the core innovations of the 40 Milestone 1 award‑winning entrants. These teams are pursuing a spectrum of strategies—from mitochondrial‑targeted small molecules and metformin‑rapamycin combos to AI‑driven nutrition plans, senolytic drugs, and...
A Study in Regulatory Conservatism versus Clinical Innovation
At today’s ODAC meeting, the FDA will review AstraZeneca’s oral selective estrogen receptor degrader camizestrant for hormone‑receptor‑positive, HER2‑negative breast cancer with ESR1 mutations. Late‑stage data showed a 56% reduction in risk of disease progression and consistent safety, along with improved...

Cardiovascular Health 2026
Recent studies highlight pitavastatin’s pleiotropic benefits beyond LDL‑C reduction, including functional HDL elevation, enhanced cholesterol‑efflux capacity, and antioxidative actions in dyslipidemic patients. Pre‑clinical work shows the drug strengthens blood‑brain barrier integrity and mitigates lipopolysaccharide‑induced BBB dysfunction, suggesting neuroprotective potential. Real‑world...

Your Weekly TechBio News: High-Throughput Screening
High‑Throughput Screening (HTS) remains a cornerstone of modern drug discovery, allowing researchers to evaluate millions of chemical compounds against biological targets in a single campaign. Recent advances in robotics, miniaturized assay formats, and cloud‑based data pipelines have dramatically increased throughput...

Generally Good Indian Pharma Companies
An audit of top‑tier Indian generic manufacturers evaluated options for 20 therapeutic compounds, ranking firms by market capitalization, API vertical integration, ANDA volume, and GMP compliance. Cipla, Abbott, Sun Pharma, Zydus, Biocon and others were highlighted for specific products such...

Pharmaceutical Executive Daily: Rocket Pharmaceuticals Sells Pediatric Disease Priority Review Voucher
Rocket Pharmaceuticals agreed to sell its rare‑pediatric disease priority‑review voucher for $180 million in cash, providing non‑dilutive funding as the PRV program was reauthorized in early 2026. Teva Pharmaceutical announced a definitive agreement to acquire Emalex Biosciences for up to $900 million,...

March 2026 Patent Highlights
The March 2026 Patent Highlights page aggregates the latest drug‑discovery milestones, from 38 first‑time small‑molecule approvals by Europe’s EMA, China’s NMPA and Japan’s PMDA to a deep dive on protein‑structure advances and machine‑learning tools. It spotlights a newly optimized HPK1 inhibitor...

Teva Enters $900 Million Agreement to Acquire Emalex
Teva Pharmaceutical Industries announced a $900 million acquisition of Emalex Biosciences, securing the late‑stage pediatric Tourette syndrome candidate ecopipam. The deal includes a $700 million upfront payment and up to $200 million in milestone fees, with the transaction slated to close in Q3 2026....

FDA Knew of Better Method to Detect COVID Vaccine Safety Signals — But Refused to Use It
Senator Ron Johnson released a report showing that FDA officials during the Biden administration knew a superior empirical Bayesian data‑mining method existed for detecting COVID‑19 vaccine safety signals in VAERS but ordered staff to continue using the older, flawed tool....

Thermo Fisher Scientific Launches Applied Biosystems PowerFlex Thermal Cycler
Thermo Fisher Scientific unveiled the Applied Biosystems PowerFlex Thermal Cycler, a next‑generation PCR instrument that emphasizes speed, precision, and flexibility. The system features a 10.1‑inch touchscreen, AI‑driven Smart Help, and two configuration options—a 96‑well plate and a 3 × 32‑well layout with...
Nanozymes Against Brain Tumors
Researchers at Empa and HOCH Health Ostschweiz are developing biocompatible nanozymes that can be applied directly during brain‑tumor surgery to attack astrocytoma cells. The nanozymes act like enzymes, generating reactive‑oxygen species and activating drug precursors, and they are triggered by...

Meet the Brand New Excuse for Medical Failures; It’s a Doozy
Google AI released research indicating roughly 10% of patients may not respond to GLP‑1 weight‑loss drugs because of specific genetic variations. The finding is framed as a scientific explanation for drug inefficacy, suggesting that patient genetics, not the medication, drive...

Reduced Ghrelin Receptor Activity Improves Mitochondrial Function and Muscle Function in Aged Mice
Researchers demonstrated that reducing activity of the ghrelin receptor (GHSR‑1a) improves muscle endurance and mitochondrial function in aged mice. Both genetic knockout and the inverse‑agonist PF‑5190457 increased markers of mitochondrial biogenesis and mitophagy, enhancing fatigue resistance. The interventions did not...
Arguing for an Emphasis on Comparative Organelle Biology
Researchers argue that aging studies should shift from a gene‑by‑gene focus to holistic comparisons of organelle structures across species. While genome‑centric approaches have identified hallmarks of aging, they often fail to explain why interventions that extend lifespan in short‑lived models...

Word Games: How Moderna Is Selling Its Newest Vaccine without Using the “V” Word
Moderna’s $776 million federal award for a bird‑flu vaccine is under scrutiny after U.S. officials targeted mRNA technology, prompting the company to warn it may halt late‑stage vaccine programs. Simultaneously, Moderna and Merck are advancing an mRNA‑based cancer treatment, which Merck...

CRL Release Update – No Litigation War yet, but New Citizen Petition Beats the Drums
The FDA has begun posting Complete Response Letters (CRLs) for unapproved NDAs, ANDAs, and BLAs, releasing 127 documents to date, including 36 that had never been publicly disclosed. A Washington‑based law firm filed a Citizen Petition on April 20, 2026,...

Peptides / Bioregulators
The AGI House in San Francisco hosted the inaugural California Peptide Club, drawing over 100 invite‑only attendees to discuss self‑optimization peptides. Organiser Julius Ritter highlighted the fragmented information landscape and showcased stacks ranging from growth‑hormone releasers to cognitive enhancers. Days later, HHS...

FOXO4-DRI Is Fascinating, but Was Never Intended for Human Use, What Are the Takeaways?
FOXO4‑DRI is an experimental senolytic peptide that selectively eliminates senescent cells by disrupting the FOXO4‑p53 interaction, prompting p53‑mediated apoptosis. Pre‑clinical studies across vascular, reproductive, musculoskeletal and renal models report improved endothelial function, restored testosterone production, chondrocyte rejuvenation, and reduced frailty....

Peptides / Bioregulators
A new study examined 6,441 gray‑market peptide samples covering 14 compounds, measuring purity, dose accuracy, and endotoxin levels. Between 41.6% and 71.1% of the products failed basic pharmaceutical standards, and 2.4% contained no active peptide at all. Endotoxin contamination appeared...

Semiglutide Regenerates Cartilage Loss Through Weight Loss Independent Metabolic Restoration Mechanism
Semaglutide, a GLP‑1 receptor agonist originally developed for diabetes, is showing promise as a cartilage‑regenerating therapy. Preclinical pair‑fed mouse studies demonstrate cartilage protection independent of weight loss, while a 24‑week pilot in humans reported a 17% increase in cartilage thickness....
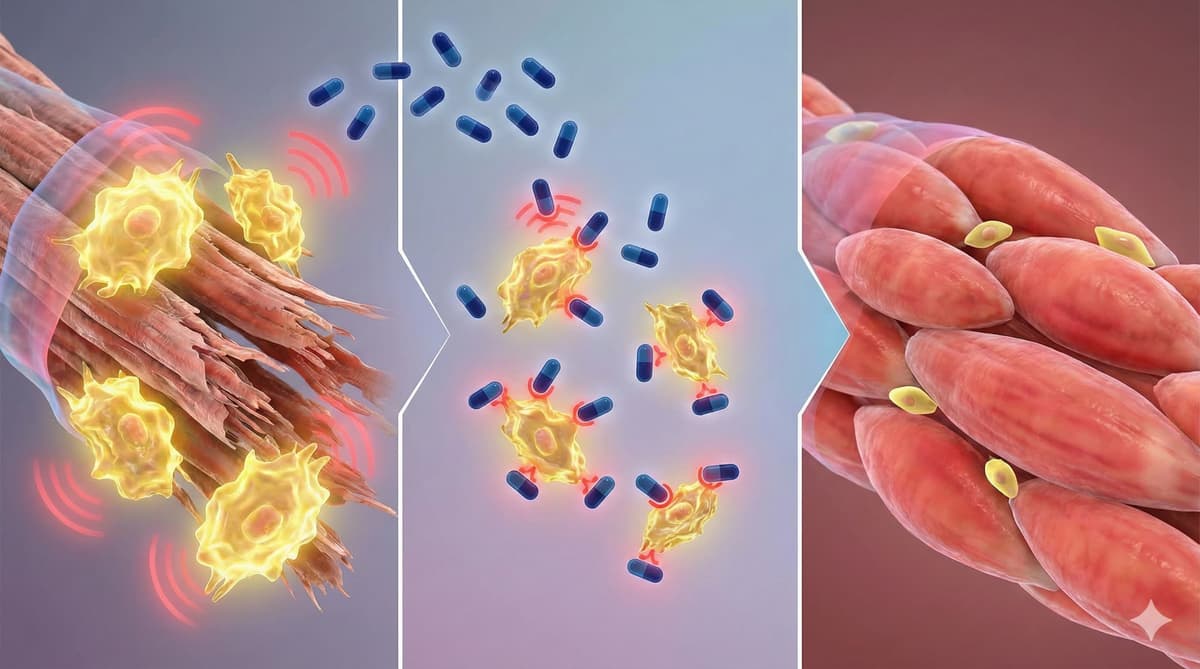
HIV Drug (Maraviroc) Reverses Muscle Aging by Purging “Zombie Cell” Signals
Researchers are exploring the HIV CCR5 antagonist maraviroc as a senomorphic agent that could blunt muscle aging by dampening chronic SASP signaling. Modeling suggests a 75 mg once‑daily dose achieves high CCR5 occupancy, but human data on sarcopenia are absent. The...

Alamar Biosciences IPO Raises $219.9M for Blood-Based Biomarker Tools
Alamar Biosciences closed an upsized IPO, raising $219.9 million by pricing 12.9 million shares at $17 each, and began trading on Nasdaq under the ticker ALMR. The California‑based firm offers the NULISA platform, which detects low‑level protein biomarkers in blood, and its...

Kailera Therapeutics IPO Raises $718.8M for Obesity Drug Pipeline
Kailera Therapeutics completed a $718.8 million IPO, pricing 44.9 million shares at $16 each and listing on Nasdaq under KLRA. The company’s lead candidate, ribupatide, is a once‑weekly injectable GLP‑1/GIP dual agonist currently in three global Phase III trials targeting obesity and type 2...
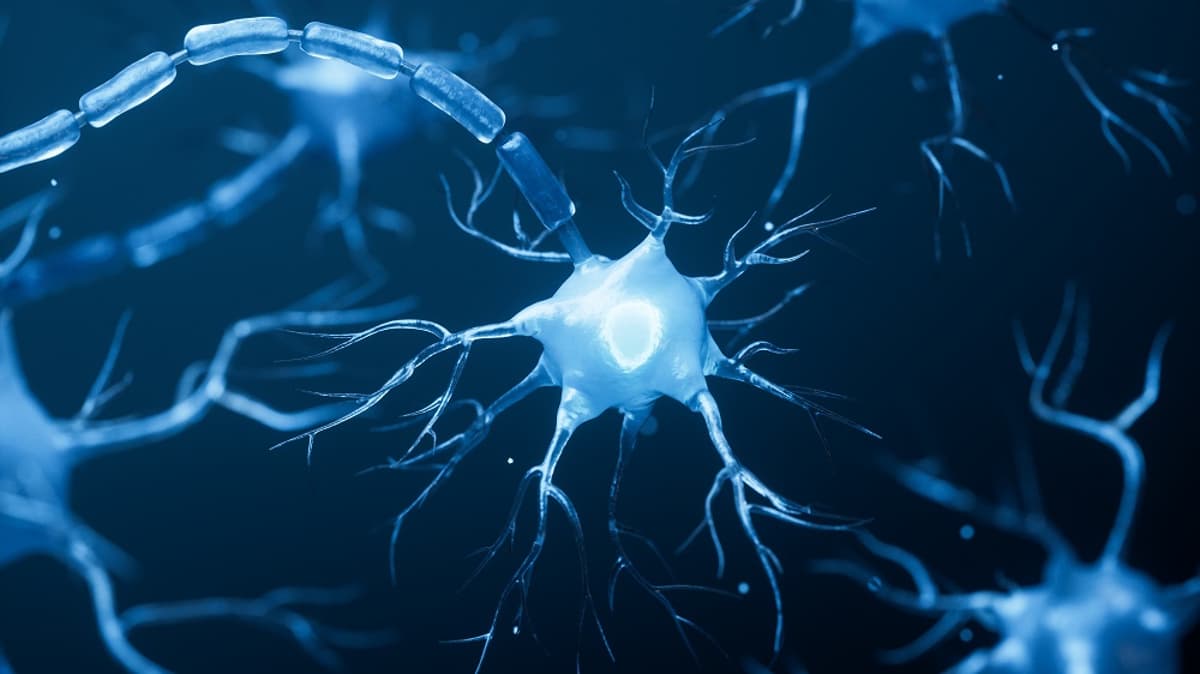
A Popular Senolytic Treatment Causes Brain Damage in Mice
A recent PNAS study shows that the widely used senolytic cocktail dasatinib plus quercetin (D+Q) impairs myelination in the mouse corpus callosum. The treatment altered oligodendrocyte morphology within minutes, reduced myelin thickness, and triggered endoplasmic reticulum stress, without killing the...

With Cell and Gene Therapies Proliferation, Specialty Pharmacy Faces New Pricing, Distribution Pressures
Cell and gene therapies are moving from ultra‑rare indications to broader patient populations, prompting a fundamental redesign of specialty‑pharmacy distribution and pricing models. At the 2026 Asembia meeting, experts warned that current flash‑title and buy‑and‑bill channels may not scale to...

Profluent and Lilly: The Next Gene Editor Will Be Designed by AI
Profluent, an Air Street Capital portfolio company, announced a multi‑program partnership with Eli Lilly to create AI‑designed recombinases for kilobase‑scale gene editing. The deal includes an upfront cash payment, committed R&D funding, and up to $2.25 billion in development and commercial milestones...
Ashvattha Therapeutics Announces Presentations Highlighting Mechanism of Action for Migaldendranib in Diabetic Macular Edema and Neovascular Age-Related Macular Degeneration
Ashvattha Therapeutics presented Phase 2 data on its subcutaneous nanomedicine migaldendranib (MGB) for diabetic macular edema and neovascular age‑related macular degeneration at ARVO. The two‑stage trial showed stable central subfield thickness for up to 12 weeks and maintained visual acuity without...

Cellares and Cabaletta Bio Sign 10-Year Commercial Supply Agreement to Scale Rese-Cel
Cellares has entered a 10‑year commercial supply agreement with Cabaletta Bio to manufacture rese‑cel, the company’s autologous CAR‑T therapy for autoimmune diseases, using its fully automated Cell Shuttle and Cell Q platforms. The deal secures long‑term capacity to produce thousands of...
FDA Approves Breztri for Patients 12 Years and Older with Asthma
The FDA has approved Breztri Aerosphere, a single‑inhaler triple‑combination of budesonide, glycopyrrolate, and formoterol, for maintenance treatment of asthma in patients aged 12 and older. This marks the first U.S. approval of a triple therapy that adds a long‑acting muscarinic...
Targeting Senescent Cells as a Treatment for Chronic Obstructive Pulmonary Disease
Researchers have introduced BCLXL-PROTAC, a proteolysis‑targeting chimera that degrades the anti‑apoptotic protein BCLXL in senescent lung cells. In primary small‑airway epithelial cells and fibroblasts from COPD patients, the compound induced caspase‑3‑mediated apoptosis and lowered classic senescence markers such as p21,...

Senescent Macrophages Are Important in Liver Aging and Liver Disease
Researchers identified a distinct p21‑positive, TREM2‑positive senescent macrophage population that accumulates in aging and fatty livers. These cells drive chronic inflammation through a senescence‑associated secretory phenotype linked to type I interferon signaling. In mouse models, senolytic agents that selectively eliminate these...

How to Get Pfizer & Moderna mRNA Out of Your Body
The article explains that Pfizer‑BioNTech and Moderna COVID‑19 vaccines rely on lipid‑nanoparticle‑encapsulated synthetic mRNA that is chemically altered with N1‑methylpseudouridine. This modification cloaks the RNA from innate immune sensors and dramatically slows enzymatic breakdown, extending its intracellular lifespan. Consequently, the...

Samsung Bioepis Releases Second Quarter 2026 US Biosimilar Market Report
Samsung Bioepis released its Q2 2026 U.S. Biosimilar Market Report, the thirteenth edition of the quarterly series. The report documents 92 FDA‑approved biosimilars across 20 molecules, with 67 (73 %) already launched, and notes a 60 % adoption rate for adalimumab biosimilars. It...
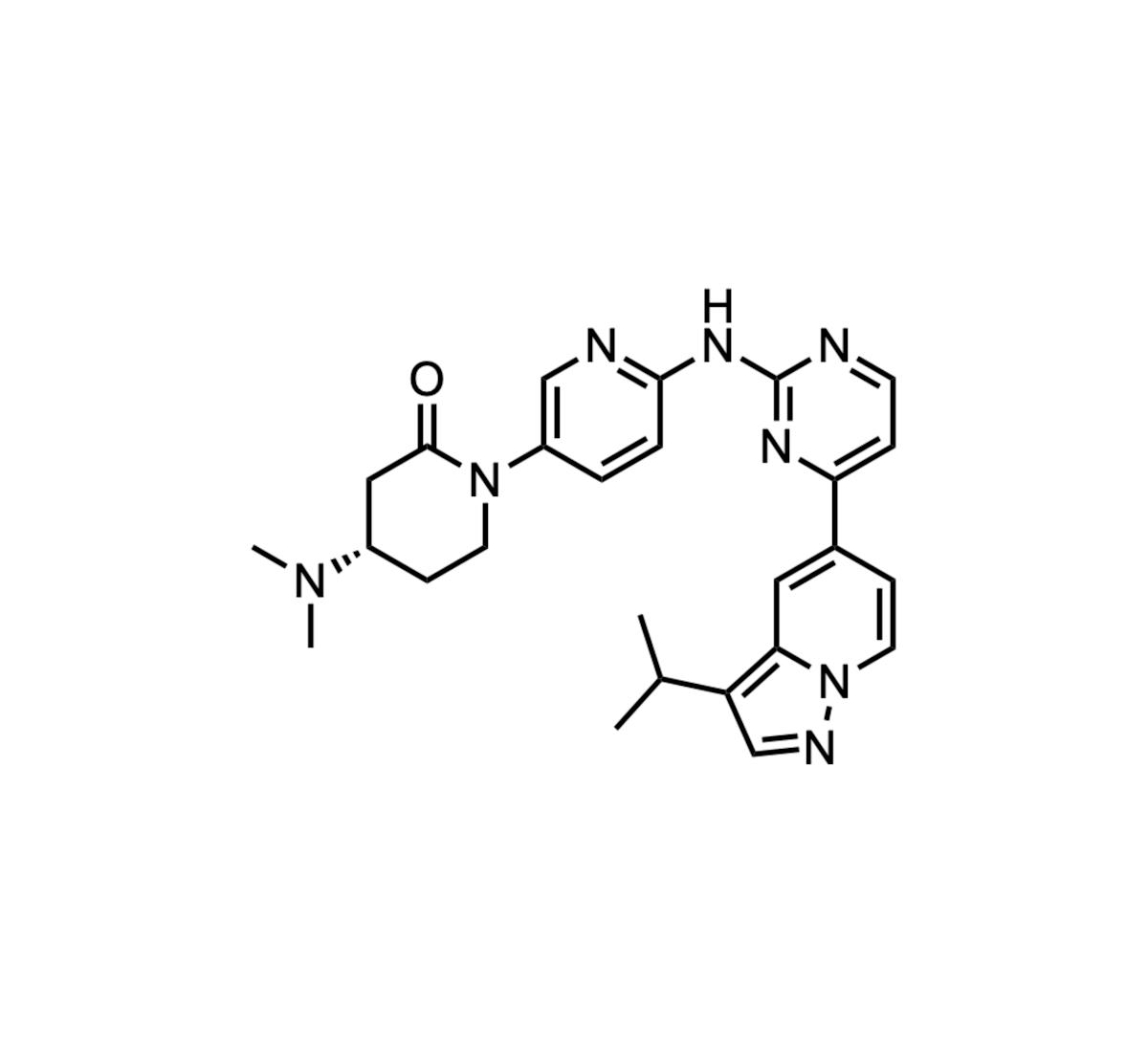
GDC-4198
Regor Therapeutics discovered GDC‑4198, an oral CDK4/2 inhibitor now owned by Genentech. The drug is in Phase 1/2 trials for advanced solid tumors, with a focus on breast cancer. It combines low‑nanomolar CDK4 potency with comparable CDK2 activity and roughly 20‑fold...
Mundipharma Announces Positive Topline Results From Phase III ReSPECT Trial Assessing ▼REZZAYO® (Rezafungin) for the Prophylaxis of Invasive Fungal Diseases...
Mundipharma reported that its Phase III ReSPECT trial met the primary endpoint, showing rezafungin (REZZAYO®) was non‑inferior to standard antifungal regimens for fungal‑free survival at Day 90 (60.7% vs 59.0%). The once‑weekly echinocandin demonstrated a comparable safety profile with fewer drug‑drug interactions...

Asembia AXS26: What the Rise in CGTs Means for the Supply Chain
Cardinal Health’s VP Joel Wayment told Pharmaceutical Commerce that the surge in cell and gene therapies is forcing a redesign of the supply chain toward reusable, multi‑use shipping systems and geographically closed packaging models. These changes aim to cut waste,...
Fragments vs DsbA: Towards a Chemical Probe
Researchers targeting the bacterial oxidoreductase DsbA—a key virulence factor—have advanced fragment‑based efforts toward a chemical probe. Initial screens identified fragments binding a shallow groove and a hidden cryptic pocket, but affinities were modest (~150 µM). By designing molecules that extend beyond...

Can We Truly Deliver Gene Therapies to Patients Without Solving the Cost and Scale Challenges of AAV Production?
Gene‑therapy developers are confronting a manufacturing bottleneck as adeno‑associated virus (AAV) vectors must be produced at commercial scale. FUJIFILM Biotechnologies proposes 2,000‑liter single‑use bioreactors combined with an integrated CDMO model to boost yield, cut cost per dose, and preserve product...
Evidence for MLKL to Be Important in Hematopoietic Stem Cell Aging
Researchers have identified the RIPK3‑MLKL signaling axis as a central driver of hematopoietic stem cell (HSC) aging. Activated MLKL accumulates in HSC mitochondria, impairing self‑renewal and lymphoid differentiation without causing necroptotic cell death. The study links multiple stress responses—such as...
Reviewing the Inability of Anti-Amyloid Immunotherapies to Affect Alzheimer's Disease
A recent Cochrane meta‑analysis of ten anti‑amyloid monoclonal antibodies—including aducanumab, lecanemab and donanemab—shows only trivial cognitive gains and modest functional improvement in patients with mild Alzheimer’s disease, despite clear plaque clearance. The studies also reveal an elevated risk of amyloid‑related...

New Bird Flu Vaccine Shows Promise Against Multiple H5N1 Strains
University of Nebraska–Lincoln researchers have unveiled a nanodisc‑based vaccine that protects mice and dairy calves from multiple H5N1 bird‑flu strains. The platform uses a prime‑boost regimen combining intramuscular and intranasal delivery to generate systemic and mucosal immunity. Preclinical trials showed...

Precipio (PRPO): Advanced Blood Diagnostics
Precipio (PRPO) showcased its dual‑model strategy in a live MicroCapClub interview, highlighting a low‑cost BCR‑ABL assay developed for under $100,000—far cheaper than the $5‑10 million industry norm. The company reported a 30% revenue increase in 2025, achieving its first profitability inflection...

Understanding How Plants Pause and Restart Growth Can Help Develop Climate‑resilient Crops
Researchers identified the genetic switch that lets plants pause growth during cold, salt or drought stress and resume within roughly 24 hours once conditions improve. Using Arabidopsis roots as a rapid assay, they pinpointed Cyclin‑dependent Kinase A;1 (CDKA;1) as a...
What Your CD3 T Cell Engager Is Missing
CD3 T‑cell engagers have become a cornerstone of bispecific immunotherapy, linking T cells to cancer cells via the CD3 receptor. The article argues that despite their success, these molecules often provide only the primary activation signal, neglecting a critical secondary...

The Cure for Death Means Billionaires Will Live Forever—And Be Rich Forever
U.S. billionaires enjoy a dramatically higher life expectancy, with 20% living past 80 compared to just 3.8% of the general population. Their longevity stems from access to premium healthcare, personal trainers, and cutting‑edge nutrition. Meanwhile, leaders like Putin and Xi...

Free Radicals Podcast (Longevity / Biotech Oriented)
Kexin Huang, the a16z‑backed founder of Pho, argues that biology is entering an "Agentic Biology" era where AI agents orchestrate research rather than merely analyze data. His Integrated Biology Environment (IBE), embodied in the Biomni platform, acts like an IDE...

Free Radicals Podcast (Longevity / Biotech Oriented)
Nathan Cheng argues that aging remains untreated due to a coordination failure rooted in cultural "deathism," despite roughly 100,000 daily deaths from age‑related diseases. He highlights a stark $5 B versus $100 B+ funding gap between longevity and cancer research, underscoring the...