
New Antidepressants: Tackling Treatment Resistant Depression
New rapid‑acting antidepressants are reshaping treatment for the roughly one‑third of patients with treatment‑resistant depression who do not benefit from SSRIs or SNRIs. FDA‑approved agents such as esketamine and the newer oral combo Auvelity provide relief within minutes to weeks, while psychedelic‑based therapies like psilocybin (COMP360) and LSD analogs are advancing through phase 3 trials. Next‑generation neuroplastogens, exemplified by Enveric’s EB‑003 and Elkedonia’s Elk1 inhibitor, aim to deliver oral, non‑hallucinogenic efficacy. An executive order signed in April 2026 is accelerating regulatory pathways, spurring investor interest and precision‑medicine research.

Novartis Reveals More Data Behind Pluvicto Expansion Bid
Novartas reported new PSMAddition trial data showing that adding its radioligand therapy Pluvicto to standard hormonal treatment cuts PSA progression by 58% in hormone‑sensitive metastatic prostate cancer. Deep PSA reductions below 0.2 ng/mL were achieved by 87.4% of patients versus 74.9%...
Amgen’s Rare Disease Drug Tavneos Tied to 20 Deaths in Japan
Amgen’s rare‑disease drug Tavneos has been linked to 20 deaths among roughly 8,500 Japanese patients, primarily due to vanishing bile duct syndrome, a severe form of drug‑induced liver injury. The Japanese distributor Kissei Pharmaceutical has warned doctors against initiating new...

Regeneron Inks a ~$2.3B Deal with Parabilis Medicines to Advance Antibody-Helicon Conjugates Across Multiple Therapeutic Areas
Regeneron has struck a strategic research collaboration with Parabilis Medicines to develop antibody‑Helicon conjugates (AHCs) across several therapeutic areas. The agreement provides Parabilis with $125 million, including $50 million upfront and a $75 million equity‑financing commitment, and makes it eligible for roughly $2.2 billion...

Sandoz Reports the EC Approval of Bysumlog and Dazparda (Biosimilars, Humalog and NovoRapid)
The European Commission has granted approval for Sandoz's biosimilar insulin pens Bysumlog (insulin lispro) and Dazparda (insulin aspart). Both products are authorized as prefilled pens for diabetes treatment in adults, children and patients as young as one year, demonstrating efficacy...
Regeneron Misses Again as Melanoma Combo Bows to Merck’s Keytruda in Phase 3
Regeneron reported that its investigational LAG‑3 antibody fianlimab, combined with the PD‑1 blocker Libtayo, failed to show statistically significant survival benefits versus Merck’s Keytruda in a Phase 3 first‑line melanoma trial. Median progression‑free survival was 11.5 months for the high‑dose combo...

Magic Mushrooms Could Be Effective Treatment for Cocaine Addiction, Study Shows
Researchers published a small clinical trial showing that a single dose of psilocybin increased the likelihood of abstaining from cocaine compared with a placebo. The study involved 19 participants receiving psilocybin and 17 receiving diphenhydramine, all of whom engaged in...

BIO Supports and Seeks Refinements to FDA’s Plausible Mechanism Framework
The FDA has issued draft guidance introducing a Plausible Mechanism Framework to evaluate safety and efficacy of individualized, disease‑targeted therapies when traditional trials are infeasible. The Biotechnology Innovation Organization (BIO) issued a supportive comment letter, praising the agency’s direction while...

Supercharging Immune Cells May Help Control HIV Long-Term
Scientists have repurposed CAR‑T cell therapy, originally used for cancer, to target HIV. In a small Phase 1 trial, two participants who received the engineered T cells remained off antiretroviral drugs with undetectable viral loads for nearly two years and one...

AstraZeneca Reports the US FDA Approval of Baxfendy (Baxdrostat) To Treat Hard-to-Control Hypertension
AstraZeneca’s Baxfendy (baxdrostat) received U.S. FDA approval for use with other antihypertensives in patients with uncontrolled or treatment‑resistant hypertension. The approval is based on the Phase III BaxHTN trial, which enrolled 796 participants and showed significant blood‑pressure reductions versus placebo. In...

The Future of Radiopharma Is Being Built by Nuclear Geeks in SLC
Nusco, a Salt Lake City physics firm, is building a 190,000‑sq‑ft radioisotope production facility featuring a novel ion‑source linear accelerator capable of producing up to 12 isotopes simultaneously. The plant, designed for 24/7 operation and earthquake resilience, aims to meet...

Japan’s MHLW Approves Boehringer’s Jascayd for IPF and PPF
Boehringer Ingelheim’s Jascayd (nerandomilast) has been approved by Japan’s Ministry of Health, Labour and Welfare for treating idiopathic pulmonary fibrosis (IPF) and progressive pulmonary fibrosis (PPF). It is the first phosphodiesterase 4B inhibitor with antifibrotic and immunomodulatory properties to receive such...

Lotte Biologics Expands Antibody Manufacturing Agreement with Ottimo Pharma
Lotte Biologics announced on May 18, 2026 an expanded collaboration with South Korean biotech Ottimo Pharma to manufacture its PD‑1/VEGFR2 dual‑paratopic antibody, OTP‑01. The agreement builds on a June‑2025 deal and adds commercial process development support at Lotte’s Syracuse Bio Campus in...

Genentech Reports FDA Approval of Tecentriq and Tecentriq Hybreza for Adjuvant Muscle-Invasive Bladder Cancer With ctDNA-Guided Treatment
Genentech’s Tecentriq and Tecentriq Hybreza received FDA approval as adjuvant therapies for adults with muscle‑invasive bladder cancer (MIBC) who show circulating tumor DNA (ctDNA) minimal residual disease after cystectomy. The approval is linked to Natera’s Signatera CDx assay, which was...

AstraZeneca and Daiichi Sankyo’s Enhertu Receives US FDA Approval in Early Breast Cancer for Two Indications
The U.S. FDA approved AstraZeneca and Daiichi Sankyo’s antibody‑drug conjugate Enhertu (trastuzumab deruxtecan) for two early‑stage HER2‑positive breast‑cancer indications. In the neoadjuvant setting, Enhertu combined with trastuzumab‑pertuzumab (THP) earned approval after the DESTINY‑Breast11 trial showed a 67.3% pathologic complete response...

Can Revolution’s 'Miracle' Pancreatic Cancer Drug Be Topped? Immuneering, Actuate Say Yes
Revolution Medicines reported Phase 3 data showing its oral RAS inhibitor daraxonrasib more than doubled median overall survival for advanced pancreatic cancer to 13.2 months versus 6.7 months on chemotherapy. The results triggered a 40 % share surge and will support global regulatory filings,...
4 Spaces Primed for the Next Wave of Gene Therapies
The FDA’s recent approval of Regeneron’s Otarmeni for hereditary hearing loss marks the first non‑oncology gene‑therapy clearance, underscoring a shift toward treating central‑nervous‑system, ophthalmic, cardiovascular and muscular disorders. Companies such as Lexeo, Lilly/AskBio, uniQure, REGENXBIO/AbbVie, Tenaya, Medera and Sarepta are...
This Startup Aims to Upend Biologic Drug Production With Implantable ‘Cell Factories’
Duracyte, a Rice‑University spin‑out, is developing an implantable "cell factory" that produces therapeutic proteins inside patients, eliminating the need for traditional biologic manufacturing and injection pens. The capsule‑sized device houses genetically engineered human cells, supplies them with nutrients from the...

A Revolutionary Cancer Treatment Could Transform Autoimmune Disease
Researchers are expanding CAR T cell therapy, originally a cancer breakthrough, to treat autoimmune diseases such as multiple sclerosis, lupus, and stiff‑person syndrome. Early trials, including a 2025 Nebraska study and a 2025 Kyverna trial of 26 stiff‑person patients, report functional...

Why J&J Thinks Its New Psoriasis Pill Could Be One of Its Biggest Drugs Ever
Johnson & Johnson’s newly approved oral psoriasis pill Icotyde targets the IL‑23 receptor, making it the first once‑daily tablet in a class dominated by injectables. The drug entered the market in March and logged roughly 1,500 prescriptions within its first...

Researchers Identify First Suite of Human Antibodies Against Measles Virus
NIH‑funded researchers have isolated and structurally mapped the first comprehensive set of human monoclonal antibodies against measles virus, revealing nine distinct epitopes on the H and F surface proteins. One antibody, 4F09, locked the fusion protein and cleared virus from...
![[Comment] Emerging Β-Lactam and Β-Lactamase Inhibitor Strategies for Complicated Urinary Tract Infections](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/9fed89640d11fbd33efb7e76f58daf8c.webp)
[Comment] Emerging Β-Lactam and Β-Lactamase Inhibitor Strategies for Complicated Urinary Tract Infections
Complicated urinary tract infections (cUTIs) and acute pyelonephritis remain leading causes of hospitalisation and antibiotic consumption worldwide. Rising rates of ESBL‑producing and carbapenem‑resistant Gram‑negative bacteria are eroding the efficacy of existing β‑lactam regimens. Recent phase‑3 data show that novel β‑lactam/β‑lactamase...

FDA Approves Two Separate Indications for Fam-Trastuzumab Deruxtecan-Nxki in HER2-Positive Early-Stage Breast Cancer
The FDA has approved fam‑trastuzumab deruxtecan‑nxki (Enhertu) for two new indications in HER2‑positive early‑stage breast cancer: a neoadjuvant regimen followed by taxane, trastuzumab and pertuzumab, and an adjuvant regimen for patients with residual invasive disease. In the DESTINY‑Breast11 neoadjuvant trial,...

The BioPharm Brief: Oncology Momentum, CAR-T Advances, Strategic Expansion
AstraZeneca’s exploratory POTOMAC trial showed that combining its checkpoint inhibitor Imfinzi with BCG lowered early recurrence risk in patients with high‑risk non‑muscle‑invasive bladder cancer. At ASGCT 2026, Imviva presented early remission data from an allogeneic CAR‑T platform targeting lupus, hinting at...
Antiviral Ensitrelvir Cuts Risk of COVID-19 in Household Contacts by Two-Thirds, Study Finds
A Phase III trial published in the New England Journal of Medicine shows that the oral antiviral ensitrelvir, given within 72 hours of an index case’s symptom onset, cuts the risk of symptomatic COVID‑19 in household contacts by roughly two‑thirds. The study...
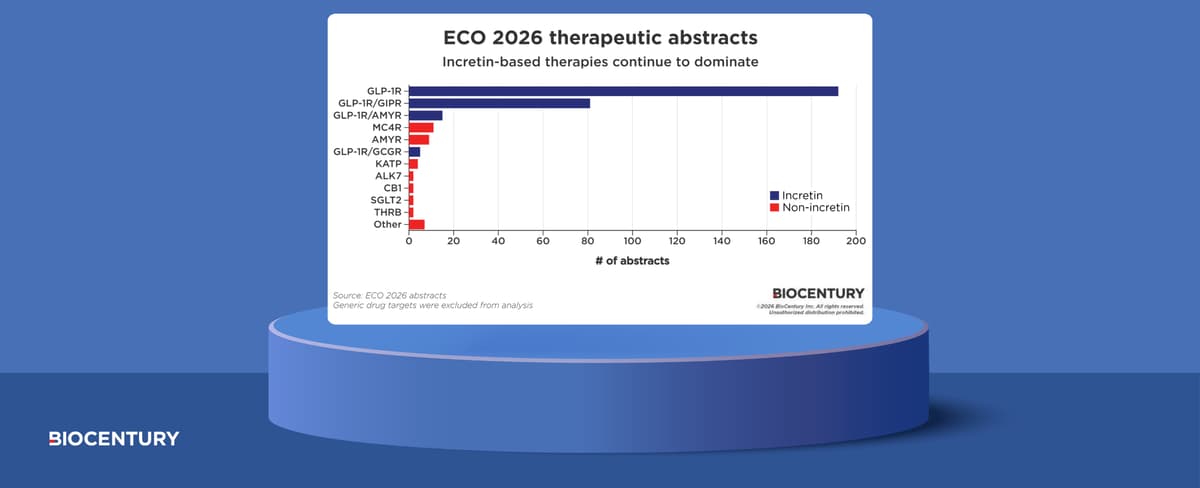
Incretins Continue to Dominate Obesity Conferences
Recent obesity conferences were dominated by incretin‑based therapies, with GLP‑1 receptor agonists such as semaglutide and the dual GIP/GLP‑1 agent tirzepatide taking center stage. New trial data presented showed weight‑loss results of 15‑20 percent, reinforcing the clinical potency of these...
Novel CAR T Cell Therapy Moves Into Clinical Studies
The University of Texas MD Anderson Cancer Center and its joint venture CTMC received FDA clearance to proceed with an Investigational New Drug application for a novel CAR‑T cell therapy targeting CD94‑positive T/NK‑cell lymphomas. The therapy will enter a Phase 1...
ECO 2026: Indirect Comparison Favours Wegovy Pill over Foundayo
At the 33rd European Congress on Obesity, Novo Nordisk presented a post‑hoc indirect comparison of oral semaglutide (Wegovy pill) versus orforglipron (Foundayo). Using simulated treatment comparison and matching‑adjusted indirect methods, the analysis showed Wegovy achieved roughly 3 percentage‑points greater weight...
New mRNA Therapy Destroys Cancer by Improving T Cell Priming
MIT researchers engineered lipid‑nanoparticle mRNA encoding NF‑κB‑inducing kinase (NIK) or IRF8 to reprogram immature dendritic cells into the cDC1 phenotype. The immune‑remodeling mRNAs amplified CD8+ T‑cell priming, leading to complete tumor regression in 70‑80% of mice across colorectal and metastatic...
Bacterial Energy Enzyme Reveals Dual-Trigger Sodium Pump Mechanism, Offering Antibiotic Clues
Researchers used modified AI tools and supercomputer‑scale molecular dynamics to capture the hidden motions of the bacterial sodium‑pumping enzyme Na⁺‑NQR. The simulations revealed a dual‑trigger mechanism where sodium binding and electron transfer drive conformational changes in subunits NqrD and NqrE,...

Psilocybin Offers Fast-Acting Alternative to Traditional Antidepressants
A phase‑2, double‑blind trial in Sweden found that a single 25 mg dose of psilocybin produced rapid antidepressant effects, cutting MADRS scores by an average of 9.7 points within eight days versus 2.4 points for an active placebo. The benefit persisted...

Translational Intelligence: The CDMO's Unspoken Role in the ATMP Journey
Advanced therapy medicinal product (ATMP) developers face a critical translation gap between research‑stage processes and GMP‑compliant manufacturing, where the process itself becomes the product. This gap, not a funding issue, creates structural risk that can halt programs and deter investors....

Candel Reports Prostate Cancer Drug's Long-Term Data Ahead of FDA Filing
Candel Therapeutics released long‑term follow‑up results for its investigational prostate cancer therapy, showing durable efficacy and a favorable safety profile. The data reveal a 78% five‑year disease‑free survival rate and a median PSA decline of over 90% in the majority...

Aardvark's Next Move After Clinical Hold; Alumis' Verdict on Acelyrin Asset
Aardvark Therapeutics announced that the FDA clinical hold on its rare‑disease HERO and OLE studies has been lifted, allowing the company to unblind the trial data. The unblinding will provide critical efficacy and safety readouts for the targeted condition. In...

CAR-T Therapy for Stiff Person Syndrome Nears Approval
Kyverna Therapeutics is close to securing regulatory approval for its CAR‑T cell therapy aimed at treating stiff person syndrome (SPS), a rare autoimmune neurological disorder with no approved drugs. The therapy uses engineered T cells to eliminate the B‑cell populations...

Resurge Therapeutics Presents Clinical Results on Its Intraprostatic Drug Elution (IPDE) Platform for Benign Prostatic Hyperplasia (BPH) at AUA 2026
Resurge Therapeutics presented data from two early‑phase studies of its intraprostatic drug elution (IPDE) platform for benign prostatic hyperplasia (BPH) at the AUA 2026 meeting. In the transperineal cohort of 20 patients, IPSS dropped 15 points and peak urinary flow...

#ASGCT26: A Zillow-Like Marketplace for Abandoned Gene Therapies Goes Live
Two nonprofit groups, the American Society of Gene & Cell Therapy and the Orphan Therapeutics Accelerator, unveiled a Zillow‑style online marketplace that lists abandoned cell and gene therapy assets. The platform, launched at the ASGCT26 conference, aggregates more than 30...

Arna Pharma and Slate Run Complete JV for New US Company
Arna Pharma and Slate Run Pharmaceuticals have completed a joint venture to create a U.S.-based specialty pharmaceutical company focused on branded products, 505(b)(2) medicines, and specialized generics. The partnership launched its first collaborative product, Aridol, and plans to add more...

NICE Backs Stomach Cancer, PAH Drugs for NHS Use
The UK’s health technology assessment body NICE has endorsed AstraZeneca’s immunotherapy Imfinzi for peri‑operative treatment of aggressive stomach and gastro‑oesophageal junction cancers, and approved Merck & Co.’s Winrevair as an add‑on therapy for pulmonary arterial hypertension (PAH). More than 1,500 patients in...
CAR T-Cell Therapy Bolstered by Stiffening up Cancer Cells First
Researchers have discovered that pre‑treating cancer cells to increase their stiffness markedly improves the effectiveness of CAR T‑cell therapy in mouse models of aggressive melanoma. The physical alteration of tumor mechanics enhances immune cell infiltration and tumor killing, offering a novel...

Kyowa Kirin Secures FDA Approval for Crysvita Dosing Update
Kyowa Kirin has secured FDA approval to expand Crysvita’s dosing regimen for adults with X‑linked hypophosphataemia (XLH). The update allows a 0.5 mg/kg dose up to 90 mg every two weeks, with the option to increase to 1 mg/kg after four weeks if serum...

Kyowa Kirin Secures FDA Approval for Crysvita Dosing Update
Kyowa Kirin has secured FDA approval for a dosing update to Crysvita (burosumab‑twza) for adults with X‑linked hypophosphataemia (XLH). The new regimen permits a 0.5 mg/kg dose every two weeks, with the option to increase to 1 mg/kg after four weeks if serum...

Biogen Reports P-II (CELIA) Trial Results on Diranersen in Early Alzheimer’s Disease
Biogen disclosed phase‑II (CELIA) results for intrathecal diranersen in 416 patients with early Alzheimer’s disease. The trial did not meet its primary dose‑response endpoint on CDR‑SB at week 76, but all dosing arms, especially 60 mg every‑four‑weeks, showed a slowing of clinical...

AstraZeneca’s Fasenra Receives the US FDA Approval for Hypereosinophilic Syndrome
The U.S. FDA has granted approval for AstraZeneca’s biologic Fasenra (benralizumab) to treat patients aged 12 and older with hypereosinophilic syndrome (HES) lacking a non‑hematologic secondary cause. Approval follows the Phase III NATRON trial, which enrolled 133 patients and compared 30 mg...

Racura Oncology Advances CPACS Trial to Next Dose Level
Racura Oncology (ASX:RAC) received Safety Review Committee clearance to double the RC220 dose from 40 mg/m² to 80 mg/m² for Cohort 2 of its CPAPS Phase 1 trial, pairing the drug with doxorubicin. The first three Cohort 1 patients showed no dose‑limiting toxicities, and all...

Biogen Finds Alzheimer’s Path for Tau ASO Despite Mixed Data
Biogen reported Phase 2 data for its tau‑targeting antisense oligonucleotide (ASO) in Alzheimer’s disease, showing a roughly 30% reduction in cerebrospinal fluid tau levels. Cognitive endpoints, however, delivered only modest, statistically non‑significant improvements versus placebo. The safety profile remained clean,...

Viagra Shows Promise as Potential Treatment to Halt Peyronie’s Disease
A Phase‑II trial of 133 men with acute Peyronie’s disease tested an off‑label combination of a PDE5 inhibitor (sildenafil or tadalafil) and the SERM tamoxifen. After three months, 43% of participants reported a meaningful reduction in penile curvature versus 15%...

60 Degrees Pharmaceuticals Inc (SXTP) Q1 2026 Earnings Call Transcript
United Therapeutics reported $782 million in Q1 2026 revenue, with Tyvaso contributing $458 million and showing 9% year‑over‑year growth despite seasonal headwinds. Phase III data revealed ralinepag delivering a threefold reduction in PAH disease progression and meeting all primary endpoints. The company announced a...

Citius Oncology Inc (CTOR) Q2 2026 Earnings Call Transcript
CTI BioPharma reported a cash balance of $71.9 million at the end of Q2 2021, while operating losses widened to $19.5 million as the company prepared for a potential U.S. launch of pacritinib. The FDA accepted the pacritinib NDA with priority review, setting...

BioCardia Inc (BCDA) Q1 2026 Earnings Call Transcript
BioCardia reported Q1 2026 results, noting reduced SG&A expenses, a modest net loss, and a cash balance of $5.3 million that extends its runway into 2026. The company secured FDA breakthrough device designation for its CARDIAMP cell therapy and received positive...