
Hyperbranched Biorefinery Molecule‐Regulated Switchable Adhesion and Noninvasive Healing
Researchers have created a reversible biomedical adhesive using a hyperbranched polysaccharide produced via microbial fermentation. The nanoconfined structure supplies abundant dynamic disulfide bonds, delivering both high adhesive strength and a broad, controllable adhesion range (296 N/m down to 17 N/m). In animal wound models the adhesive achieved a 94.8% closure rate after ten days, far surpassing the 76.1% rate of standard controls. This work demonstrates that biorefinery‑derived molecules can overcome traditional bio‑adhesive limitations while remaining sustainable.
One-and-Done Heart Disease Prevention? Scientists Show It May Be Possible.
Scientists reported that a single infusion of an experimental gene‑editing drug lowered LDL cholesterol by up to 62 percent in an interim analysis of 35 patients. The reduction remained durable in a subgroup treated 18 months earlier, suggesting a potentially...

Agios Reports the EC Approval of Pyrukynd for Anemia in Alpha or Beta Thalassemia
The European Commission has granted approval for Agios' oral pyruvate kinase activator Pyrukynd (mitapivat) to treat anemia in adults with both non‑transfusion‑dependent and transfusion‑dependent alpha‑ or beta‑thalassemia. The decision follows positive results from the global Phase III ENERGIZE and ENERGIZE‑T trials,...

Pharming Reports the EC Approval of Joenja (Leniolisib) for Activated Phosphoinositide 3-Kinase Delta Syndrome (APDS)
The European Commission has granted marketing authorization for Pharming’s oral PI3Kδ inhibitor Joenja (leniolisib) to treat patients aged 12 and older with activated phosphoinositide 3‑kinase delta syndrome (APDS) across all 30 EEA states. Approval is based on a Phase II/III...

Caliway Secures FDA Clearance for CBL-514 Clinical Trial
Caliway Biopharmaceuticals received FDA clearance for its CBL‑0302 IND application, clearing the path for a pivotal global Phase III trial of CBL‑514 aimed at reducing abdominal subcutaneous fat. The double‑blind, placebo‑controlled study will enroll roughly 320 participants in the United States, Canada...

Late-Breakers to Look Out for at ASCO 2026
ASCO 2026’s late‑breaker session will unveil a slate of pivotal trials that could reshape treatment algorithms across liver, bladder, pancreatic, prostate, multiple myeloma and several rare cancers. AstraZeneca will present EMERALD‑3 and POTOMAC data, pushing its Imfinzi‑based combos into first‑line...
Oral Wegovy Backed by CHMP at May Meeting
The European Medicines Agency’s CHMP gave a positive opinion on Novo Nordisk’s oral formulation of Wegovy, marking the first EU approval for an oral GLP‑1 weight‑loss drug. The endorsement paves the way for a launch in select non‑US markets in...

EU Opens Public Consultation on Biotech Act II, Targeting Industrial Fermentation and Biomanufacturing
The European Commission opened a public consultation on a proposed Biotech Act II, extending the EU’s biotech framework to cover industrial fermentation, cultivated biomass, and precision‑fermentation products. Stakeholders from alternative‑protein and industrial‑biotech firms submitted feedback urging clearer labeling, safety assessments, and...

FEATURE: From Lab Idea to $2.25 Billion: Ultrasound Destroys Cancer without Scalpels
Biomedical engineer Zhen Xu’s 25‑year effort produced histotripsy, an ultrasound‑based method that liquefies tumor cells without incisions. Her start‑up HistoSonics, founded in 2009, was valued at $2.25 billion after a majority‑stake investment. The technique earned FDA approval for liver cancer in...

DNA Methylation Variability May Improve Precision Cancer Treatment Strategies
A new review in Genes & Diseases highlights DNA methylation heterogeneity as a driver of tumor microenvironment complexity. It distinguishes intra‑tumor and inter‑patient methylation variability and links these patterns to tumor evolution, immune escape, and metabolic reprogramming. Advances in quantitative...

Low-Dose Ketamine Shows Promise for Easing Chronic Fatigue
NIH researchers ran a randomized, double‑blind crossover trial with ten adults experiencing chronic fatigue from cancer, fibromyalgia, lupus and ME/CFS. A single low‑dose ketamine infusion lowered fatigue scores by 21% on day three, meeting the study’s 20% benchmark, while the...
Kura Oncology’s SWOT Analysis: Biotech Stock Faces Launch Test
Kura Oncology secured FDA approval in November 2025 for Komzifti (ziftomenib) to treat relapsed or refractory NPM1‑mutated acute myeloid leukemia, propelling its stock nearly 100% higher over the past year. However, Q4 2025 sales fell short of analyst expectations, highlighting early‑launch...
Bispecifics in the Community: Infrastructure, Education, the Future
Bispecific antibodies are becoming a pivotal oncology therapy, delivering durable responses in relapsed or refractory hematologic cancers and moving into earlier treatment lines. Their step‑up dosing schedules and intensive toxicity monitoring demand new workflows, remote therapy monitoring, and robust coordination...

Deep Phenotyping Reveals Skin Remodeling in Sclerosis Treatment
A recent study employing deep phenotyping—integrating high‑resolution imaging, transcriptomics, and proteomics—has uncovered significant skin remodeling in patients undergoing experimental sclerosis therapy. The data show a 30% reduction in skin thickness and a 45% drop in collagen deposition after six months...

Ionis Pharmaceuticals’ SWOT Analysis: Stock Gains Momentum on FDA Approval
Ionis Pharmaceuticals (NASDAQ:IONS) secured FDA approval for REDEMPLO, the first siRNA therapy targeting familial chylomicronemia syndrome (FCS), in November 2025. The drug’s clean label, quarterly at‑home dosing and broad indication have positioned it for rapid uptake, with payer agreements covering...

Final Frontier for Meds? UK Startup Sends Drug-Making Into Space
BioOrbit, a UK biotech, launched its Box‑E microgravity crystallisation unit to the International Space Station on a SpaceX flight, aiming to produce ultra‑pure protein crystals for self‑injected cancer drugs. The six‑week orbital test leverages weightlessness to create stable crystal structures...

Venus Remedies Gets Saudi FDA Approval for Speciality Oncology Therapy
Venus Remedies Limited announced that the Saudi Food and Drug Authority has granted marketing authorisation for its specialty oncology drug Plerixafor, a stem‑cell mobiliser used in autologous transplants. This marks the first global approval for the therapy, positioning Saudi Arabia...

After Early Semaglutide Lead, Torrent Pharma Eyes US Liver Drug Resmetirom for India Launch
Torrent Pharmaceuticals is conducting a Phase 3 trial of Resmetirom, the first US‑approved drug for metabolic dysfunction‑associated steatohepatitis (MASH), with plans to launch in India once patent barriers lift. The company already commands a strong position in the Indian semaglutide market,...
Pim1 Identified as Promising Therapeutic Target for Inflammatory Arthritis Treatment
Researchers have pinpointed the serine/threonine kinase Pim1 as a pivotal driver of abnormal Th17 cell differentiation in inflammatory arthritis. Elevated Pim1 levels were detected in CD4⁺ T cells from rheumatoid arthritis and ankylosing spondylitis patients, and mice lacking Pim1 in...
Gene-Based Therapies Could Transform Future Pancreatitis Treatment
Pancreatitis remains a largely untreatable inflammatory disease, with current care limited to symptom management. Gene‑based therapies—spanning augmentation, inhibition, and editing—are emerging to address the genetic drivers across lipid‑metabolism, trypsin regulation, ductal secretion, and ER‑stress pathways. Recent preclinical work shows AAV‑mediated...

ASCO to Spotlight Pancreatic Cancer Innovation Beyond KRAS
The page is BioCentury’s cookie policy, outlining five categories of cookies used on its website. Strictly necessary cookies support authentication and navigation, while functional cookies enable personalization. Marketing, advertising, and analytics cookies are used to tailor offers, display ads, and...
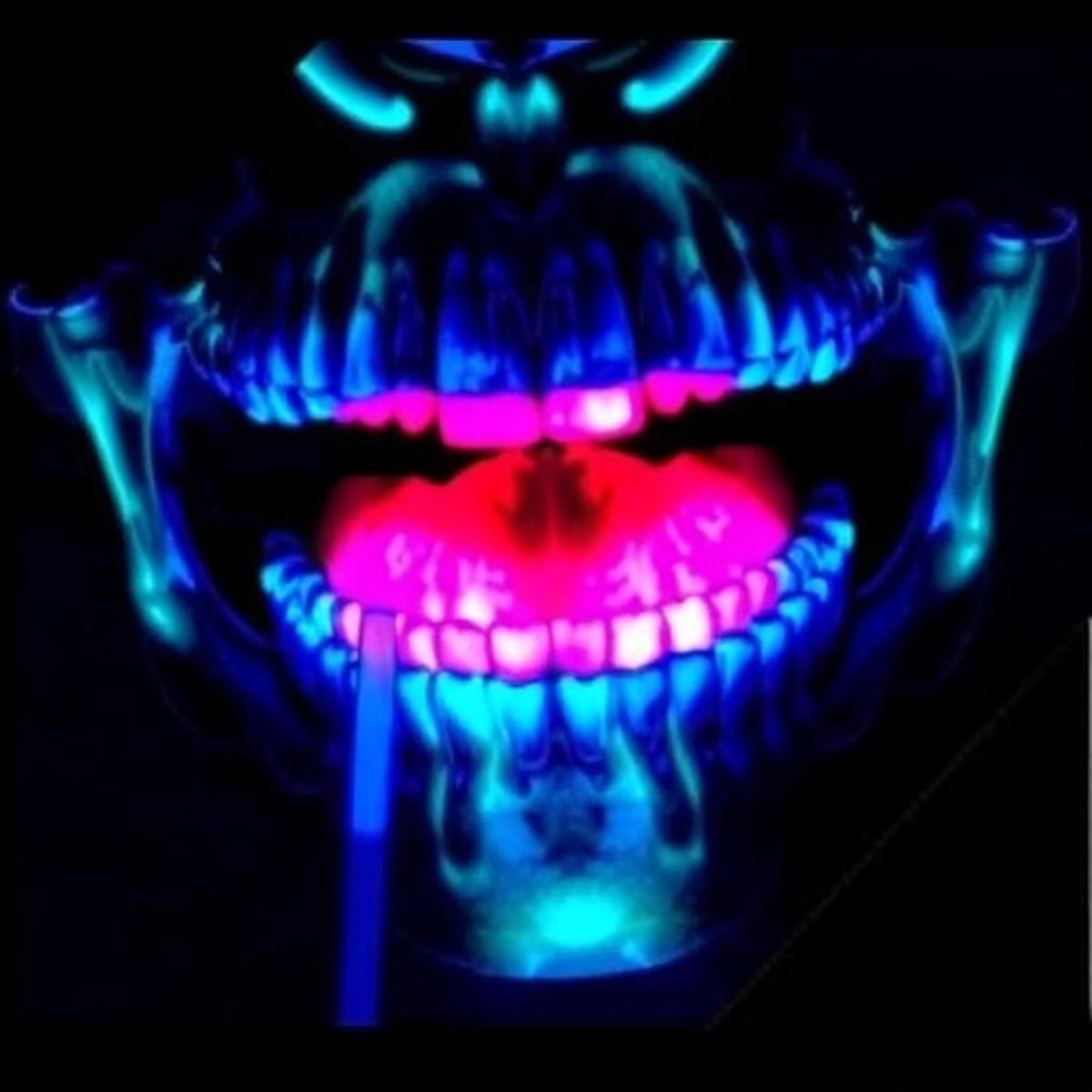
NIR Fluorescence Surgery Enhances Oral Cancer Removal
Near‑infrared (NIR) fluorescence imaging is being integrated into oral cancer surgery to highlight malignant tissue that standard visual inspection can miss. A multi‑center trial of 120 patients demonstrated a 30% reduction in positive surgical margins and shaved roughly 12 minutes...

New Study Could Improve Testing and Treatment for Rare Brain, Spinal Cord, and Eye Cancers
Researchers at Fudan University identified hepatitis A virus cellular receptor 1 (HAVCR1) as a fluid‑based biomarker that distinguishes primary central nervous system lymphoma (PCNSL) and its eye‑only variant, primary vitreoretinal lymphoma (PVRL), from non‑cancerous conditions. In a study of 199 lymphoma...
New Indicator for Response to Therapy in Pediatric Cancers Identified
Researchers at the University of Birmingham reported that a high aneuploidy score can predict which children with relapsed solid tumors respond to a combined low‑dose irinotecan and PARP‑inhibitor regimen. The Phase I/II eSMART arm enrolled 70 patients across the UK, France,...

From Decades to Years - AI Could Speed Search for Brain Drugs Hiding in Plain Sight
Scientists at the UK Dementia Research Institute are using artificial intelligence to sift through patient data, voice recordings, eye scans and lab‑grown brain cells, aiming to repurpose existing drugs for neurological conditions such as motor neurone disease (MND). By training...

Abivax Presents First Quarter 2026 Financial Results and Reports Three-Year Interim Data From Study 108, a Phase 2a/2b Open-Label Extension...
Abivax reported Q1 2026 results, highlighting €491.6 million (≈$540 million) in cash and a runway extending into Q4 2027. The company released three‑year interim data from Study 108, showing that 68% of 130 ulcerative colitis patients remained in clinical remission after 144 weeks of obefazimod...

FDA Approves Hepcludex, First Treatment for Chronic Hepatitis D
The U.S. Food and Drug Administration has granted approval to Hepcludex (bulevirtide‑gmod), marking the first FDA‑cleared therapy for chronic hepatitis D in adults without cirrhosis or with compensated cirrhosis. Approval follows the phase 3 MYR301 trial, which showed a 48% combined response...

FDA Clears Gilead's Hepatitis D Drug, Four Years After Prior Rejection
Gilead’s hepatitis D therapy Hepcludex (bulevirtide) has finally secured FDA approval after a prior rejection over manufacturing and distribution flaws. The drug becomes the first U.S.‑cleared treatment for chronic hepatitis D, a rare but aggressive liver infection affecting roughly 70,000 Americans. Phase 3...

Enzymes Involved in Cholesterol Transport May Point to New Cancer Therapies
Scientists at Sanford Burnham Prebys and the University of Illinois Chicago identified phosphatidylinositol 5‑phosphate 4‑kinases (PI5P4Ks) as critical for cholesterol trafficking in TP53‑mutant cancers. In mouse models, deleting PI5P4K α and β prevented tumor formation by causing lysosomal cholesterol mislocalization...

Skape Bio Unlocks Generalizable GPCR Drugs Using AI Protein Design
Skape Bio, founded by former UW protein‑design researcher Chris Norn, has unveiled an AI‑driven platform that creates miniprotein therapeutics for G‑protein‑coupled receptors (GPCRs). A recent Nature paper shows functional miniproteins targeting 11 diverse GPCRs, including agonists validated on three receptors....

Human Gut Organoids with Functional Nerves Developed that Can Be Mass Produced
Researchers at Cincinnati Children’s Hospital and Nantes Université have unveiled a 3D‑printed scaffold system that accelerates the growth of human gut organoids to transplantation maturity in 14 days—half the previous timeline. The confined culture system yields centimeter‑scale intestinal, colon and stomach...
Experimental mRNA Vaccine May Protect Against Multiple Ebola Viruses
Researchers have created an experimental mRNA vaccine that protects rodents from three orthoebolavirus strains, including the Bundibugyo virus driving the current outbreak in the Democratic Republic of the Congo and Uganda. The study shows the vaccine elicits robust antibody responses...
Expanded Label for Efgartigimod Offers Fast-Acting Treatment for gMG: James F. Howard, MD
On May 11, 2026, the FDA broadened the indication for efgartigimod, approving it for all adult patients with generalized myasthenia gravis, including AChR‑positive, MuSK‑positive, LRP4‑positive, and triple‑seronegative subtypes. The decision follows the phase‑3 ADAPT SERON trial, which enrolled 119 patients...
Team Finds Markers of Inflammatory Breast Cancer in Blood
University of Texas researchers have discovered blood‑based RNA biomarkers that reliably differentiate inflammatory breast cancer (IBC) from other breast cancer subtypes. Using the Thermostable Group II Intron Reverse Transcriptase (TGIRT) sequencing platform, they captured complex and fragmented RNAs missed by...

First Generic Drug Approvals
The FDA’s Center for Drug Evaluation and Research approved 19 first‑generic drug applications between January and March 2026, spanning therapeutic areas such as oncology, infectious disease, psychiatry, renal care, and pain management. Notable entries include the first generic bortezomib injection...
ASCO26: 5 Data Snapshots Ahead of the Year’s Biggest Cancer Drug Meeting
Ahead of the ASCO Annual Meeting, several late‑stage oncology trials were previewed in newly released abstracts. Merck’s antibody‑drug conjugate sacituzumab tirumotecan (sac‑TMT) combined with Keytruda cut disease‑progression risk by 65% and achieved a 70% response rate in first‑line non‑small cell...

OSE’s Tedopi-Keytruda Combo Clears Phase II Ovarian Cancer Hurdle
French biotech OSE Immunotherapeutics announced positive Phase II data for its cancer vaccine Tedopi combined with Merck's Keytruda in platinum‑sensitive recurrent ovarian cancer. The combination improved median progression‑free survival to 4.1 months versus 2.8 months with best supportive care, cutting the risk of...

Focus on Biosimilar Adoption: Driving Uptake Through Strategy
Teva’s U.S. Value & Access team has moved beyond simple payer formulary placement, adopting a holistic ecosystem approach that coordinates payers, providers, pharmacies, and health systems to accelerate biosimilar uptake. By redesigning contracts to include step‑therapy, lower patient cost‑sharing, and...
What Is Immunotherapy and How Does It Treat Cancer and Other Conditions?
Immunotherapy research has surged, with clinical trials climbing from 1,257 between 2006‑2016 to 4,591 in the past decade. Cancer treatments dominate the field, featuring checkpoint inhibitors, CAR‑T cells, and mRNA‑based vaccines now approved for more than 30 tumor types. Researchers...

Cognitive Effects Vary by Therapy for Advanced Prostate Cancer
A phase‑2 ARACOG trial presented at ASCO showed that men with advanced prostate cancer receiving darolutamide experienced significantly less cognitive decline over 24 weeks than those on enzalutamide. The study enrolled 111 patients (median age 71) and evaluated five computer‑based neurocognitive tests,...

The NMPA Grants Conditional Approval to Boehringer Ingelheim’s Hernexeos for 1L HER2-Mutant NSCLC
China's National Medical Products Administration granted conditional approval to Boehringer Ingelheim’s oral HER2‑mutant NSCLC drug Hernexeos (zongertinib). The decision is based on a Phase Ib trial of 74 treatment‑naïve patients that delivered a 75.7% overall response rate and a median...

LSX Nordic Congress 2026: What Came Out of the Biotech Partnering Event
At the LSX Nordic Congress 2026 in Copenhagen, life‑science leaders emphasized that Nordic biotech firms must look beyond their small domestic markets to achieve growth. Executives highlighted a scarcity of risk‑willing capital, especially for Series A and B rounds, prompting...

Biogen, Denali to Drop Drug in Non-Genetic Parkinson’s After Mid-Stage Study Flop
Biogen and Denali Therapeutics announced that their LRRK2 inhibitor BIIB122 failed to meet primary endpoints in a Phase 2b trial of early‑stage Parkinson’s disease. The mid‑stage study, which targeted patients without the LRRK2 genetic mutation, showed no statistically significant slowing...
Moderna’s Once-Rebuffed mRNA Flu Shot to Face Scrutiny From FDA Adcomm
The FDA will hold its Vaccines and Related Biological Products Advisory Committee on June 18 to evaluate Moderna’s investigational mRNA flu vaccine, MFLUSIVA, aimed at adults 50 and older. Earlier this year the agency issued a refusal‑to‑file letter, arguing the...

Tempus AI Launches ArteraAI Prostate Test for Metastatic Prostate Cancer
Tempus AI announced the clinical launch of the ArteraAI Prostate Test, a CLIA‑certified and CAP‑accredited prognostic assay for metastatic hormone‑sensitive prostate cancer (mHSPC). The test combines a digital pathology AI algorithm with Tempus’ next‑generation sequencing data to generate personalized risk...

QuberTech Secures Funding Boost for Solution that Promises to Turn Dandelions Into Sustainable Rubber
QuberTech, a UK biotech firm, announced a £3.4 million (≈$4.3 million) funding round to scale its engineered‑dandelion platform that produces natural rubber. The capital will fund field trials, greenhouse expansion, and a pilot processing line aimed at commercial‑scale output. The company positions...

NICE Changes Stance on Genmab's Cervical Cancer Drug
Two months after rejecting Genmab’s Tivdak, the UK health technology body NICE has reversed course, recommending the antibody‑drug conjugate for adults with recurrent or metastatic cervical cancer. The decision follows a revised economic model that incorporates the innovaTV 301 trial results...

Allergan Aesthetics (AbbVie) Reports CHMP Positive Opinion for Boey to Temporarily Improve Glabellar Lines in Adults
Allergan Aesthetics, a unit of AbbVie, received a positive opinion from the European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP) recommending Boey (trenibotulinumtoxinE) for the temporary improvement of moderate-to-severe glabellar lines in adults. The recommendation covers all...
The Problem at the Heart of Drug Discovery: Lexogen & Ochre Bio on the Power of AI on Human Data
Lexogen, an RNA transcriptomics and NGS service provider, teamed up with Ochre Bio, a biotech developing AI‑driven RNA therapies for chronic liver disease. The partnership leverages Lexogen’s high‑throughput sequencing to generate human‑first data that trains Ochre’s predictive models. Together they...
Biogen, Denali Pull Parkinson's Drug After Failed Trial
Biogen has halted development of BIIB122, its LRRK2‑inhibitor partner with Denali, after the phase 2b LUMA trial failed to slow Parkinson’s disease progression despite achieving about 30% target inhibition. The study, which included both LRRK2‑mutated and idiopathic patients, missed its primary...