
Architecting Life: Authoring the Future of Species with Dr. Adrian Woolfson
Dr. Adrian Woolfson argues that DNA must be treated as a programmable engineering material, enabling the design of living systems from houses to organs. By decoding DNA's generative grammar, humanity could author genomes and potentially rewrite its own code, ushering in unprecedented longevity and disease eradication. Woolfson stresses that this power brings profound ethical risks, demanding a moral framework for safe and responsible deployment. The discussion builds on his decade‑long advocacy for exponential technologies that could transform scarcity into abundance.

Agenus Names BAP Pharma as Exclusive Global Partner for BOT+BAL Access Programs
Agenus appointed BAP Pharma as its exclusive global partner to manage early‑access programs for the botensilimab‑balstilimab (BOT+BAL) immunotherapy combo. The collaboration will handle France’s government‑reimbursed Autorisation d’Accès Compassionnel (AAC) pathway and paid named‑patient programs in several other markets. Agenus has...

Charles River Highlights Effectiveness of VCGs in Toxicology
Charles River Laboratories published a retrospective analysis of 20 nonclinical toxicology studies that replaced traditional concurrent control groups with virtual control groups (VCGs). The review found 100% concordance in No Observed Adverse Effect Level (NOAEL) determinations and demonstrated up to...
Star Therapeutics Receives FDA Rare Pediatric Disease and Breakthrough Therapy Designations for VGA039 in Von Willebrand Disease Prophylaxis
Star Therapeutics announced that the FDA has granted both rare pediatric disease and Breakthrough Therapy designations to its lead candidate VGA039, a monoclonal antibody aimed at preventing bleeding in von Willebrand disease (VWD). The designations support the ongoing Phase 3 VIVID‑6 study,...

Simulations Plus Announces Collaboration with Lonza and U.S. FDA to Advance Predictive Frameworks for Complex Oral Drug Products
Simulations Plus announced a funded collaboration with CDMO Lonza and the U.S. FDA to create a mechanistic, predictive framework for amorphous solid dispersion (ASD) oral drugs. The partnership will combine Lonza's advanced in‑vitro dissolution testing with Simulations Plus' GastroPlus and...

From PhD to Staff Data Scientist in Biotech
Jordan works as a Staff Data Scientist at Recursion in Fort Collins, where he integrates computational workflows with experimental biology to accelerate drug discovery. His daily duties include building high‑dimensional analysis pipelines, collaborating with biologists on experiment design, and presenting...

The AI Drug Discovery Capital Stack in 2026: Who Has Raised the Most, Why Their Technical Approaches Actually Differ, and...
The essay maps AI‑driven drug‑discovery firms’ capital stacks as of April 2026, highlighting that Eikon, Xaira, Isomorphic Labs and Recursion sit at the top of disclosed funding. It separates the sector into four technical lanes—structure foundation models, generative chemistry, phenomics/perturbational biology,...
Patients Stay Cancer-Free Three Years After Clinical Trial
A phase II trial at UCL tested pembrolizumab as neoadjuvant therapy for 32 patients with stage II‑III MMR‑deficient/MSI‑high bowel cancer. After up to nine weeks of immunotherapy before surgery, 59% showed no detectable tumor and none experienced recurrence over a median...

The Gut Microbe in INDY Related Longevity in Flies
Researchers investigated how the longevity‑associated Indy gene influences the gut microbiome in Drosophila. Indy heterozygous flies displayed lower bacterial load and greater microbial diversity as they aged, while still achieving lifespan extension even in germ‑free conditions. The study linked Indy...
PEPITEM as a Potential Therapy for Autoimmune Arthritis
Researchers at the University of Birmingham have identified a decline in the anti‑inflammatory peptide PEPITEM as a key driver of worsening inflammatory arthritis with age. Laboratory tests showed that adding synthetic PEPITEM restores white‑blood‑cell responsiveness to adiponectin in early‑stage rheumatoid...

Prostate Cancer - I’m Asking for some Specific Advice/Thoughts to Determine My Physical (Cell-Level Age) versus Chronological Age
The large TRAVERSE trial of about 5,200 hypogonadal men found no increase in prostate‑cancer incidence with testosterone replacement therapy—12 cases on treatment versus 11 on placebo—though the study’s 33‑month follow‑up and 60% dropout limit statistical power. Mechanistically, androgen‑receptor saturation occurs...

Dr. Kaeberlein's Optispan Podcast Series - Rapamycin and More
The Optispan podcast hosted by Dr. Kaeberlein outlines a translational protocol for 3‑hydroxyanthranilic acid (3HAA), a mouse‑tested longevity molecule. Using FDA BSA scaling, the human equivalent dose (HED) is calculated at roughly 1.1 g per day for a 70‑kg adult. Safety...
AACR26 Innovative Early Stage Developments to Watch Out For
At the AACR annual meeting in San Diego, four cutting‑edge oncology programs were showcased in a single session. Each candidate is at or just beyond the threshold for first‑in‑human trials, spanning bispecific antibodies, RNA‑based therapeutics, CRISPR‑edited cell therapies, and novel...

Major Antineoplastic Mechanisms of Combination Ivermectin-Mebendazole
Recent preclinical analyses highlight the anticancer potential of combining ivermectin and mebendazole, two antiparasitic drugs repurposed for oncology. Ivermectin suppresses proliferative signaling pathways such as Wnt/β‑catenin, Akt/mTOR, and STAT3, while mebendazole disrupts microtubule polymerization, inducing G2/M arrest. Together they inhibit...

AACR San Diego 2026: New Drugs on the Horizon
The AACR 2026 Annual Meeting in San Diego unveiled 11 first‑time disclosed oncology candidates spanning small‑molecule degraders, bispecific antibodies, T‑cell engagers and ADCs. Highlights include NEO‑811, a CRBN‑mediated molecular glue targeting HIF‑1β for VHL‑deficient renal cancer, and AZD8359, a STEAP2‑directed T‑cell...

What Patients Value in Data Reuse for Oncology Research: A Multi-Stakeholder Qualitative Study to Inform the European Health Data Space...
A new multi‑stakeholder qualitative study examined what oncology patients value when their health data are reused for research. Conducted in Belgium, the research highlights patients’ demand for transparent consent, robust data security, and clear societal benefits. Findings are intended to...

Psychedelics Go Mainstream
President Donald Trump issued an executive order to speed up research and access to psychedelic therapies, allocating $50 million in federal funding and instructing regulators to dismantle long‑standing barriers. The move validates a growing investment thesis that the psychedelic sector will...
ATF5 as a Point of Tradeoff in Muscle Mass versus Muscle Quality
Researchers discovered that deleting the transcription factor ATF5 in mice prevents the typical age‑related loss of skeletal muscle mass, but this comes at the cost of reduced muscle quality and endurance. ATF5‑deficient mice showed lower activation of mitochondrial quality‑control proteins,...

Mitrix Bio as an Example of the Trend Towards Alternative Paths to Initial Human Data
Mitrix Bio reported preliminary Phase 1 safety results for large‑dose mitochondrial infusions, showing no immediate adverse effects in two older participants. The company simultaneously opened Right‑to‑Try clinics in Dallas, Newport Beach and Palm Beach, offering the experimental therapy under a patient‑driven model. Its...

GLP-1 May Only Be the Beginning, Not the End of the Story
Researchers led by Richard DiMarchi and Matthias Tschöp published a paper in Molecular Metabolism showing that triple agonist retatrutide can drive weight loss even when GLP‑1 signaling is blocked. Their preclinical work demonstrates that co‑activating GIP and glucagon receptors produces...
Fully Defined 3D Culture Substrate for Cancer Research
AMSBIO announced that its fully defined MatriMix 511 extracellular matrix enables patient‑derived colorectal cancer cells to form robust 3D organoids. In a Kyoto University study, the organoids preserved stage‑specific tumor biology and expressed metastatic markers, outperforming alternative matrices. MatriMix’s composition...

MHT and Mortality: Reassuring Data From a New Study
A new nationwide Danish cohort study of over 800,000 women examined long‑term menopausal hormone therapy (MHT) and mortality. Researchers tracked participants for an average of 14.3 years, including more than 100,000 MHT users, some with ten or more years of...

Rogan Tipped the Scales on Psychedelic Research in the US
Joe Rogan appeared at the White House as President Trump signed an executive order to fast‑track psychedelic research and clinical trials. The directive directs federal agencies to accelerate approval pathways for psychedelic therapeutics aimed at PTSD, veteran mental health, and...

What to Know Before Buying GLP-1 Drugs Online
The FDA warns that many online ads for GLP‑1 weight‑loss drugs promote compounded versions that lack the rigorous testing of brand‑name products like Ozempic, Wegovy, and Mounjaro. These compounded formulations can vary in concentration, contain unapproved ingredients, and have been...

Joe Rogan Reveals Trump IMMEDIATELY Offered Him FDA Approval for Unbelievable New Treatment...
President Donald Trump issued an executive order accelerating federal research on ibogaine, a Schedule I psychedelic, to create FDA pathways for veteran mental‑health treatment. The order follows claims that ibogaine can address depression, PTSD and substance abuse, with the FDA expected...

Dasatinib and Quercetin as Senolytic May Cause Brain Damage
A March 2026 PNAS study shows that the senolytic combo dasatinib and quercetin (D+Q) triggers demyelination in the corpus callosum of aged mice. The researchers used intermittent oral doses of 5 mg/kg dasatinib and 50 mg/kg quercetin, identical to regimens linked to...

The Longevity Effects of Reduced IGF-1 Signaling Depend on the Stability of the Mitochondrial Genome (Paper April 2026)
The study shows that reducing IGF‑1 signaling via Pappa loss does not extend lifespan in Polg D257A mutator mice, which harbor unstable mitochondrial DNA. While Pappa deletion improves several health metrics—splenomegaly, anemia, inflammation, muscle and cardiac function—the longevity benefit seen in...

A Closer Look at Body Composition in Obesity Drug Trials
A new systematic review of 36 randomized controlled trials, published in Annals of Internal Medicine, reveals that obesity medications often cause a sizable loss of lean mass. In 65% of drug trials, more than a quarter of total weight loss...

Is VC6TF the OSK Reversal Cocktail?
Researchers at the Sinclair Lab have identified a five‑molecule mix called VC6TF that chemically mimics the OSK (Oct4, Sox2, Klf4) gene‑therapy cocktail used to reset cellular age. The core of the “three‑chemical” version discussed by Dr. Sinclair includes CHIR‑99021, RepSox...

Weekly Reads: Prasad Successor, Sammy Hagar Stem Cells, More on OSK Trial, Finnish Speaker Needed
Vinay Prasad is set to leave his role as head of the FDA's Center for Biologics Evaluation and Research (CBER within two weeks, prompting speculation about the agency’s future direction. A new flexible oversight framework for rare‑disease gene and cell therapies...

The Next Chemical Cage Has a Beautiful Door
President Biden signed an executive order fast‑tracking psychedelic drugs, granting the FDA priority vouchers for serotonin‑2A agonists. The move follows a public ceremony with Joe Rogan, RFK Jr., and FDA Commissioner Marty Makary, signaling a shift from traditional plant‑based use to pharmaceutical...
HSS Studies Contribute Early Evidence to Help Guide Emerging Perioperative Considerations for Patients Using GLP‑1 Medications
At the ASRA annual meeting, Hospital for Special Surgery researchers presented two studies on peri‑operative management of patients taking GLP‑1 agonists. A multicenter ultrasound assessment of 354 elective surgery patients found no statistically significant difference in full‑stomach incidence between GLP‑1...

Compass Pathways Commends White House Executive Order to Accelerate Research and Access for Psychedelic Treatments
Compass Pathways welcomed the White House Executive Order aimed at speeding up research and access to psychedelic therapies for serious mental illness. The biotech highlighted its COMP360 synthetic psilocybin, which has delivered statistically significant results in two Phase 3 trials for...

INVITATION: Join Me for a Live Session on Personalised Drug Repurposing in Cancer Care
Amanda, the Metabolic Nutritionist, will co‑host a live conversation with author and metabolic oncology researcher Travis Christofferson on drug repurposing in precision oncology. The event, organized by Astron Health, will explore how approved medications can be matched to cancer patients...

Zai Lab Presents New Preclinical Data Suggesting ZL-1503, an IL-13/IL-31Rα Bispecific Antibody, Provides Rapid Itch Relief and Reduction in Inflammation...
Zai Lab unveiled preclinical data for ZL-1503, a bispecific antibody that blocks IL-13 and IL-31Rα, showing sustained itch suppression and inflammation reduction for up to 112 days after a single dose. The study demonstrated dose‑dependent efficacy across skin, lung, nasal...

GPT-Rosalind Lands: What OpenAI’s First Domain-Specific Life Sciences Model, the Codex Life Sciences Plugin & the Trusted Access Program Actually...
OpenAI unveiled GPT‑Rosalind on April 16, its first domain‑specific model built for life‑sciences tasks such as biochemistry, genomics, and protein engineering. Access is limited to a Trusted‑Access program that currently includes Amgen, Moderna, Thermo Fisher Scientific, the Allen Institute and...
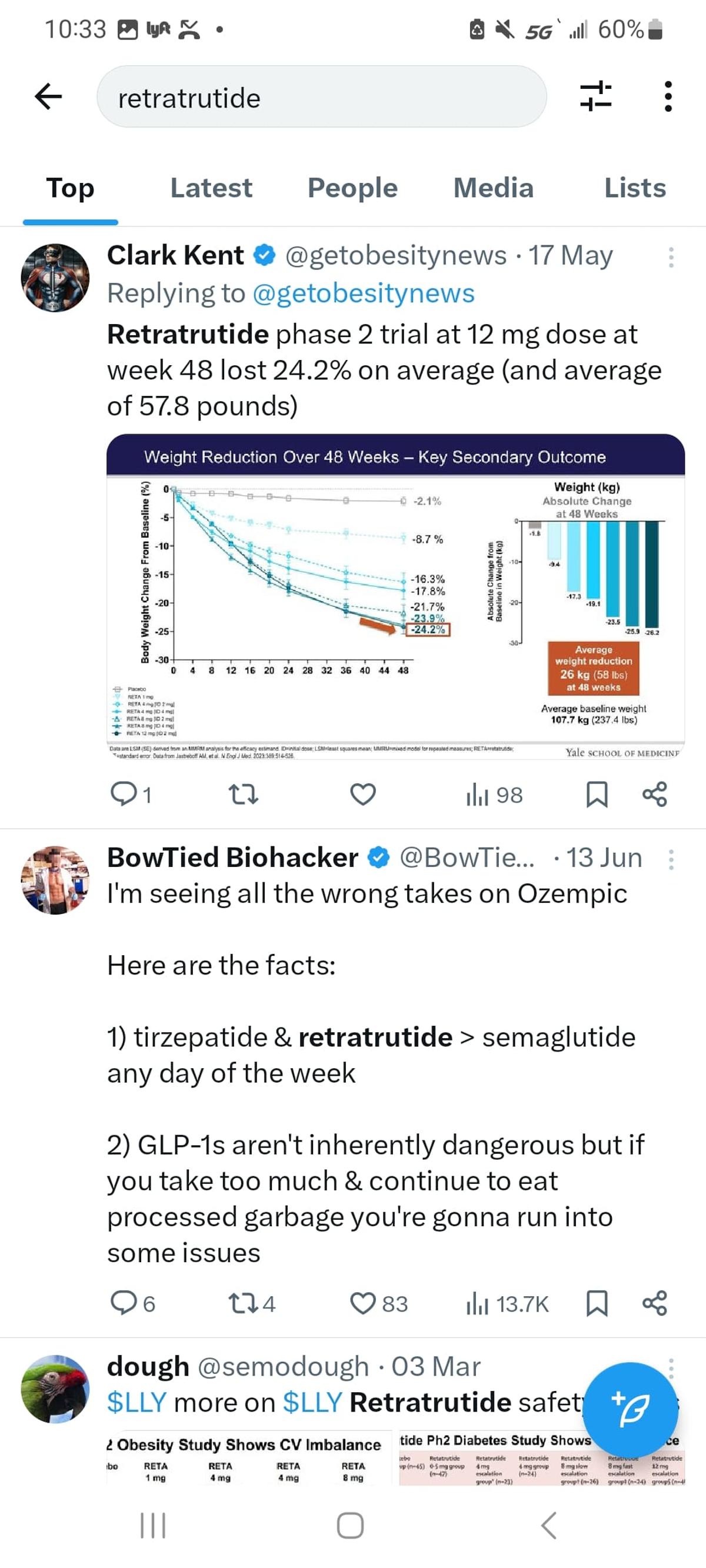
Retatrutide - Possibly Better than Semaglutide B/C Lower Nausea/Side Effect Profile, but Higher Heart Rate
Retatrutide, a triple‑agonist peptide, is generating buzz for delivering dramatic weight loss at doses of 8‑12 mg, rivaling semaglutide while causing fewer nausea complaints. Early users note that appetite suppression diminishes after several weeks, yet the drug continues to support weight‑maintenance...

Hazel Szeto, SS-31 Peptide, the World's First FDA-Approved Mitochondria-Targeted Drug (Longevity Summit, 2025)
SS‑31 (elamipretide), the first FDA‑approved mitochondria‑targeted drug, is being promoted on TikTok as a vision enhancer, despite its approval only for a rare mitochondrial disease. Clinical data for age‑related macular degeneration (AMD) show the drug failed to meet endpoints, with...

Imeglimin. A New and Novel Drug Thats Better than Metformin
Imeglimin, a mitochondrial‑targeted antidiabetic agent, is emerging as a novel alternative to metformin. Pre‑clinical studies show it restores mitochondrial function in skeletal muscle and pancreatic β‑cells, enhancing insulin secretion and reducing oxidative stress. Phase‑3 trials report HbA1c reductions of 0.6‑0.9%...

Agenus Reports Phase II Data Demonstrating Immune Reprogramming and Durable Survival with Botensilimab, Balstilimab and agenT-797 in PD-1 Refractory Gastroesophageal...
Agenus presented Phase II data showing that a combination of botensilimab, balstilimab and the allo‑iNKT cell therapy agenT‑797 achieved a 77% disease‑control rate in PD‑1‑refractory gastroesophageal adenocarcinoma. Patients who received an induction cycle of agenT‑797 before the full regimen experienced median...

SAGA Diagnostics to Present New Pathlight™ MRD Data for Ovarian and Metastatic Breast Cancer at AACR 2026
SAGA Diagnostics will present two abstracts at AACR 2026 showcasing its Pathlight™ structural‑variant‑based circulating tumor DNA (ctDNA) assay. In metastatic breast cancer, the test achieved a 77% detection rate, with ultrasensitive reads predicting therapeutic response and preceding radiologic progression. In...

Zai Lab Presents New Data Demonstrating Zocilurtatug Pelitecan (Zoci) Induces Rapid and Robust Intracranial Responses in Small Cell Lung Cancer...
Zai Lab reported that its DLL3‑targeting ADC zocilurtatug pelitecan (Zoci) generated a 53.7% confirmed intracranial objective response rate (iORR) in extensive‑stage small‑cell lung cancer (ES‑SCLC) patients with brain metastases, rising to 62.5% at the 1.6 mg/kg dose. In a separate cohort...
New HPV Test Kit for At-Home Use Secures FDA Clearance
The FDA has cleared Waters Corporation’s Onclarity HPV Self-Collection Kit, allowing individuals to collect a vaginal sample at home and send it to a lab for analysis with the BD Onclarity HPV Assay. The kit detects high‑risk HPV strains and...
Bringing AI-Driven Protein-Design Tools to Biologists Everywhere
OpenProtein.AI has launched a no‑code, web‑based platform that gives biologists instant access to powerful protein‑language models and design tools. The suite includes the PoET transformer and its newer PoET‑2 version, which delivers higher accuracy while using a fraction of the...

Anti-Amyloid Alzheimer’s Drugs Show Limited Benefit in Cochrane Review
A Cochrane review of 17 randomized trials involving 20,342 participants found that anti‑amyloid antibodies deliver little to no clinically meaningful improvement in memory, cognition, or dementia severity after roughly 18 months of treatment. The analysis also identified a markedly higher...

The Interventions Testing Program Shows that Another Eleven Compounds Do Not Slow Aging in Mice
The National Institute on Aging’s Interventions Testing Program evaluated eleven small‑molecule and supplement candidates—including astaxanthin, meclizine, mitoglitazone, pioglitazone, α‑ketoglutarate, mifepristone, methotrexate, and an atorvastatin‑telmisartan combo—in genetically heterogeneous UM‑HET3 mice and found none extended lifespan. Earlier studies that suggested modest benefits...

Industry Report: Advanced Therapy Market May Triple by 2030
Cardinal Health’s 2026 Advanced Therapy Report projects that the U.S. advanced therapy market will nearly triple, rising from 61 approved cell and gene treatments today to around 180 by 2030. The study, based on input from 160 physicians and administrators,...

Morgan Stanley Updates Bicycle Therapeutics Plc (BCYC) Outlook Amid Pipeline Refocus
Morgan Stanley lowered its price target for Bicycle Therapeutics plc (BCYC) to $12 from $13, while maintaining an Equal Weight rating, reflecting a revised valuation after the company refocused its pipeline on BT5528 and next‑generation Bicycle conjugate programs. The shift...

Jefferies Sees Reduced Upside for Arbutus Biopharma Corporation (ABUS) on Higher Appeal Probability
Jefferies lowered its price target for Arbutus Biopharma (ABUS) from $7.00 to $5.50 while keeping a Buy rating, citing a higher probability that Moderna will win its appeal in a patent dispute. The appeal outcome affects a potential $1.3 billion contingent...

Mizuho Sees Strong Risk-Reward in Corbus Pharmaceuticals Holdings, Inc. (CRBP) Near Cash Value
Mizuho Securities raised its price target on Corbus Pharmaceuticals (CRBP) to $40, maintaining an Outperform rating after the company’s fourth‑quarter results. The firm highlighted that CRBP is trading near its cash balance, which it says caps downside risk. Mizuho expects...