Aspen Neuroscience Begins First Personalized Brain‑Repair Trial for Parkinson’s
Aspen Neuroscience announced the start of its ASPIRO Phase 1/2a trial, the first personalized brain‑repair therapy for Parkinson’s disease. Eight patients received autologous dopamine‑producing cells, reporting roughly two extra hours of “Good ON” time per day. The trial marks a shift from symptom‑management to cellular replacement in neurodegeneration.

Foundayo’s Liver Failure Blip Weighs Down Lilly Shares but Analysts Unconcerned
A single hepatic failure case linked to Eli Lilly’s new weight‑loss pill Foundayo appeared in the FDA’s adverse‑event database, prompting a brief sell‑off that pushed the stock down about 3% before rebounding to close up 0.48%. Lilly quickly investigated and...

CareFusion 213, LLC - 722729 - 04/30/2026
The FDA issued Warning Letter 320‑26‑72 to CareFusion 213, LLC, a BD subsidiary, citing extensive CGMP violations at its El Paso sterile drug facility. Inspectors documented over 2,500 customer complaints involving foreign particles, missing components, and compromised seals, and found the company’s investigations,...

Intelligent Remedies, Inc. - 681941 - 01/23/2026
The FDA issued a warning letter (CMS #681941) to Intelligent Remedies, Inc., alleging that ten of its products—including Pryenda, Athrombosyn, and VIRAsol—are marketed with therapeutic claims that classify them as new drugs. The agency says the claims violate the Federal Food,...

Ray’s Vitamins - 726694 - 04/24/2026
The FDA issued a warning letter to Ray’s Vitamins after discovering that its product “Yeicob Ácido Hialurónico” contains undeclared diclofenac and dexamethasone, classifying it as an unapproved new drug and a misbranded drug. The agency cited violations of sections 301(d), 505(a) and...

Twist Bioscience Q2 2026 Loss Widens as Synthetic DNA Demand Slows
Twist Bioscience Corp posted a second‑quarter 2026 net loss of $44.0 million, or $0.71 per share, compared with a $39.3 million loss a year ago. Revenue rose 19.3% to $110.7 million, but the widening loss highlights pressure on the synthetic‑DNA market as demand...

Sun Pharma to Acquire Organon for $12 Billion, Its Largest U.S. Deal Yet
Sun Pharmaceutical Industries Ltd. announced a $12 billion all‑cash acquisition of New Jersey‑based Organon & Co., the biggest U.S. outbound deal for an Indian drugmaker this year. The transaction pushes Sun Pharma into the specialty‑medicine arena and tests its ability to...

Insulet Launches Pivotal Trial of Fully Closed‑Loop Insulin System for Type 2 Diabetes
Insulet Corp. has begun a pivotal clinical trial, called Evolve, enrolling up to 350 adults with Type 2 diabetes across 40 U.S. sites. The study tests the company’s first fully closed‑loop insulin delivery system, a move intended to broaden automated insulin...
Phase 3 FUZION Data Show Guselkumab Benefit in Perianal Fistulizing Crohn Disease
Late‑breaking Phase 3 FUZION data presented at DDW 2026 show that guselkumab significantly improves combined fistula remission in adults with perianal fistulizing Crohn disease. At 24 weeks, remission rates were 28.3% with 100 mg every eight weeks and 27.0% with 200 mg every four weeks,...

From PhD to Regional Medical Director
Jill, a PhD‑trained scientist, now serves as Regional Medical Director (Medical Science Liaison) in Denver, overseeing scientific communication, clinical strategy, and KOL engagement for cancer therapies. Her path moved from postdoc to scientific project manager, then to MSL before attaining...
Do GLP-1 Drugs Like Ozempic Prevent Cancer?
GLP‑1 receptor agonists such as Ozempic, Wegovy, and the newly approved oral drug Foundayo have shown mixed evidence regarding cancer prevention. Some observational studies link them to lower obesity‑related cancer risk and improved survival, while other data show no association...

Japan’s Organoid Farm Scales Up Cultivated Meat Production & Teases New Facility
Japan’s Organoid Farm, a JGC Holdings subsidiary, completed a 200‑litre bioreactor demonstration that produced scaffold‑free cultivated beef using a patented bovine cell line capable of continuous division. The scaffold‑free suspension culture simplifies processing and cuts raw‑material and cleaning costs, providing...
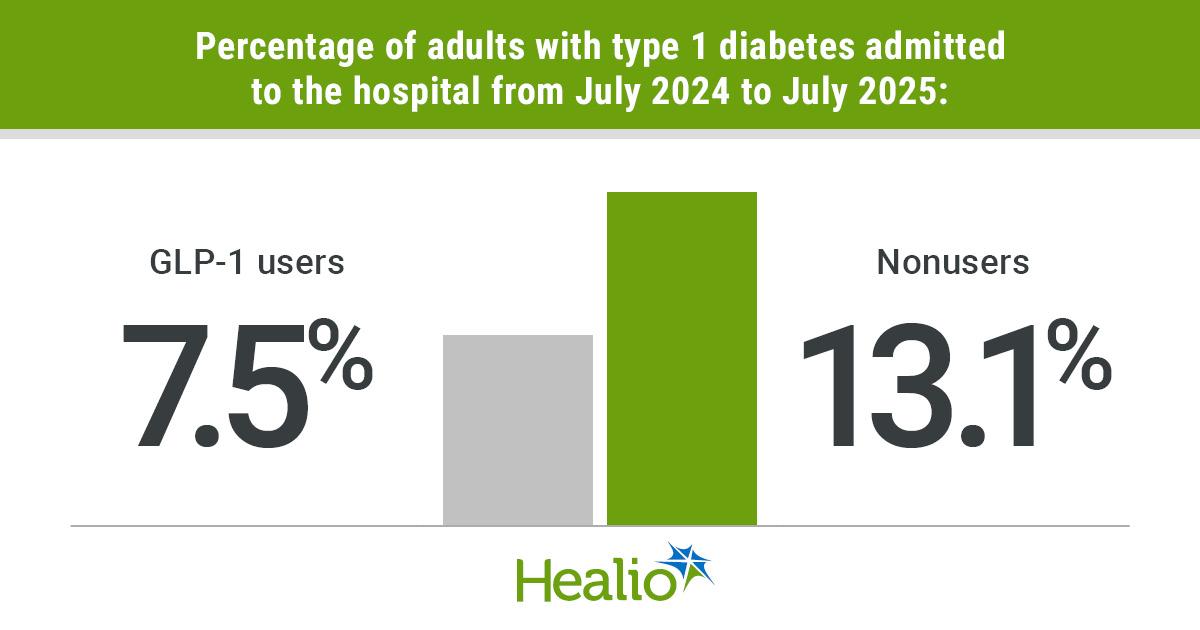
GLP-1s May Not Raise DKA, Pancreatitis Risk in Type 1 Diabetes
A single‑center study of 7,377 adults with type 1 diabetes found that none of the 255 patients using GLP‑1 receptor agonists were hospitalized for diabetic ketoacidosis (DKA) or pancreatitis over a one‑year period. Overall hospital admission rates were significantly lower for...

Disc Medicine Reports First Quarter 2026 Financial Results and Provides Business Update
Disc Medicine reported Q1 2026 results, highlighting the completion of enrollment in its Phase 3 APOLLO trial of bitopertin for erythropoietic protoporphyria, with topline data expected in Q4 2026. The company also announced that Phase 2 data for DISC‑0974 in myelofibrosis‑related anemia will be...

Genetic Variant Determines Individual GLP‑1 Drug Response
As a medical school professor, I get asked why GLP-1 drugs work miracles for some and barely budge for others. A new Nature paper from 23andMe gives part of the answer: your genes. Researchers ran a genome-wide association study in 27,885...

Odyssey Therapeutics (ODTX) IPO Deck
Odyssey Therapeutics, a clinical‑stage biopharma specializing in precision medicines for autoimmune and inflammatory disorders, unveiled its initial public offering deck in May 2026. The company aims to raise capital to advance its Phase 2‑tested pipeline, which targets conditions such as rheumatoid...
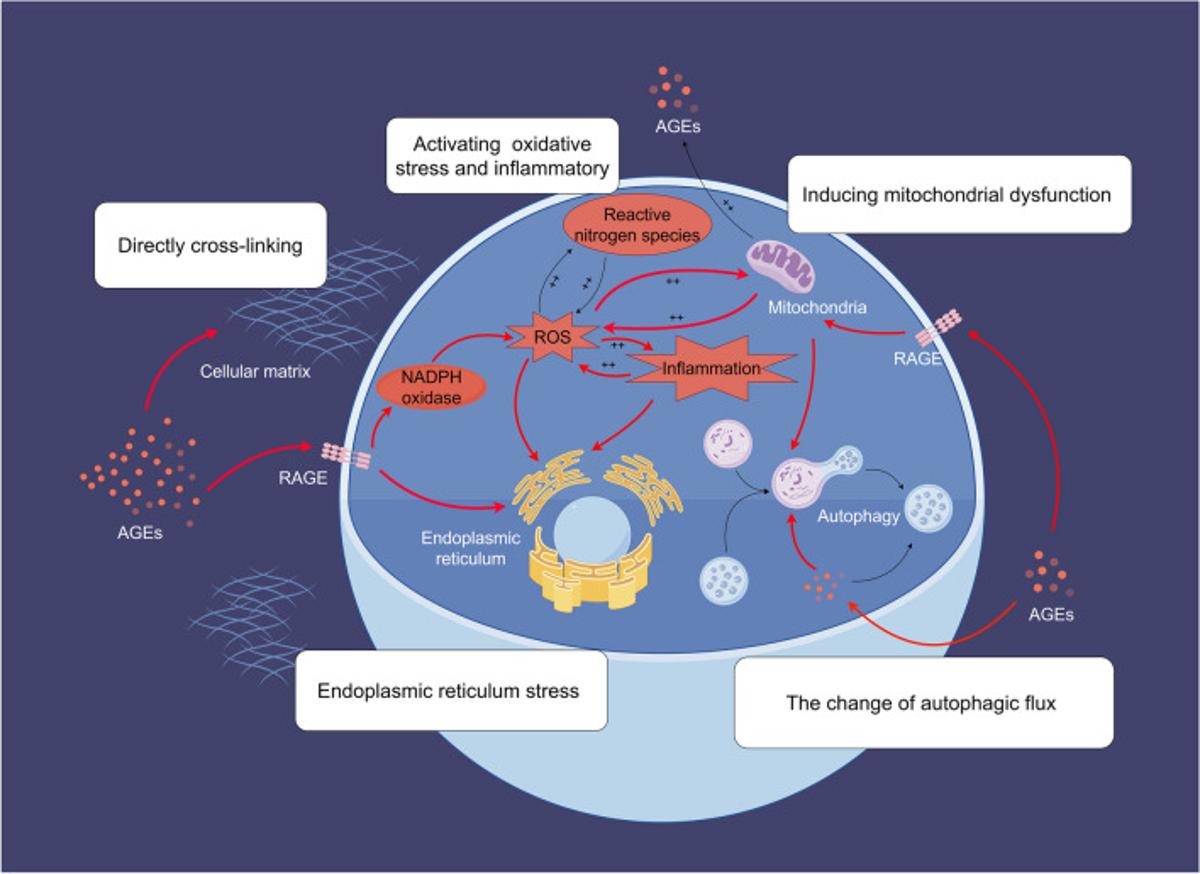
RAGE Gene Therapy Cuts Inflammation, Not Lifespan
It's a gene therapy that targets the RAGE pathway (Receptor for Advanced Glycation End-Products) that mediates inflammation and oxidative stress signaling. So far, the mice data doesn't show significant longevity benefits and mostly reduced inflammation and vascular stiffness and kidney damage. On...

Pointed Ironies: SERD Wars, ADC Hype, and What Really Works in Breast Cancer
The FDA rejected camizestrant at the ODAC meeting, while approving vepdegestrant a day later. AstraZeneca’s vepdegestrant leverages ctDNA to detect ESR1 mutations early, allowing a treatment switch while patients remain on a CDK4/6 inhibitor backbone. In contrast, Arvinas pursued a...
Minerva Neurosciences Provides First Quarter 2026 Financial Results and Business Updates
Minerva Neurosciences announced the start of its global confirmatory Phase 3 trial of roluperidone for negative symptoms of schizophrenia, enrolling about 380 patients across 40 sites, with the first patient screened in March 2026. The company reported a GAAP net loss...

The Technology Behind the Story: 3D Skin Grafts and the World of Scarpetta
The article highlights how 3D‑printed skin grafts are transitioning from laboratory experiments to clinical tools for complex wounds. Researchers at Columbia University have demonstrated patient‑specific, three‑dimensional grafts that fit irregular body parts like a glove, reducing surgery time and improving...

Myqorzo ACACIA Study Achieves Dual Primary Endpoint Success
$CYTK Here we go! Myqorzo ACACIA study results in nHCM --> BOTH co-primary endpoints hit with statistical significance. There may be debate about magnitude of improvements for Myqorzo via KCCQ-CSS and peak V02. But, if you were dreaming for a...

STAT+: Cytokinetics Drug Myqorzo Meets Twin Efficacy Goals in Study of Genetic Heart Disease
Cytokinetics announced that its Phase 3 ACACIA trial met both primary efficacy endpoints for Myqorzo in patients with non‑obstructive hypertrophic cardiomyopathy, showing significant symptom relief and improved cardiovascular fitness. Myqorzo is already approved for the obstructive form of HCM, and this...

Patents Hide Life‑Saving Drug Clues Await Discovery
What if the next life-saving drug is buried in a patent diagram? In early drug discovery, progress can hinge on a single decision. The evidence exists, but some of the most valuable insights stay invisible to conventional search, especially inside...

Agents Without Moats Will Falter in Fierce Market
Pointed ironies in the SERD wars, plus ADC hype, and what really works in breast cancer... How agents with no moat, no differentiation, and no clear advantage will struggle in a tough competitive market: https://t.co/2Efcy5ItNM https://t.co/FBDM2eDJd4
Slow Alzheimer’s Diagnoses ‘Mean UK Patients Missing Out on Experimental Treatments’
Alzheimer's Research UK warns that delayed or imprecise diagnoses are keeping UK patients out of a surge of experimental drug trials. While global trials hit a record 192 this year, fewer than 1,000 UK participants are enrolled in phase‑3 studies....
CYTK Phase 3 Surges After Long Road to Success
$CYTK Phase 3 hits + Been a long ride in that one, but sweet win https://t.co/daXv0EdCSa
NHS to Roll Out ‘1-Minute’ Immunotherapy Jab to Tens of Thousands with Cancer
The NHS is introducing a sub‑cutaneous form of pembrolizumab (Keytruda) that can be administered in just one minute, replacing the traditional two‑hour IV infusion. The rapid jab is approved for 14 cancer types, including lung, breast, head‑and‑neck, and cervical cancers,...

Boehringer’s Next‑Gen GLP‑1 Survodutide Cuts Weight 16% in Phase 3 Trial
Boehringer Ingelheim announced that its experimental GLP‑1/glucagon dual‑agonist survodutide produced an average 16.6% body‑weight reduction in a 725‑person Phase 3 study, far surpassing placebo. The result positions survodutide as a potential next‑generation tool for biohackers focused on metabolic optimization.
Fluorinated Amphiphilic Dendrimer to Improve PET Imaging of Cancer
Researchers engineered a fluorinated amphiphilic dendrimer nanocarrier radiolabeled with gallium‑68 to serve as a PET imaging agent. Fluorination lowered liver retention, accelerated renal clearance, and refined biodistribution, producing markedly higher tumor uptake in mouse models of glioblastoma and pancreatic adenocarcinoma....

Vertex Q1 2026 Revenue Hits $2.99B, CF Sales Drive Growth and UAE Gene Therapy Deal
Vertex Pharmaceuticals posted Q1 2026 total product revenue of $2.99 billion, an 8% year‑over‑year rise led by its cystic fibrosis franchise. The company also highlighted a landmark collaboration with Abu Dhabi’s Stem Cells Centre to deliver the first CRISPR‑Cas9 gene therapy...
Can Hydra Biology Inform Strategies to Extend Life in Other Species?
Hydra vulgaris displays negligible senescence thanks to a perpetual pool of stem cells that continuously replace its tissues. Researchers suggest that inserting Hydra‑like gene expression patterns into the short‑lived rotifer Brachionus manjavacas could extend the rotifer's healthspan and lower age‑related...

Inhibiting ID1 and ID3 Reduces Lung Fibrosis
Researchers identified that the transcription factors ID1 and ID3 are markedly elevated in fibroblasts from idiopathic pulmonary fibrosis (IPF) patients. Using both small‑molecule inhibitors and gene‑silencing techniques, they suppressed these proteins in mouse models, which halted fibroblast activation and triggered...
U of T Engineers Create Polymer Bristle Coating that Blocks Proteins and Germs on Medical Surfaces
University of Toronto engineers have developed a silicone‑based polymer bristle coating that prevents protein adhesion, a key step in bacterial colonisation. The non‑toxic surface could replace harsh disinfectants in hospitals, reducing chemical exposure and the risk of resistant strains.

IBM, Cleveland Clinic & RIKEN Simulate 12,635‑Atom Protein, Quantum Milestone
Scientists from IBM, Cleveland Clinic and Japan's RIKEN have used IBM’s 156‑qubit Heron processors together with the Fugaku and Miyabi‑G supercomputers to simulate a protein complex of 12,635 atoms – the biggest biologically relevant molecule ever tackled with quantum hardware....

RESEARCH: HBOT in CANCER (Hyperbaric Oxygen Therapy) - 2025 Review Paper From Sichuan University, Chengdu, China
A 2025 review paper from Sichuan University evaluates hyperbaric oxygen therapy (HBOT) as an adjunct in cancer treatment. The authors synthesize pre‑clinical data showing HBOT reverses tumor hypoxia and enhances the potency of radiotherapy and certain chemotherapies. They also examine...

Full Asset Acquisitions Avoid Margin Drag of Royalties
Seedy is not long enough $ABVX, however I’m old enough to remember when $ABBV spent $21B to buy half of ibrutinib.

Cibus Transfers Gene-Edited Herbicide-Tolerance Traits in Rice to Interoc Ahead of Latin American Launch
Cibus Inc. has transferred gene‑edited rice material that confers herbicide tolerance to its Latin American partner Interoc, meeting a scheduled development milestone. Interoc will begin testing and scaling production to launch Cibus‑enhanced seed varieties across the region. The transfer leverages...

Oxford Builds and Tests Structured Human Brain Tissue Using 3D Printing
Oxford University researchers have engineered layered human cortical tissue using stem cells, 3D printing and micro‑fluidics, then successfully implanted it into living mouse brains. The grafted tissue integrated with host neurons, formed functional synapses and reduced lesion size in traumatic‑brain‑injury...

Lannett Company and Sunshine Lake Pharma Report US FDA Approval of Langlara (Biosimilar, Lantus)
The U.S. Food and Drug Administration has granted interchangeable biosimilar status to Langlara, a copy of Sanofi’s Lantus insulin glargine. Langlara is approved for adults and children with type 1 diabetes and for adults with type 2 diabetes, based on data showing...
Can Ivermectin and Mebendazole Treat Cancer?
The Wellness Company posted a self‑reported study of its ivermectin 25 mg/mebendazole 250 mg capsule, claiming an 84% clinical‑benefit ratio and that 33% of participants showed no evidence of disease. The analysis drew on 197 baseline surveys and 122 six‑month follow‑ups, but relied solely...

Mirum Pharmaceuticals Reports P-IIb (VISTAS) Trial Data on Volixibat in Primary Sclerosing Cholangitis (PSC) Patients with Cholestatic Pruritus
Mirum Pharmaceuticals announced that its Phase IIb VISTAS trial met the primary endpoint, showing volixibat (20 mg BID) reduced cholestatic pruritus in primary sclerosing cholangitis (PSC) patients by 2.72 points on the Adult ItchRO scale versus a -1.08 point change with...

Canagliflozin - Another Top Longevity Drug
The class of sodium‑glucose cotransporter‑2 (SGLT2) inhibitors has moved from glucose‑lowering pills to a cornerstone of cardiometabolic care. Large‑scale trials and real‑world registries consistently show that agents such as canagliflozin, dapagliflozin and empagliflozin cut heart‑failure admissions, slow chronic kidney disease...

STAT+: Johnson & Johnson Advances IBD Therapy, Despite Trial Miss
Johnson & Johnson's combination therapy JNJ-4804, pairing Tremfya and Simponi, failed to meet the primary clinical remission endpoint in two Phase 2b trials for ulcerative colitis and Crohn's disease, though it outperformed each drug alone. The DUET study results were...
Neonatal Steroids Affect Preterm Infant Body Composition
A recent correction in Pediatric Research by Kraemer et al. re‑examines how neonatal corticosteroid therapy alters body composition of preterm infants at hospital discharge. Using dual‑energy X‑ray absorptiometry, the study finds a relative increase in fat mass and modest reductions...

CDC HoSt-TT Certification for Siemens Healthineers Total Testosterone Test Expands Patient Access to Gold Standard Equivalent Results
Siemens Healthineers’ Atellica IM Testosterone II (TSTII) assay has received CDC Hormone Standardization Program certification for total testosterone (HoSt‑TT), confirming its results match the gold‑standard LC‑MS/MS method. The assay, available on Atellica IM and CI analyzers, is the only fully automated immunoassay to...

Sarepta, Amylyx and Neumora Look Ahead to Key Catalysts as Q1 Earnings Roll In
Sarepta Therapeutics, Amylyx, and Neumora Therapeutics are set to report Q1 results amid pivotal milestones. Sarepta faces scrutiny after two DMD patient deaths and awaits FDA review of Amondys 45 and Vyondys 53, plus a sirolimus safety study. Amylyx, after pulling its...

The FDA Wants to Make Many Popular Prescription Drugs OTC—A Great Idea. Here’s Why It’s Unlikely to Happen
FDA Commissioner Marty Makary argues that most prescription drugs should be reclassified as over‑the‑counter unless safety concerns exist. The article highlights how prescription status inflates prices by routing purchases through insurance, citing lower cash prices for OTC versions of Claritin,...

Quantum Computing Moves Closer to Drug Discovery with Enzyme Study
A quantum‑computing team has successfully modeled the active site of a key enzyme, demonstrating that quantum simulations can capture chemical reactions with unprecedented accuracy. The study, conducted on a 127‑qubit superconducting processor, reproduced experimental binding energies within a few kilojoules...

Telomeres: History, Health and Hallmarks of Aging
Bill Andrews, a co‑discoverer of human telomerase, argues that telomere shortening is the primary limiter of human lifespan and that systemic activation of telomerase can reverse biological aging. He promotes small‑molecule activators such as TAM‑818 and botanical blends like Telo‑Vital,...

Molecular Hydrogen May Reduce Fatigue and Support Physical Function in People with Long COVID
A single‑blind, 14‑day pilot trial published in *Nutrients* examined hydrogen‑rich water versus regular water in 32 adults with long‑COVID. Participants drinking the hydrogen‑infused water reported statistically significant reductions in fatigue and showed measurable gains in six‑minute walk distance (42‑62 m), chair‑stand...