
Blood as the Mirror of Aging
Recent research positions blood as both a diagnostic mirror and a therapeutic lever for aging. Multi‑omics studies show plasma proteins, metabolites, and extracellular vesicles reflect chronological and organ‑specific age, while heterochronic parabiosis and young plasma transfers demonstrate that youthful circulation can rejuvenate tissue function in animal models. Emerging interventions such as plasma dilution, therapeutic plasma exchange, and targeted factor modulation aim to remove pro‑aging signals and amplify rejuvenating components. The field is rapidly moving from mechanistic insight toward clinical applications targeting healthspan and neurodegeneration.
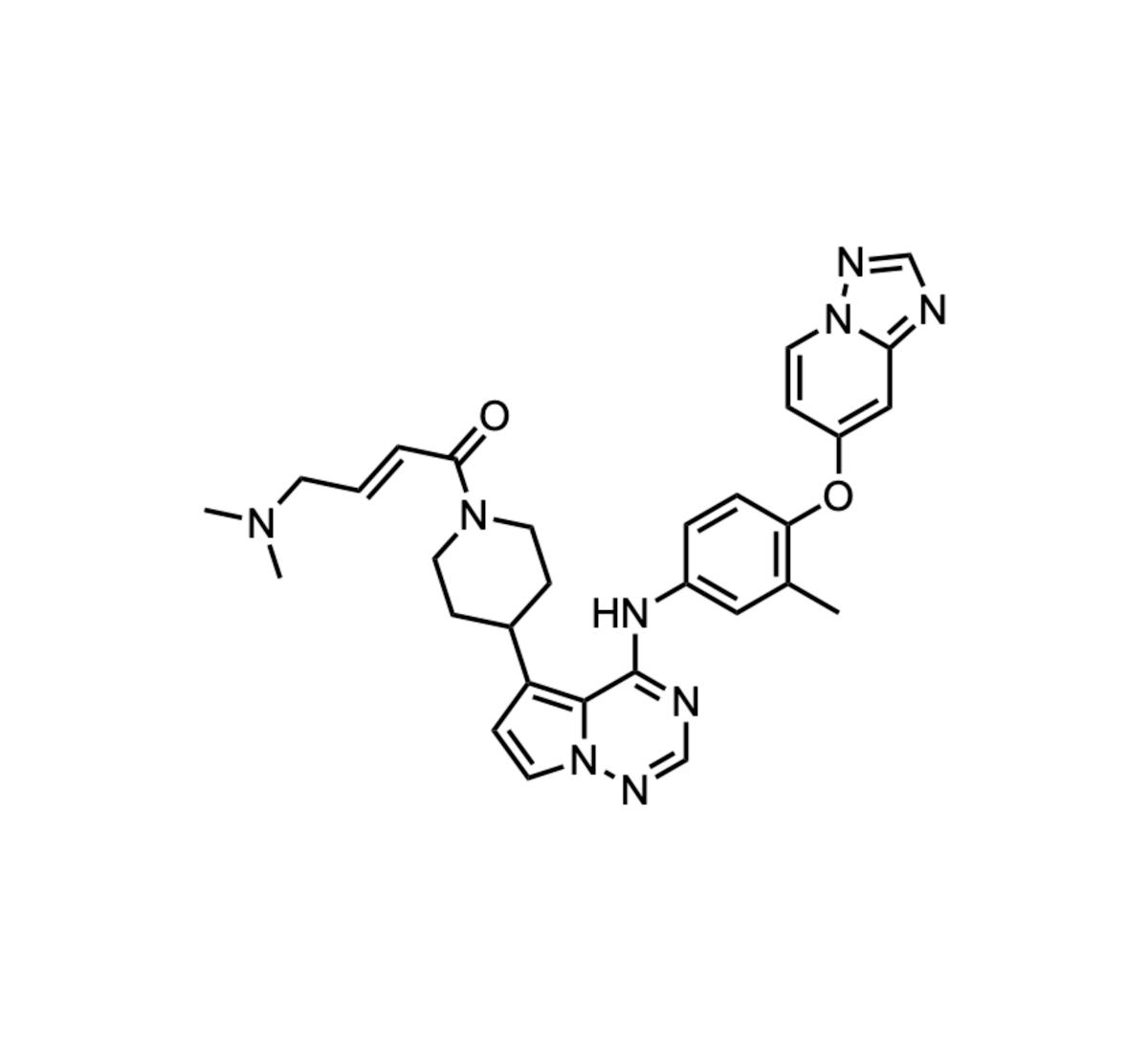
IAM1363
Iambic Therapeutics of San Diego announced the initiation of a Phase 1 clinical trial for an oral covalent inhibitor targeting HER2‑mutant cancers. The molecule, identified through an AI‑guided high‑throughput screening campaign, binds irreversibly to the mutant HER2 kinase domain. Preclinical data...

(Not) Getting Misled by Crystal Structures Part 6: Low Ligand Occupancies
A recent study re‑refined roughly 10,000 protein‑ligand structures from the PDB and found that while only 10% originally reported ligand occupancies at or below 0.9, re‑refinement raised that figure to 35%. Fragment‑sized ligands (<300 Da) were especially prone to occupancy loss,...

Module 4, Section 2: All About Assays
The Module 4, Section 2 briefing provides a concise overview of modern assay platforms used in early‑stage drug discovery. It references key literature on PRMT5 fragment‑based screening that produced the MRTX1719 candidate, as well as thermal‑shift, surface plasmon resonance (SPR), and polymerase...

Drug Side Effects Are Often the Main Effects
The article argues that drug side effects are not peripheral accidents but integral parts of a medication’s primary pharmacological action. It explains that the same biochemical pathways that deliver therapeutic benefits also generate adverse outcomes, using aspirin, blood‑pressure agents, and...
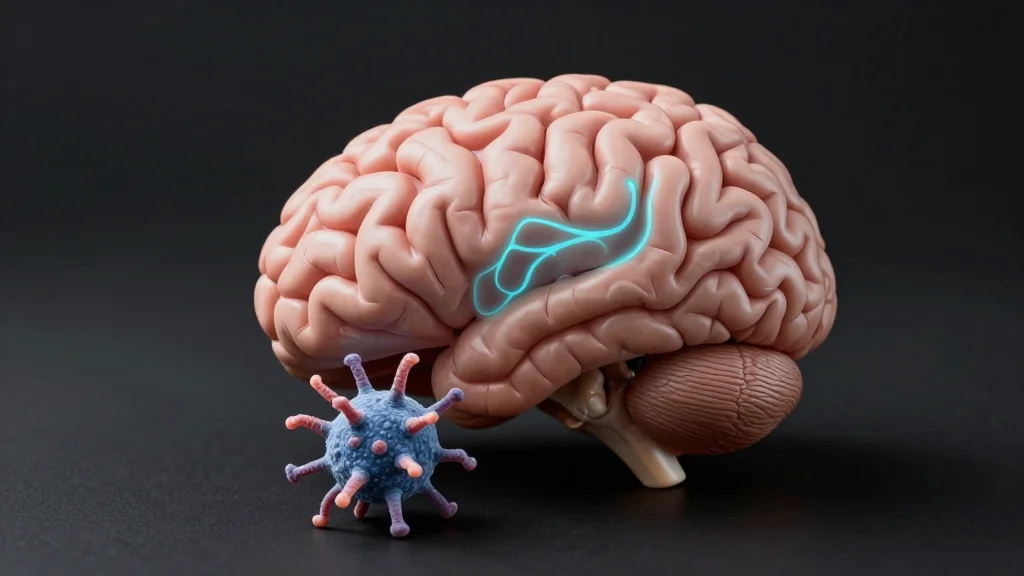
A Mechanism Linking Protein Aggregation to STING Activation and Inflammation in the Aging Brain
Researchers identified S‑nitrosylation of the immune sensor STING at cysteine‑148 as a key driver of neuroinflammation in Alzheimer’s disease. The modified protein, SNO‑STING, was abundant in human Alzheimer’s brains, cultured microglia, and mouse models. Blocking this chemical change reduced microglial...

MAHA Vs. The FDA: Dredging up Old Anti-Regulation Revisionist History
A new essay in Science-Based Medicine denounces the “health‑freedom” narrative that seeks to dismantle the FDA. It traces the agency’s authority back to the 1962 Kefauver‑Harris amendment, which linked drug approval to rigorous safety and efficacy trials. The author dismantles...

Viewpoint: How ‘Health Care Guru’ Joe Rogan Circumvented the FDA’s Skepticism on Psychedelics
Joe Rogan directly messaged President Donald Trump about the therapeutic potential of psychedelics, prompting the president to sign an executive order that fast‑tracks FDA review of these drugs. The order creates a priority‑voucher system that accelerates approvals for psychedelic manufacturers,...

Hidden Ingredient in GLP-1 Tablets Raises New Gut Health Questions
Oral semaglutide tablets rely on the absorption enhancer SNAC (salcaprozate sodium) to cross the stomach lining, but only 0.4%‑1% of the drug reaches the bloodstream. A 21‑day rat study published in the Journal of Controlled Release found that the majority...
![Special Situation Setup: CVV Sum Of The Parts Mispricing + Biorem Q4 Management Briefing [Geowire Weekly No. 237]](/cdn-cgi/image/width=1200,quality=75,format=auto,fit=cover/https://geoinvesting.com/wp-content/uploads/2026/05/0503_thumbnail.png)
Special Situation Setup: CVV Sum Of The Parts Mispricing + Biorem Q4 Management Briefing [Geowire Weekly No. 237]
GeoInvesting’s weekly wrap‑up highlighted a special‑situations opportunity in CVV after a “blowout” quarter that may be mispriced under a sum‑of‑the‑parts analysis. The newsletter also featured Biorem Inc.’s Q4 2025 management briefing, where the CEO reported record backlog growth and stronger...

RESEARCH: NICLOSAMIDE in CANCER and Other Diseases - 2025 Review Paper From Henan, China
A 2025 review paper from Henan, China, evaluates niclosamide—a decades‑old anti‑parasitic—as a repurposed oncology agent. The analysis compiles pre‑clinical data across breast, lung, pancreatic and colorectal cancers, and highlights early‑phase clinical trials showing modest tumor responses. Researchers also discuss formulation...

FDA Closes the 503B Bulks Door on Semaglutide, Tirzepatide, and Liraglutide
On April 30, 2026 the FDA issued a proposal to exclude semaglutide, tirzepatide and liraglutide from the 503B Bulks List, arguing there is no clinical need for outsourcing facilities to compound these GLP‑1 drugs. The move follows the resolution of...

Rethinking Blood Thinners for Atrial Fibrillation Patients
At the American College of Cardiology meeting, a three‑year trial demonstrated that the Watchman left‑atrial‑appendage closure device provides stroke protection comparable to lifelong anticoagulation while causing far fewer bleeding events. The findings challenge the entrenched belief that atrial fibrillation patients...

After Heart Attack, Therapeutic Plasma Exchange (TPE) Rescues the Aging Heart
Researchers at UC Berkeley demonstrated that therapeutic plasma exchange (TPE) performed 24 hours after a heart attack can nearly reverse damage in aged mice, the equivalent of humans in their 60s. By replacing half of the plasma with saline‑albumin solution, the...
NomosLogic Founder Matt Hardy Launches Lyceum and Odyssey on Dendrite Lite
NomosLogic unveiled two consumer‑facing experiences, Lyceum and Odyssey, on its Dendrite Lite platform. Lyceum delivers a personalized genomic‑literacy quiz generated from a user’s own DNA, while Odyssey presents the genome as a seven‑chapter narrative. Both tools leverage NomosLogic’s Hardy Bridge...

BREAKING: 146 NEUROLOGICAL AND PSYCHIATRIC CDC/FDA SAFETY SIGNALS WERE BREACHED WITH COVID SHOTS
A recent Substack post by Nicolas Hulscher alleges that COVID‑19 mRNA vaccines breached 146 CDC/FDA safety signals, citing astronomical relative‑risk figures such as a 3,000‑fold increase in brain clots and a 7.4% national rate of cognitive disability. The author claims...
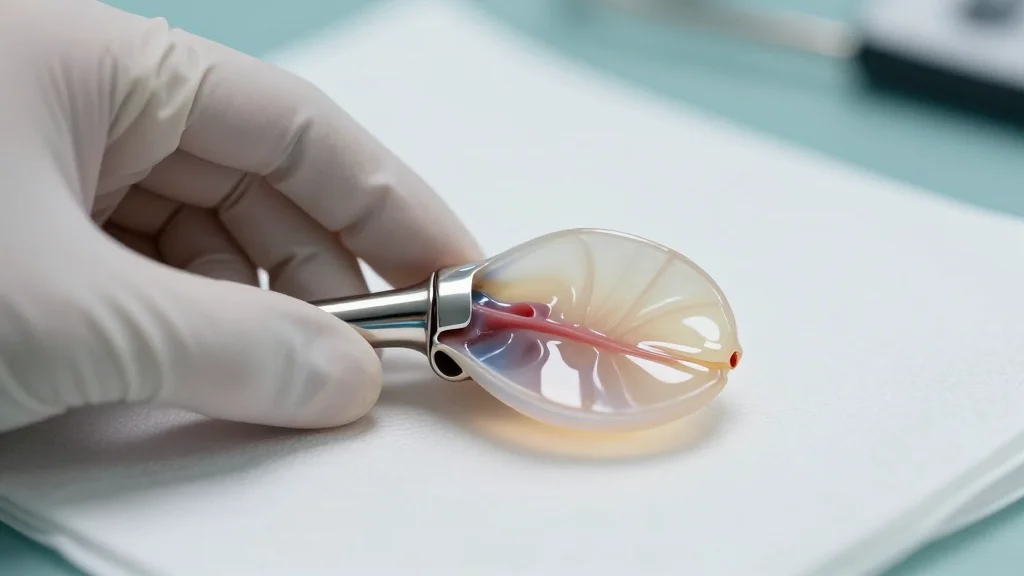
Ten-Year Pivotal Data Demonstrate Long-Term Durability of Edwards Lifesciences’ Resilia Tissue
Edwards Lifesciences released 10‑year results from the COMMENCE aortic trial, confirming that its RESILIA tissue surgical valves retain high durability. At ten years, 97.9% of patients were free from structural valve deterioration and 97.8% avoided reoperation. The data also show...

The Preclinical Signal in Routine Abdominal CT
A Mayo‑MD Anderson team unveiled REDMOD, a radiomics AI model that flags pancreatic ductal adenocarcinoma (PDAC) signals on routine abdominal CTs previously read as normal. The model delivers 73% sensitivity and 88% specificity, offering a median lead time of about...

Promising New Technique Uses Nanoparticles to Detect Pancreatic Cancer
Scientists at Oregon Health & Science University have unveiled a blood‑based assay that uses an electronic jolt to harvest tumor‑derived nanoparticles, achieving 97% accuracy in detecting pancreatic cancer. The technique, validated in a blinded study of 36 participants, outperforms the...

Monthly Features – April 2026
The LikelyStory blog’s April 2026 roundup spotlights two new releases: TK Thoits’s *SETTUP*, a fast‑paced medical thriller that pulls back the curtain on the multibillion‑dollar clinical‑trial industry, and Bear Pardun’s *The Knight’s Last Stand*, an epic fantasy where a lone...

Incyte Announces FDA Approval of Jakafi XR™ (Ruxolitinib) Extended-Release Tablets for the Treatment of Myelofibrosis, Polycythemia Vera and Graft-Versus-Host Disease
Incyte announced FDA approval of Jakafi XR, an extended‑release ruxolitinib tablet, for adult intermediate‑ or high‑risk myelofibrosis, hydroxyurea‑intolerant polycythemia vera, and steroid‑refractory acute or chronic graft‑versus‑host disease in patients 12 years and older. The 55 mg once‑daily tablet is bioequivalent to the 25 mg...
E-Scopics Announces New FDA Clearance for Hepatoscope® With Expanded Capabilities in Managing Liver Disease Complications, Advanced Elastography Imaging Features, and...
E‑Scopics announced FDA clearance for enhancements to its Hepatoscope® platform, adding a dedicated spleen exam workflow and upgraded 2D transient elastography (2DTE) that complies with IEC 63412‑1. The device now runs on macOS laptops with Apple M3 chips and integrates HL7 FHIR R4...

Katherine Szarama, PhD, Named Acting CBER Director: Who’s Next?
Vinay Prasad stepped down as director of the FDA's Center for Biologics Evaluation and Research (CBER), and Katherine Szarama, PhD, was named acting director. Szarama, a biologist with prior experience as CBER deputy director and stints at CMS, Emerson Collective...

Pharmaceutical Executive Daily: FDA Approves Auvelity
The FDA approved Axsome Therapeutics’ Auvelity for agitation associated with Alzheimer’s disease, marking the first non‑antipsychotic and only the second drug cleared for this indication. Earlier, the FDA’s Oncologic Drugs Advisory Committee gave AstraZeneca’s Truqap a 7‑1 favorable benefit‑risk vote,...

Update on Brad Stanfield's Rapamycin Clinical Study in NZ
Brad Stanfield’s New Zealand rapamycin trial enrolled older adults on a 12‑week protocol, with participants typically taking 6 mg every other week. The study measured functional outcomes such as the chair‑stand test, sparking debate over whether short‑term dosing can reveal longevity benefits. Commentators...

Asembia AXS26: How CGTs Are Reshaping Specialty Pharmacy
Cell and gene therapies (CGTs) are fundamentally changing the specialty pharmacy landscape, moving beyond traditional buy‑and‑bill models toward highly coordinated, patient‑specific workflows. The therapies demand specialized treatment‑center activities such as apheresis, conditioning, and strict cold‑chain logistics. High upfront costs create...
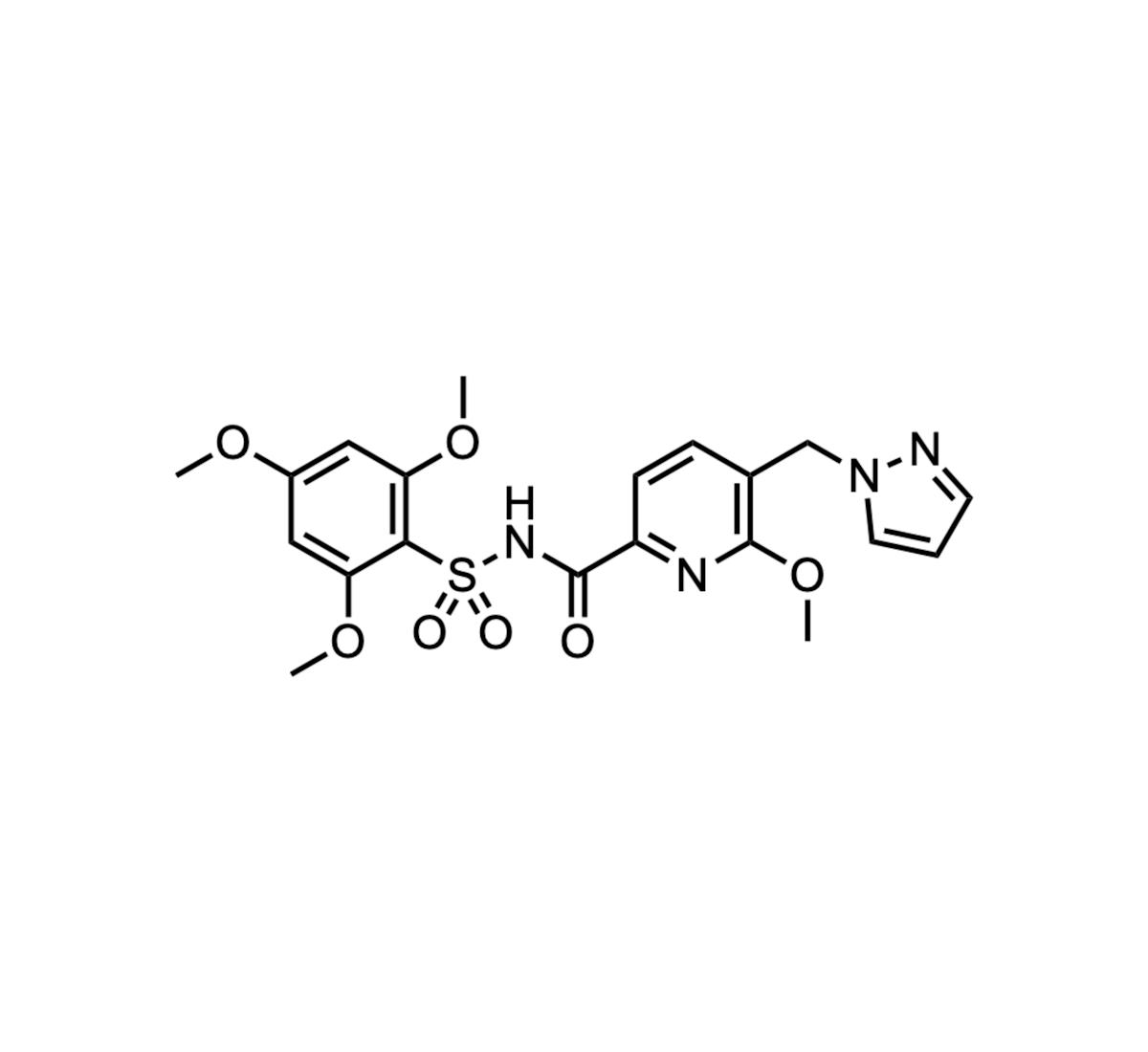
OP-3136
OP‑3136, a KAT6A‑selective inhibitor, entered Phase 1/2 trials for advanced hormone‑receptor‑positive breast cancer. The drug mimics the pyrophosphate of acetyl‑CoA using an acyl‑sulfonamide scaffold, delivering high specificity for the epigenetic writer KAT6A. Olema Pharmaceuticals is testing OP‑3136 in combination with SERDs...

Sydnexis to Present New Data From Phase 3 STAR Trial of SYD-101 at ARVO 2026 Annual Meeting
Sydnexis announced it will unveil new subgroup analysis data from the Phase 3 STAR trial of its low‑dose atropine eye drop SYD‑101 at the ARVO 2026 meeting in Denver. The analysis focuses on children with fast‑progressing myopia, a cohort that typically...

World Asthma Day 2026: New Anti-Inflammatory Drugs and More for Asthma Control
World Asthma Day on May 5 highlighted the persistent gap in access to anti‑inflammatory inhalers, a core theme of the Global Initiative for Asthma. The FDA approved AstraZeneca’s Breztri Aerosphere, the first single‑inhaler triple therapy that combines an inhaled corticosteroid, a...

New Semaglutide for Alcohol Use Disorder Trial Shows Big Drops in Drinking
A Lancet‑published, double‑blind, 26‑week trial found once‑weekly semaglutide markedly reduced alcohol consumption in participants with alcohol use disorder and obesity. Across primary drinking endpoints, the semaglutide arm showed statistically significant declines compared with placebo, despite both groups receiving identical cognitive‑behavioral...

Arrowhead Pharmaceuticals Receives TGA Approval of REDEMPLO® (Plozasiran) in Australia, Expanding Global Access for Patients with Familial Chylomicronemia Syndrome (FCS)
Arrowhead Pharmaceuticals announced that Australia’s Therapeutic Goods Administration approved REDEMPLO® (plozasiran), the first siRNA therapy for familial chylomicronemia syndrome (FCS) in the country. The drug, administered subcutaneously every three months, targets apoC‑III to dramatically lower triglycerides. In the Phase 3 PALISADE...

Friday Hope: H. Erinaceus (Lion’s Mane): A Mushroom Which May Help Those Suffering From Long COVID/Spike Disease/Injury
The post reviews pre‑clinical data showing that Hericium erinaceus (Lion’s Mane) suppresses NF‑κB, COX‑2 and iNOS while activating Nrf2, thereby reducing inflammation, oxidative stress and supporting neuronal health. Mouse studies demonstrate improved mitochondrial membrane potential, ATP production and antioxidant enzyme...

ARCHIMED to Acquire Esperion Therapeutics in a $1.1 Billion Deal
ARCHIMED has agreed to acquire Esperion Therapeutics for approximately $1.1 billion, offering shareholders $3.16 in cash per share—a 58% premium—and a contingent value right (CVR) that could add up to $100 million based on future sales milestones. The transaction values Esperion at...

Space-Efficient Cryogenic Sample Storage
Azenta Life Sciences introduced the CryoArc™ Pico Automated Storage System, a compact LN‑2 cryogenic platform that maintains samples at –190 °C. Designed for biobanking, clinical research, and cell‑gene therapy labs, the system integrates barcode‑driven software for sample tracking, chain‑of‑custody, and CFR 21...

Fecal Microbiota Transplantation Reduces MDM2 Expression and Risk of Liver Cancer
Researchers demonstrated that fecal microbiota transplantation (FMT) from young to old mice suppresses age‑related MDM2 overexpression and prevents liver cancer development. In the study, none of the FMT‑treated older mice developed tumors, whereas two of eight control mice did. Treated...

Zepbound Soars and Lilly Advises Patience on Foundayo
Eli Lilly reported a blockbuster first‑quarter, with revenue jumping 56% to $19.8 billion driven by soaring sales of its obesity drugs Zepbound and Mounjaro. Zepbound alone generated over $4 billion, up about 80% year‑over‑year, while Mounjaro nearly doubled to close to $9 billion. The...
New Genetic Discovery Could Spell This Aggressive Cancer’s Downfall
UCLA researchers uncovered a genetic weakness in small cell neuroendocrine carcinoma (SCNC) by creating prostate‑derived organoid models and running genome‑wide CRISPR screens. The screens identified the transcription factor E2F3 as a synthetic‑lethal partner of RB loss, and inhibiting E2F3 halted...

We Tried to Get Trump to Back Psychedelic Therapy — Here’s What Happened
The authors drafted a bipartisan policy blueprint to accelerate psychedelic‑based treatments, targeting veterans with PTSD, addiction and depression. After Joe Rogan shared the proposal, it amassed over 1.5 million views on X, prompting meetings with health‑policy influencer Calley Means and a...

Asembia ASX26: The Strain on the Development of Infrastructure for Sustainable C&G Therapy
Cell and gene therapies promise transformative treatments but remain hampered by steep costs across research, manufacturing, and delivery. At the Asembia ASX26 Summit, Aradigm Health CEO Will Shrank highlighted the fragmented U.S. payment landscape as a core barrier to sustainable...
Should Regulators Rely More on AI to Accelerate Drug Approvals?
Regulators, led by the FDA, are testing AI tools to speed drug approval processes. The agency’s pilot projects include AI‑driven real‑time monitoring of clinical trials and automated data review. Proponents argue AI can cut analysis time and uncover hidden patterns,...

FDA Moves to Speed Review Support for Psychedelic-Related Treatments
The FDA announced a new fast‑track mechanism for psychedelic‑based therapies, issuing Commissioner’s National Priority Vouchers to three programs targeting depression and PTSD with psilocybin or methylone. It also granted an IND for DemeRx NB to study noribogaine hydrochloride as a...

Varenicline
Varenicline (Chantix) received FDA approval in 2006 as a partial α4β2 nicotinic receptor agonist, offering a middle‑ground approach between nicotine replacement and bupropion. Its mechanism delivers enough receptor activation to ease cravings while antagonizing nicotine’s rewarding effects. The drug quickly...

Merck Announces First Dose in Phase 3 Study with Enpatoran for Lupus Patients with Active Skin Manifestations
Merck announced the first patient has been dosed in its global Phase 3 ELOWEN program, testing the oral TLR7/8 inhibitor enpatoran in lupus patients with active skin manifestations. The double‑blind, placebo‑controlled studies—ELOWEN‑1 and ELOWEN‑2—will enroll roughly 200 participants each across 266...

CGT Global Appoints Charlotte Ivancic to Board of Advisors, Strengthening Strategic Leadership in Cell and Gene Therapy Policy and Market...
CGT Global announced the appointment of Charlotte Ivancic to its Board of Advisors, bringing over 25 years of federal health policy and legislative experience. Ivancic, a partner at FGS Global and former senior aide to Senate Majority Leader Bill Frist,...

What We Know (and Don’t Know) About Peptide Safety
Peptide safety is neither automatically assured by mimicking endogenous hormones nor inherently hazardous because research is incomplete. The risk profile depends on the specific peptide, the depth of clinical evidence, product purity, and real‑world usage conditions. Early‑phase trials provide maximum...

The FDA Real Time Clinical Trial Announcement Quietly Dissolves Phase Gates, Breaks Biotech Capital Markets Plumbing, and Opens a Founder...
The FDA announced on April 28 that real‑time clinical trials (RTCTs) are now live, with AstraZeneca’s phase‑2 lymphoma study and Amgen’s phase‑1b small‑cell lung cancer trial streaming data through Paradigm Health. The agency claims a 20‑40 % reduction in development timelines, but...

Xtalks Featured Member: Parminder Singh Bhasin, Senior Director, Head of Analytical Research and Project Management, Intech Biopharm. Ltd
Parminder Singh Bhasin, Senior Director at Intech Biopharm Ltd., has been highlighted as an Xtalks Featured Community Member. With more than 26 years in pharmaceutical R&D, he oversees analytical research, method validation, and project management for complex generics, pMDI, nasal...

A Combination Treatment Is Claimed to Produce Sizable Life Extension in Aged Mice
Seragon funded a pre‑clinical trial of SRN‑901, a proprietary oral cocktail that blends urolithin A, quercetin, nicotinamide riboside, alpha‑lipoic acid and the company’s SRN‑820. In 18‑month‑old mice on a Western diet, the regimen extended median remaining lifespan by 33% and cut...

EBook: The Roles of Endpoint, Real-Time and Digital PCR in Molecular Research
The new BioTechniques eBook outlines how endpoint PCR, quantitative real‑time PCR (qPCR) and digital PCR (dPCR) complement each other in modern molecular research. It explains that endpoint PCR remains a cost‑effective tool for qualitative detection, qPCR adds fast and reproducible...

Thursday: Three Morning Takes
The New York Times revealed that biologists are using AI chatbots to research how to modify and release deadly pathogens, highlighting a growing bio‑security risk. In New York, communist mayor Zohran Mamdani announced a second‑home tax targeting hedge‑fund titan Ken...