Today's Healthcare Pulse

FDA greenlights durvalumab combo for high‑risk bladder cancer
The FDA approved durvalumab (Imfinzi) combined with Bacillus Calmette‑Guerin for BCG‑naïve, high‑risk non‑muscle invasive bladder cancer. The POTOMAC trial enrolled 1,018 patients and showed a 32% reduction in disease recurrence risk (hazard ratio 0.68, p=0.015). Durvalumab is given at 1,500 mg IV every four weeks for up to 13 cycles.
Also developing:
By the numbers: Apogee Therapeutics raises $1.3B royalty financing

Protara Therapeutics Shows 55% 12‑Month Complete Response in BCG‑Naïve NMIBC Phase 2 Trial
Protara Therapeutics disclosed that its intravesical cell therapy TARA‑002 achieved a 55% complete response rate at 12 months in a fully enrolled BCG‑naïve cohort of the Phase 2 ADVANCED‑2 trial. The data, presented at the AUA 2026 meeting, underscore durable efficacy and a favorable safety profile, positioning TARA‑002 as a possible new standard for high‑risk non‑muscle‑invasive bladder cancer.
Thailand Classifies Hantavirus as Dangerous Communicable Disease, Mandates 42-Day Quarantine for High-Risk Contacts
Thailand has officially classified hantavirus as a dangerous communicable disease, making it the 14th illness on the nation’s legal list. The move triggers mandatory reporting within three hours and a 42‑day quarantine for high‑risk contacts. Authorities are tightening screening at...

United Therapeutics Corporation Announces FDA Clearance to Proceed with UHeart Xenotransplantation Clinical Trial
United Therapeutics received FDA clearance to begin the EXPRESS clinical trial of its UHeart xenotransplant, a pig‑derived heart with ten gene edits. The phase‑1/2/3 study will initially enroll up to two end‑stage heart‑failure patients, with safety and efficacy data reviewed...

Oregon's Healthcare Oversight Law Sparks Private‑Equity Concerns After Five Years of Inaction
Oregon’s 2021 law giving the state health department authority to block hospital, hospice and practice acquisitions has failed to block any transaction in five years, yet its mere existence is unsettling private‑equity firms. The law is credited with two high‑profile...

Isomorphic Labs Secures $2.1 B Series B to Accelerate AI Drug Discovery
Isomorphic Labs, the London‑based AI biotech founded by DeepMind’s Demis Hassabis, closed a $2.1 billion Series B round led by Thrive Capital. The capital will expand its IsoDDE platform and integrate AlphaFold 3, underscoring growing investor belief that AI can reshape pharmaceutical...
Rice and MD Anderson Unveil PrecisionView Handheld AI Microscope for Early Cancer Detection
Scientists from Rice University and MD Anderson Cancer Center introduced PrecisionView, a handheld AI‑powered endomicroscope that delivers cellular‑level images across a field of view five times larger and a depth of field eight times greater than conventional devices, aiming to...

CMS Finalizes Removal Of Standardized Plan Options, Non-Network Plans Addition In 2027 Exchange Rule
The Centers for Medicare & Medicaid Services (CMS) issued its 2027 health‑insurance exchange rule, stripping away the long‑standing standardized plan requirements and opening the marketplace to non‑network insurers. The rule also expands catastrophic‑only plans, giving consumers lower‑cost entry points. These...
FDA Grants First ctDNA-Guided Approval for Genentech's Tecentriq in Bladder Cancer
Genentech, part of Roche, secured FDA approval for Tecentriq (atezolizumab) and Tecentriq Hybreza as adjuvant treatments for muscle‑invasive bladder cancer patients with circulating tumor DNA residual disease. The decision, based on Phase III IMvigor011 data, shows a 36% drop in disease‑free...
![[Comment] Emerging Β-Lactam and Β-Lactamase Inhibitor Strategies for Complicated Urinary Tract Infections](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/9fed89640d11fbd33efb7e76f58daf8c.webp)
[Comment] Emerging Β-Lactam and Β-Lactamase Inhibitor Strategies for Complicated Urinary Tract Infections
Complicated urinary tract infections (cUTIs) and acute pyelonephritis remain leading causes of hospitalisation and antibiotic consumption worldwide. Rising rates of ESBL‑producing and carbapenem‑resistant Gram‑negative bacteria are eroding the efficacy of existing β‑lactam regimens. Recent phase‑3 data show that novel β‑lactam/β‑lactamase...
Agenus Announces Publication of Phase 1b Botensilimab and Balstilimab Data in Post-Immunotherapy Hepatocellular Carcinoma in Liver Cancer
Agenus published Phase 1b data on botensilimab plus balstilimab in 19 patients with treatment‑refractory hepatocellular carcinoma who had progressed after prior immunotherapy. The combination achieved a 17% objective response rate, a 50% clinical benefit rate at 18 weeks, median progression‑free survival...

CMS’ Health Care Fraud Clampdown Could Needlessly Burden Nursing Homes, With Advocates Urging a ‘Well-Calibrated’ Policy Instead
The Centers for Medicare & Medicaid Services (CMS) has launched the CRUSH (Comprehensive Regulations to Uncover Suspicious Healthcare) initiative to tighten fraud detection across Medicare and Medicaid, incorporating stricter provider screening, AI‑driven oversight, and faster claim deadlines. Nursing‑home providers and...

From Fax Machines to AI: Health Agencies Accelerate Modernization
Federal health leaders HRSA and CMS announced an aggressive modernization agenda aimed at replacing outdated transplant IT, automating prior‑authorization workflows, and expanding AI‑assisted care in rural areas. The organ procurement system will receive a unified, real‑time tracking platform, while CMS...

FDA Approves Two Separate Indications for Fam-Trastuzumab Deruxtecan-Nxki in HER2-Positive Early-Stage Breast Cancer
The FDA has approved fam‑trastuzumab deruxtecan‑nxki (Enhertu) for two new indications in HER2‑positive early‑stage breast cancer: a neoadjuvant regimen followed by taxane, trastuzumab and pertuzumab, and an adjuvant regimen for patients with residual invasive disease. In the DESTINY‑Breast11 neoadjuvant trial,...

The BioPharm Brief: Oncology Momentum, CAR-T Advances, Strategic Expansion
AstraZeneca’s exploratory POTOMAC trial showed that combining its checkpoint inhibitor Imfinzi with BCG lowered early recurrence risk in patients with high‑risk non‑muscle‑invasive bladder cancer. At ASGCT 2026, Imviva presented early remission data from an allogeneic CAR‑T platform targeting lupus, hinting at...

TRI-611
TRI‑611 is a CNS‑penetrant, CRBN‑mediated molecular‑glue degrader targeting ALK fusion proteins in ALK‑positive non‑small cell lung cancer (NSCLC). By recruiting a non‑G‑loop degron distal to the orthosteric site, it degrades ALK independently of the active site, sidestepping common tyrosine‑kinase inhibitor...

Top 20 Fastest Growing Pharma & Biotech Companies in the Americas for 2026
The Financial Times‑Statista 2026 ranking spotlights the 20 fastest‑growing pharma and biotech firms in the Americas, measuring compound annual growth rates from 2021‑2024. Companies such as Capricor Therapeutics, BioAtla, Ardelyx, Legend Biotech and Repare Therapeutics posted explosive revenue gains, driven...

Obesity Societies Issue First Joint Consensus on Incretin Drug Use
At the 2026 European Congress on Obesity in Istanbul, major obesity and dietitian societies released a unified consensus on the clinical use of incretin‑based therapies. The statement stresses integrated medical nutrition therapy, mental‑health screening, and a target 3:1 fat‑to‑lean‑mass loss...

Pfizer Secures EU Approval for HYMPAVZI Gene Therapy, Cuts Bleeds 93%
Pfizer has obtained European Commission marketing authorisation for HYMPAVZI (marstacimab) to treat hemophilia A or B patients with inhibitors aged 12 and older. The approval, covering all 27 EU members plus Iceland, Liechtenstein and Norway, is based on a Phase III...

METiS TechBio IPO Raises $270 M, Debuts as First Public AI‑Powered Drug Delivery Firm
METiS TechBio listed on the Hong Kong Stock Exchange, raising HK$2.11 billion ($269.5 million) and becoming the world’s first publicly listed AI‑powered drug‑delivery company. The offering was oversubscribed more than 6,900 times, propelling the stock 173% higher on debut and drawing a...

Optura Secures $17.5 Million Series A to Expand AI‑Powered ROAI™ Platform for Health Systems
Optura announced a $17.5 million Series A round led by Salesforce Ventures and Echo Health Ventures. The funding will be used to scale the company’s ROAI™ AI analytics platform across U.S. health systems, accelerating its push into predictive care and operational...

Marty Makary Steps Down as FDA Commissioner After Complaints, Adding to Regulatory Uncertainties
Marty Makary resigned as FDA commissioner after a turbulent 13‑month tenure marked by rapid regulatory experiments and political controversy. President Trump named deputy commissioner for food, Kyle Diamantas, a lawyer, as acting commissioner, placing a non‑physician at the agency’s helm. Makary’s...

CDC Says 41 Across U.S. Being Monitored for Hantavirus
The CDC announced that 41 people nationwide are being monitored for hantavirus after an outbreak on an Atlantic‑ocean cruise ship. The cohort includes 16 passengers quarantined in Omaha, Nebraska, two in Atlanta, Georgia, and others who returned home or were...

Transcript - Update on CDC's Hantavirus Response 5/15/2026
The CDC announced that no U.S. cases of the Andes‑virus hantavirus have been detected, but it is closely monitoring high‑risk individuals linked to the MV Hondius cruise ship and related flights. A 42‑day observation period has been mandated for...

Rehabilitation of Achilles Tendon Injuries Through Patient-Specific Scaffold Design Using FDM-Based 3D Printing of Thermoplastic Polyurethane (TPU)
Researchers demonstrated that thermoplastic polyurethane (TPU) scaffolds fabricated via fused deposition modeling (FDM) can be customized for Achilles tendon repair. Patient‑specific designs using AutoCAD and Cura produced spiral and lattice geometries with semi‑crystalline structure, confirmed by X‑ray diffraction. SEM revealed...
RAGE Implicated in Worsening Breast Cancer Mortality with Age
Georgetown researchers discovered that the receptor for advanced glycation end‑products (RAGE) fuels breast cancer metastasis in older hosts. In three mouse models of triple‑negative breast cancer, aged mice showed markedly more lung metastases, a surge that vanished when RAGE was...

New MRI Technique Maps 20-Plus Multiple Sclerosis Biomarkers in a Single Noncontrast Scan
Researchers at the University of Illinois Urbana‑Champaign have unveiled MRx, a multiparametric MRI method that maps more than 20 quantitative brain biomarkers in a single, non‑contrast scan. The AI‑driven acquisition and physics‑based processing deliver high‑resolution structural, physiological and molecular data...

FDA Approves First Interchangeable Biosimilars to Simponi and Simponi Aria (Golimumab) to Treat Rheumatoid Arthritis and Ulcerative Colitis
The FDA has approved Immgolis (golimumab‑sldi) and Immgolis Intri as interchangeable biosimilars to Janssen’s Simponi and Simponi Aria, respectively. Immgolis is delivered via a prefilled subcutaneous syringe, while Immgolis Intri is given as an intravenous infusion. Both are indicated for adults...
Antiviral Ensitrelvir Cuts Risk of COVID-19 in Household Contacts by Two-Thirds, Study Finds
A Phase III trial published in the New England Journal of Medicine shows that the oral antiviral ensitrelvir, given within 72 hours of an index case’s symptom onset, cuts the risk of symptomatic COVID‑19 in household contacts by roughly two‑thirds. The study...
Development of a Predictive Nomogram for Intraoperative Massive Transfusion in Patients with Placenta Accreta Spectrum: A Retrospective Cohort Study
A retrospective cohort of 87 placenta accreta spectrum (PAS) patients identified a 36.7% incidence of intraoperative massive transfusion. Using LASSO and multivariate logistic regression, the authors built a nomogram incorporating gestational bleeding, pre‑operative hemoglobin, and PAS subtype (increta/percreta vs. accreta)....

Cone Health Expands with New Employee Health Center
Cone Health announced a partnership with the Town of Mooresville, North Carolina, to open an employee health center serving municipal workers, retirees and their families. The clinic will deliver primary, preventive, and occupational care, including drug testing, screenings, and wellness...

Timely Peel Key to Better Outcomes in Epiretinal Membranes
Patients who receive epiretinal membrane (ERM) peeling within six months of retinal detachment repair achieve markedly better visual outcomes than those whose surgery is delayed. Dr. Vivek Chaturvedi presented data from a three‑part study—including a retrospective review of 55 eyes,...

Indiana Gets Federal OK for Hospital Assessment, Medicaid Payment Overhaul
Indiana received federal clearance to revamp its hospital assessment fee and Medicaid payment structure. The state will max assessments at the 6% federal ceiling, tying them to net patient revenue, with proceeds earmarked for the State Directed Payment (SDP) program....
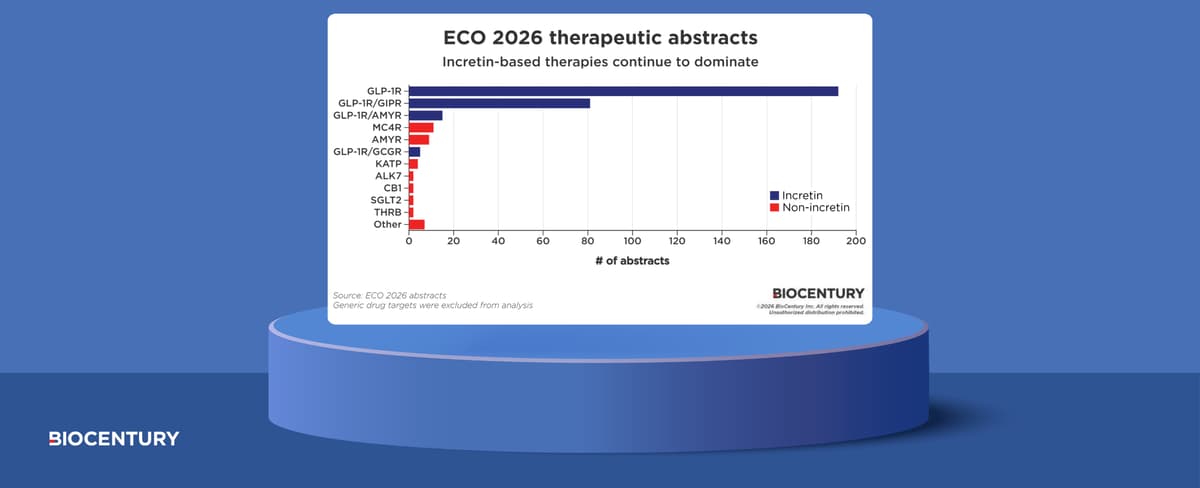
Incretins Continue to Dominate Obesity Conferences
Recent obesity conferences were dominated by incretin‑based therapies, with GLP‑1 receptor agonists such as semaglutide and the dual GIP/GLP‑1 agent tirzepatide taking center stage. New trial data presented showed weight‑loss results of 15‑20 percent, reinforcing the clinical potency of these...

Anomaly Insights Scores $17M Financing
Anomaly Insights, an AI‑powered payer intelligence startup, announced a $17 million financing round led by Sound Ventures. Existing backers Alumni Ventures, Link Ventures, Redesign Health and RRE Ventures also participated, bringing the company’s total capital raised to $34 million. The infusion is...

Changing Assault-Based STI Outcomes with Remote Care Delivery
RAINN’s Safe Access Program has partnered with Visby Medical to deliver at‑home STI testing and treatment in care‑desert regions across 17 states. Visby’s kits boast 98% accuracy and provide results in 30 minutes via a mobile app, then connect positive...

Pharmaceutical Executive Daily: FDA Approves AstraZeneca's Fasenra
The FDA has approved AstraZeneca’s Fasenra (benralizumab) for patients 12 and older with hypereosinophilic syndrome (HES), adding a rare‑disease indication to a drug already used for severe eosinophilic asthma. The approval rests on the Phase III Natron trial, which showed a...

Wearable Polygraph Tracks Deep-Body Stress Signals
Northwestern University engineers have created a sub‑8‑gram, wireless polygraph that adheres to the chest like a bandage and continuously records heart activity, respiration, sweat, blood flow and temperature. The system streams real‑time data to a smartphone, delivering an objective stress...

Heartflow Reports Significant Revenue Growth as CCTA Sees Wider Adoption
Heartflow announced a 41% year‑over‑year jump in first‑quarter 2026 total revenue to $52.6 million, with U.S. sales climbing 42% to $48.3 million. The surge is driven by expanding use of its AI‑powered FFR‑CT platform and the newly launched Plaque Analysis software. Inclusion...

Humans and Axolotls Share Key Regeneration Genes
Humans and Axolotls Share Many Regeneration-Related Genes Scientists have found that humans and axolotls share many of the same genes and molecular pathways involved in limb development and tissue regeneration. Axolotls activate these pathways as adults to...

MemorialCare Names Hospital CEO, Chief Medical Officer
MemorialCare Long Beach Medical Center appointed Gary Purushotham as CEO and Dr. Sapna Mehta, DO, as CMO, establishing a dyad leadership model. Purushotham previously led Detroit Medical Center Sinai‑Grace Hospital for nearly four years. Mehta, a Long Beach hospitalist for...
CRNA Pay by State, Adjusted for Cost of Living
The Bureau of Labor Statistics data shows certified registered nurse anesthetists (CRNAs) command some of the highest wages in health care, with Alaska leading nominal hourly pay at $149.57. When adjusted for each state’s 2025 cost‑of‑living index, West Virginia rises...

Biopharma M&A Maintains Strength Even as Large Deals Wane
Biopharma merger‑and‑acquisition activity remains robust, driven by companies scrambling to replace revenue lost to looming patent expiries. Deal volume rose about 15% year‑over‑year, pushing total transaction value to roughly $45 billion in the last twelve months. However, the market has seen...

Dear Health Care Provider Letters: Improving Communication of Important Safety Information
The FDA issued final Level 2 guidance (Docket FDA‑2010‑D‑0319) on Dear Health Care Provider (DHCP) letters, detailing when manufacturers should send them, the essential content, optimal organization, and formatting techniques. The guidance covers both drug and biologic products and applies to...

STAT+: Takeda Will Pay $13.6 Million to Settle Allegations It Paid Kickbacks to Doctors
Takeda Pharmaceuticals has agreed to pay $13.6 million to settle U.S. Department of Justice allegations that it provided illegal kickbacks to physicians. The DOJ says the company offered speaking fees and high‑end restaurant meals from January 2014 through October 2020 to boost prescriptions...
Novel CAR T Cell Therapy Moves Into Clinical Studies
The University of Texas MD Anderson Cancer Center and its joint venture CTMC received FDA clearance to proceed with an Investigational New Drug application for a novel CAR‑T cell therapy targeting CD94‑positive T/NK‑cell lymphomas. The therapy will enter a Phase 1...
Multi-Institutional Trial Explores New Lifeline for Advanced Prostate Patients
Researchers at MUSC and Emory reported Phase 2 results for opaganib, an oral drug targeting sphingolipid metabolism, added to standard androgen‑receptor therapies in metastatic castration‑resistant prostate cancer. In 66 patients, disease control at 16 weeks reached 15% with abiraterone and 9% with...

Despite 36 Years at Hospital, Surgeon Considered Contractor – Not Employee
A pioneering cardiac surgeon at Calgary’s Foothills Medical Centre, Teresa Prieur, spent 36 years wearing an Alberta Health Services (AHS) badge and using its resources, yet the Human Rights Tribunal of Alberta ruled she was a contractor, not an employee....

Amgen Expands Crackdown on What It Says Is Misuse of 340B Program
Amgen announced it will broaden its data‑submission requirements for pharmacies that dispense its products under the federal 340B drug discount program. The new policy mandates that participating pharmacies provide detailed claims data for every prescription, aiming to curb what Amgen...

Some Medicare Patients Can Now Get Free CBD
The Trump administration has launched a test program that will provide free cannabidiol (CBD) to thousands of Medicare patients. The initiative aims to collect real‑world evidence on whether CBD improves quality of life and reduces overall healthcare spending for seniors....

How AI Innovation Is Widening The Digital Health Divide
OpenAI unveiled ChatGPT Health, a generative AI tool that aggregates personal health data across portals. Early adopters like tech‑entrepreneur Sergei Polevikov reported hallucinated records and administrative friction, highlighting usability gaps. Interviews in Mobile, Alabama reveal that many low‑income users already rely...