
Emerging Nanoreactors for Precision Disease Treatment: From Principles to Biomedical Applications
Nanoreactors—engineered nanostructures that emulate cellular compartments—are reshaping precision nanomedicine. By confining reactants, integrating synergistic catalytic sites, and responding to physiological cues, they dramatically accelerate therapeutic reactions and enable controlled drug release. Recent advances span organic, polymeric, and organic‑inorganic hybrids, showing promise against Alzheimer’s disease, cancer, diabetes, rheumatoid arthritis, and chronic wounds. The review highlights mechanistic insights, platform classifications, and the remaining hurdles before clinical adoption.

Application of Hydrogel in the Capture and Detection of Circulating Tumor Cells
The review details how hydrogel‑based platforms are reshaping circulating tumor cell (CTC) capture and detection. Hydrogels provide a three‑dimensional porous matrix that enhances cell infiltration, biocompatibility that preserves CTC viability, and tunable chemistry for affinity‑driven targeting. By coupling these materials...
Medtronic Data Back Renal Denervation in the Long Term
Medtronic reported that two EuroPCR abstracts based on the global SYMPLICITY registry show its Symplicity Spyral renal denervation system delivers sustained blood‑pressure reductions over three years. One analysis of 787 patients with prior cardiovascular events recorded a 17.4 mmHg drop in office...
WVE-006 RNA Editing Therapy Achieves MZ-Like Phenotype in Alpha-1 Antitrypsin Deficiency Phase 1b/2a Trial
Wave Life Sciences reported that its investigational RNA‑editing drug WVE‑006 generated major‑variant alpha‑1 antitrypsin (M‑AAT) levels comparable to the protective heterozygous Pi*MZ phenotype in patients with homozygous Pi*ZZ AAT deficiency. The Phase 1b/2a RestorAATion‑2 trial showed 64%–59% of circulating AAT was...
EQS-News: GeoVax Comments on Escalating Bundibugyo Ebola Outbreak and Growing Need for Flexible Biodefense Vaccine Platforms
GeoVax Labs warned that the escalating Bundibugyo Ebola outbreak in Central Africa highlights the lack of licensed vaccines for less‑common Ebola strains. The company pointed to its Modified Vaccinia Ankara (MVA) platform, which has shown single‑dose protection against Zaire and...

Treating Pattern Hair Loss: How Close Are We?
VeraDermics’ oral minoxidil pill VDPHL01 cleared a pivotal phase 2/3 trial, showing a 30‑33 hairs/cm² increase in non‑vellus hair count versus 7.3 for placebo and coverage improvement in over 79% of participants. The extended‑release formulation reduces peak blood levels that have...
Scientists Found a Hidden Alzheimer’s Trigger and Shut It Down
Researchers at Indiana University identified the brain enzyme IDOL as a promising new target for Alzheimer’s therapy. Deleting IDOL from neurons in mouse models dramatically cut amyloid plaque buildup and lowered APOE levels, a key genetic risk factor. The findings...

Engineered Nanomaterials Optimize Delivery Barriers in Cancer Immunotherapy
A new review outlines how engineered nanomaterials can overcome delivery bottlenecks in cancer immunotherapy by matching material design to each step of the immunity cycle. It details active‑targeting ligands, intracellular escape mechanisms, and co‑delivery of antigens, adjuvants, mRNA or CRISPR...

Beam One-Ups Wave as Both Show Promise of Editing for AATD
Beam Therapeutics presented Phase 1/2 data for its DNA editor BEAM‑302, showing an 80% drop in mutated alpha‑1 antitrypsin (AAT) protein and lifting total AAT above the 11 µM protective threshold, with effects lasting 12 months. Wave Life Sciences reported its RNA editor...
AbbVie’s New Immunology Standard-Bearer Skyrizi Kneels to UCB’s Bimzelx in Psoriatic Arthritis
UCB’s Bimzelx outperformed AbbVie’s Skyrizi in a Phase 3 head‑to‑head trial for psoriatic arthritis, achieving 49.1% ACR50 versus 38.4% for Skyrizi at week 16. While Bimzelx also showed numerically higher minimal disease activity (43% vs 39.9%), the difference missed statistical significance. Skyrizi...

QIAGEN and NVIDIA Partner on AI Drug Discovery
Qiagen’s Digital Insights division will embed NVIDIA’s accelerated computing and BioNeMo platform into its bioinformatics suite, creating a graph‑based AI environment for drug discovery. The integration is designed to streamline target identification, biomarker discovery, and multi‑omics hypothesis generation for pharma...

BioMarin’s ENERGY 3 Trial of BMN 401 Meets One Co-Primary Endpoint
BioMarin Pharmaceutical announced that its Phase III ENERGY 3 trial of BMN 401 met one of two co‑primary endpoints, showing a statistically significant rise in plasma inorganic pyrophosphate (PPi) levels in children with ENPP1 deficiency through week 52. The trial failed to demonstrate any...

Proactivity for Drug Safety: A New Service or a New Mindset?
The letter argues that proactive drug‑safety surveillance cannot fix a system that approves drugs with insufficient benefit data and overlooks rare, severe harms. It cites historic delays in risk‑management actions for valproate and topiramate, noting that formal REMS were only...

Tuneable Peptide Biotech Parabilis Files IPO
Parabilis Medicines, the Cambridge‑based tuneable peptide biotech, filed a Nasdaq prospectus to raise roughly $100 million in an IPO under the PBLS ticker. The offering follows a $305 million private round and a multi‑billion‑dollar alliance with Regeneron, which contributed $50 million upfront and...

Eight Swedish Companies Spearheading the Country’s Biotech Scene in 2026
Sweden’s life‑science sector, now worth roughly $42 billion in turnover and employing over 52,000 people, is being propelled by eight standout biotech firms. Annexin Pharmaceuticals reported safety and early efficacy in a phase 2a ophthalmology trial, while Anocca raised $46 million to advance...

Lilly Snaps up Engage to Advance Non-Viral Genetic Medicines
Eli Lilly agreed to acquire Engage Biologics for up to $202 million in cash, adding the company’s non‑viral DNA delivery platform called Tethosome. The technology combines lipid‑nanoparticle shells with an mRNA‑encoded transport protein to move genetic payloads into cell nuclei without viral vectors....
Efficacy of Ustekinumab Combined with Partial Enteral Nutrition in Crohn’s Disease
A retrospective cohort of 124 Crohn’s disease patients showed that adding partial enteral nutrition (PEN) to ustekinumab (UST) therapy markedly improved long‑term mucosal healing. At week 54, endoscopic remission was achieved in 71.05% of the UST + PEN group versus 50.00% with...
UCB Reports P-III (BE BOLD) Trial Data on Bimzelx in Active Psoriatic Arthritis
UCB disclosed Phase III BE BOLD trial results comparing its Bimzelx (bimekizumab) to AbbVie’s Skyrizi (risankizumab) in 553 adults with active psoriatic arthritis. The primary endpoint was met, with 49.1% of patients achieving an ACR50 response at week 16 versus 38.4%...

Field-Ready Tool Identifies Rare and Zoonotic Parasitic Worms Missed by Standard Tests
Researchers at the University of Melbourne and UNSW have created a field‑ready diagnostic that uses Oxford Nanopore long‑read sequencing to profile the full community of parasitic nematodes in stool from humans and animals. Validation showed sensitivity and specificity comparable to...
Startup Creates Embryos Using Lab-Grown Sperm
Paterna Biosciences announced it has successfully generated human sperm cells from stem cells in the laboratory and used them to fertilize oocytes, producing embryos. The breakthrough was demonstrated by co‑founder and CEO Dr. Alex Pastuszak, who explained the step‑by‑step protocol...

Kraig Biocraft Labs Creates Immortalized Silk Gland Cell Line
Kraig Biocraft Laboratories announced the creation of an immortalized silk gland cell line that could serve as the foundation for a next‑generation biotechnology platform. The cells exhibit strong proliferative capacity, stable serial passaging and robust long‑term viability, while delivering high...
Sustained Therapeutics – Presents Positive Phase 2 Data for ST-01 in Podium Presentation at the American Urological Association 2026 Annual...
Sustained Therapeutics presented Phase 2 data showing its ST‑01 polymer‑lidocaine formulation significantly reduced pain in men with chronic scrotal content pain (CSCP). At the 70 mg/mL dose, 67% of patients achieved a ≥2‑point pain reduction and 83% met clinical response criteria, far...

Why the Need to Expand Industry Focus Beyond Gene Editing Toward Scalable Cell Therapy Manufacturing?
Cell therapy developers are moving beyond pure gene‑editing breakthroughs to prioritize scalable manufacturing, according to Cellular Origins CEO Dr. Edwin Stone at the 2026 ASGCT meeting. The industry is integrating manufacturing feasibility into early‑stage development, evaluating multiple delivery platforms such...

Mentari Therapeutics Announces Merger with InMed Pharmaceuticals, Concurrent $290 Million Private Placement
Mentari Therapeutics, a biotech focused on migraine prevention, announced a merger with InMed Pharmaceuticals alongside a $290 million private placement. The financing round was led by Fairmount and featured investors such as a16z Bio + Health, Venrock, Wellington Management, and Blackstone...
CRISPR-Based System Targets RNA and Kills Cells on Demand
Scientists at Utah State University have engineered a CRISPR‑Cas12a2 system that reads a specific RNA transcript and triggers uncontrolled DNA shredding, killing the host cell. The enzyme reduced yeast colonies 134‑fold and stopped proliferation of HeLa cancer cells, even when...

ESR1 Mutation Testing Questions Follow Trial
A recent clinical trial examined the role of ESR1 mutations in advanced hormone‑receptor‑positive breast cancer, revealing that patients with the mutation respond differently to endocrine therapies. The study demonstrated that a next‑generation selective estrogen degrader (SED) extended progression‑free survival compared...
BioMarin Drug Acquired in Buyout Misses Goal in Rare Disease Study
BioMarin Pharmaceutical’s BMN 401, an enzyme‑replacement therapy for ENPP1 deficiency, met its primary biochemical endpoint by raising plasma PPi levels but failed the co‑primary skeletal health goal in a Phase 3 trial of 27 children. The miss on clinical benefit and all...

JCR Pharmaceuticals Highlights Preclinical CNS Gene Therapy Data for JUST-AAV Platform at ASGCT 2026
JCR Pharmaceuticals showcased preclinical data for its JUST‑AAV platform at ASGCT 2026, highlighting enhanced central nervous system (CNS) delivery and reduced liver exposure compared with conventional AAV9 vectors. The platform uses transferrin‑receptor‑targeted capsids to cross the blood‑brain barrier, delivering therapeutic...

Lessons Learned From Drug Development Programs in Autism: Implications for Future Programs
The article synthesizes expert insights on why autism drug development has lagged, highlighting the lack of approved therapies for core symptoms and the challenges posed by biological and clinical heterogeneity. It outlines six key learnings, including the need to measure...
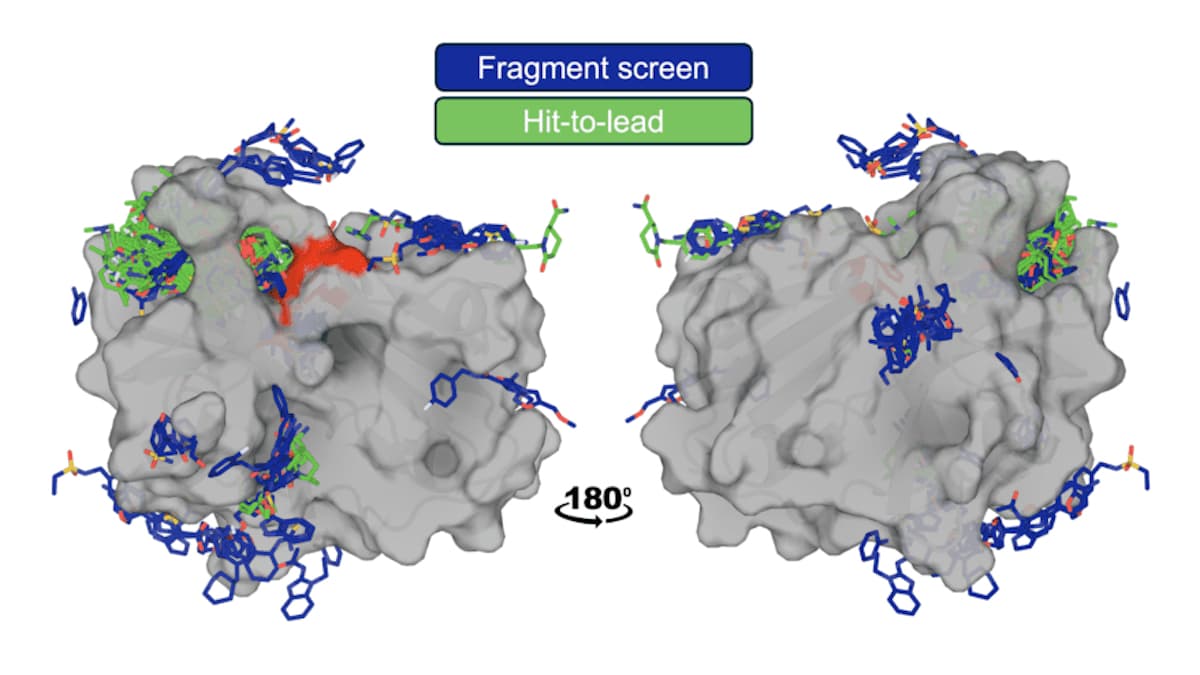
OpenBind Unveils Its First AI Model for Drug Discovery
OpenBind, a UK‑led consortium, has published its first AI‑ready dataset and a predictive model for the EV‑A71 2A protease, a target linked to hand, foot and mouth disease. The release includes 699 X‑ray structures and binding‑strength data for 601 compounds,...

Wave Aims for Monthly Dosing with RNA Editing Treatment for AATD
Wave Life Sciences announced an updated read‑out from its early‑stage trial of an RNA‑editing therapy for alpha‑1 antitrypsin deficiency (AATD). The data indicate that the treatment can restore functional protein levels with a dosing schedule that could be moved to...

Relay Doubles the Bar, Outpacing Novartis with a 60% Response in Rare Disease
Relay’s oral PI3Kα inhibitor zovegalisib posted a 60% volumetric response in a Phase 2 trial of patients with vascular malformations, far outpacing Novartis’ 11% result with Vijoice. The data, presented at the ISVAA World Congress 2026, came from 20 evaluable patients,...

Harbin Jixianglong Biotech Co., Ltd. - 723330 - 05/01/2026
The FDA issued a warning letter to Harbin Jixianglong Biotech after a November 2025 inspection revealed multiple CGMP violations involving its peptide API manufacturing. The firm repackaged and relabeled semaglutide batches from unapproved suppliers, misbranding the products and failing to...

PolleyMed, LLC - 726018 - 05/14/2026
The FDA issued a warning letter to PolleyMed, LLC for marketing its SaniiSwab™ nasal cleaning product as an unapproved new drug and for misbranding claims of FDA OTC approval. The agency’s review of the company’s website and social‑media content found...

Sourav K. Mishra, M.D. / All India Institute of Medical Sciences - 724881 - 04/29/2026
The FDA issued a warning letter to Dr. Sourav K. Mishra of AIIMS Bhubaneswar for serious violations in a bioequivalence study of doxorubicin hydrochloride liposome injection conducted for Qilu Pharmaceutical. The investigator administered the prohibited CYP3A4 inhibitor aprepitant to all...

RAS Inhibition Enters Its Second Wave
RAS inhibition has moved into a second wave of drug development that goes beyond the KRAS G12C breakthrough. After sotorasib and adagrasib secured accelerated approvals for non‑small cell lung cancer and later for KRAS G12C‑mutated colorectal cancer, companies are targeting more prevalent...

STAT+: Eli Lilly Tops Prominent Rankings on Pharma R&D Performance
Eli Lilly topped the IDEA Pharma index, claiming the No. 1 spot in both Innovation and Invention categories. The Innovation ranking weighs revenue from new products, recent approvals and major development milestones, while the Invention ranking evaluates pipeline depth, clinical trial activity and...
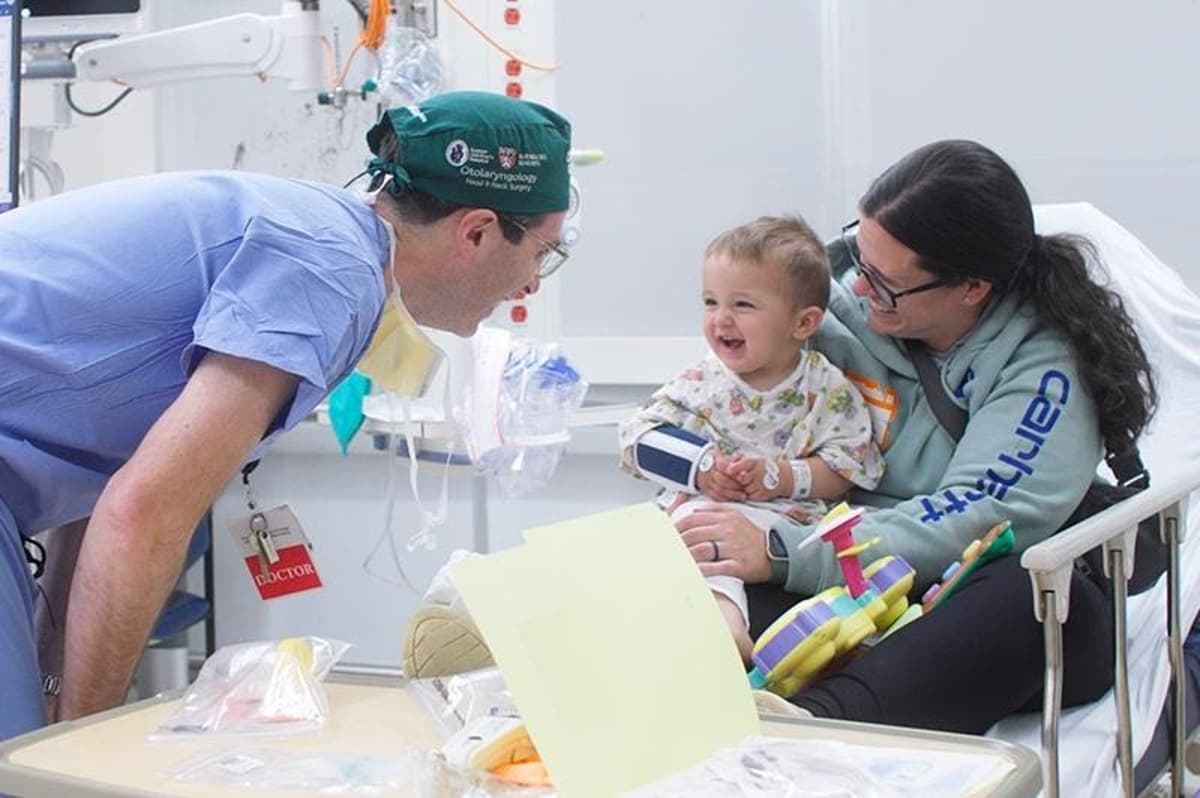
FDA Approves First-Ever Gene Therapy For Deafness, Opening Door To New Era
The U.S. Food and Drug Administration has approved the world’s first gene therapy for deafness, targeting the OTOF mutation that blocks sound transmission in the inner ear. In a pivotal trial of 20 children and teens, 80% experienced improved hearing...

In the Clinic for May 19, 2026
BioWorld’s May 19, 2026 clinic roundup aggregates a wide array of industry snapshots, special reports, and infographics covering biopharma, med‑tech, and emerging health topics. The collection highlights trends such as dynamic digital data analysis, the 2026 med‑tech outlook, mRNA vaccine research, and...

BioMarin Suffers Another Blow to Rare Disease Portfolio in Phase 3 Flop
BioMarin’s investigational enzyme replacement therapy BMN 401 lowered plasma inorganic pyrophosphate (PPi) in the Phase 3 ENERGY 3 trial for ENPP1 deficiency, but it did not translate into clinical benefit. The study enrolled almost 30 children aged 1‑12 and missed the primary Radiographic...
SERB to Pay Hansa €110M Upfront for EU Rights to Kidney Transplant Drug
SERB Pharmaceuticals will pay Hansa Biopharma €110 million (≈$119 million) upfront for exclusive rights to Idefirix across Europe, the Middle East and North Africa. The antibody‑cleaving enzyme, conditionally approved for highly sensitized kidney‑transplant patients, generated €18.7 million (≈$20 million) in 2025 sales but has...

Bayer’s Hyrnuo (Sevabertinib) Gains the US FDA Priority Review for 1L Treatment of HER2-Mutated NSCLC
Bayer’s Hyrnuo (sevabertinib) received FDA priority review for first‑line treatment of adults with locally advanced or metastatic non‑small cell lung cancer (NSCLC) harboring HER2 kinase‑domain activating mutations and no prior systemic therapy. The review follows promising interim data from Cohort F...
Common Asthma Drug May Turn Off Tumor 'Switch' Tied to Immunotherapy Resistance
A Northwestern Medicine study published in Nature Cancer shows that blocking the cysteinyl leukotriene receptor 1 (CysLTR1) with the asthma drug montelukast can reverse immunotherapy resistance in several aggressive cancers. Experiments in mouse models and analyses of human tumor samples demonstrated...
Vaccine Experts Debate Options to Combat Outbreak of Unusual Ebola Strain
The World Health Organization convened a closed meeting of vaccine experts after the Bundibugyo ebolavirus outbreak in the Democratic Republic of the Congo was declared a public‑health emergency. The outbreak has produced roughly 500 suspected cases and more than 130...

A New Genetically Modified Rice Could Improve Children’s Health. But Will It Be Grown?
The Philippines has issued its first biosafety permit for HIZ039, a genetically modified rice enriched with iron and zinc, aiming to combat childhood anemia and stunting. Laboratory data show the grain triples iron and more than doubles zinc compared with...

Protein Engineering and Testing Condensed Into One Day
Stanford researchers introduced MIDAS, a microbe‑independent deep assembly and screening method that reduces protein‑engineering cycles from weeks to a single day. By using PCR to assemble linear DNA fragments, the technique bypasses traditional bacterial cloning and plasmid preparation. The workflow...
Immune Checkpoint Regulation in Cancer Therapy and Evasion
The article reviews the multilayered regulation of immune checkpoint molecules, detailing genetic, epigenetic, transcriptional, post‑transcriptional, translational, and post‑translational mechanisms that shape checkpoint expression in tumors and immune cells. It highlights the recent clinical integration of LAG3‑targeted therapies as the newest...

MHRA’s Pound on Aligning Clinical Trial Regulation with Innovation
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) is rolling out clinical‑trial reforms aimed at more than just faster approvals. Interim director of innovation James Pound says the agency is pursuing international alignment and horizon‑scanning to anticipate emerging therapies....
New Math Method Inflates Alzheimer’s Drug Success by 29x
A Brown University research letter published in JAMA Neurology warns that the statistical technique known as quantile aggregation can dramatically overstate the efficacy of emerging Alzheimer’s drugs. Simulations showed the method inflates the perceived link between amyloid clearance and cognitive...

Coya 302 Gets Fast-Track Designation for ALS Treatment
Coya Therapeutics received FDA fast‑track designation for its experimental ALS drug Coya 302, a subcutaneous blend of low‑dose IL‑2 and CTLA‑4 immunoglobulin. The therapy is designed to boost regulatory T‑cells while suppressing inflammatory monocytes, addressing the immune imbalance that accelerates motor‑neuron...