
In a First, Scientists Are Rewinding Human Cells Back to a ‘Youthful’ State. Is This the Dawn of Immortality?
Scientists are advancing partial cellular reprogramming to reverse age‑related decline while preserving cell identity. YouthBio Therapeutics is preparing a first‑in‑human trial of its brain‑targeted YB002 program for Alzheimer’s after receiving FDA feedback. Parallel efforts such as Life Biosciences’ ER‑100 aim to rejuvenate optic‑nerve cells and restore vision. Researchers warn that precise control is essential, as animal data show tumor formation when reprogramming is mis‑timed.

FDA Vaccine Studies Censored by Trump Admin After Finding Benefits of Shots
The Food and Drug Administration, under the Department of Health and Human Services, blocked the publication of two internal studies that demonstrated the safety and efficacy of COVID‑19 vaccines, and prevented two Shingrix (shingles vaccine) abstracts from being presented at...

Tofersen, a New Treatment for A.L.S., Reverses Symptoms for Some
Tofersen, the first FDA‑approved therapy targeting the SOD1 genetic form of ALS, is showing functional gains in a subset of patients. In a case highlighted by the New York Times, 58‑year‑old Amanda Sifford’s lung capacity rebounded from 48% to 86%...
LTZ Therapeutics Secures $38M to Advance Myeloid Engager Immunotherapy Pipeline
LTZ Therapeutics announced an oversubscribed $38 million financing round led by GL Ventures, bringing its total capital to roughly $130 million since 2022. The funds will propel the Universal Myeloid Cell Engager (U‑MCE) platform, supporting the Phase 1 trial of lead bispecific antibody...

COVID-19 Prevention Gaps in Immunocompromised Patients: Muhammed Bilal Abid, MD
Dr. Muhammed Bilal Abid highlighted persistent COVID‑19 prevention gaps for immunocompromised patients, especially those with chronic lymphocytic leukemia (CLL). He cited the CANOPY trial, which reported zero symptomatic COVID‑19 cases over six months in a small CLL cohort receiving the monoclonal antibody...

Heavy-Chain BsAbs More Manufacturable than Light-Chains
Bispecific antibodies (BsAbs) have grown to 19 FDA approvals and roughly 250 candidates in development, but scaling their production remains a bottleneck. A recent study by Universidad Nacional Autónoma de México researchers compared six BsAb architectures and linked design to...

Researchers’ Spinout Focuses on Simplifying Viral Vector Purification
Researchers at North Carolina State University have spun out ChromaGenix to commercialize synthetic peptide ligands for affinity purification of viral vectors used in gene therapies. The peptide ligands are cheaper, more stable and less immunogenic than traditional protein ligands, cutting...

Moss Powering the Next Drug Frontier
Eleva is commercializing a moss‑based biomanufacturing platform that can produce complex glycoproteins difficult to express in traditional CHO or yeast systems. The German firm has advanced its first candidate, a recombinant alpha‑galactosidase for Fabry disease, into clinical trials and is...
Regulators Should Rely on Peers’ GMP Audits to Cut Inspection Burden
Biopharma manufacturing sites face an average of 2.68 GMP inspections per year, each lasting up to nine days, and preparation can take six months to a year. The International Federation of Pharmaceutical Manufacturers and Associations (IFPMA) argues that regulators should...

First U.S. Patients Treated With Microrobotic Surgery For Alzheimer’s
A microrobotic surgery trial for Alzheimer’s disease began at Baptist Health in Jacksonville, treating the first patient with moderate disease. Medical Microinstruments Inc. (MMI) plans to enroll 15 participants and monitor them for a year, aiming to clear cervical lymph‑node...
Financings for May 6, 2026
UK‑based Cytospire Therapeutics announced a £61 million (≈$82.7 million) Series A round to accelerate its EGFR‑targeted T‑cell engager pipeline for solid tumours. The capital backs multiple pre‑clinical programs and reflects strong investor confidence in next‑generation immunotherapies. Bio Korea 2026 highlighted a shift in...

Snowflake and Veeva Unlock Agentic AI in Life Sciences
Snowflake and Veeva announced a joint solution that links Veeva Vault’s read‑only data to Snowflake’s AI Data Cloud via the Openflow Connector. The integration lets life‑science firms run end‑to‑end analytics and agentic AI across clinical, safety, regulatory, quality and commercial...

Other News to Note for May 6, 2026
Cytospire Therapeutics announced a £61 million (≈$82.7 million) Series A round to advance its next‑generation T‑cell engager platform targeting the epidermal growth factor receptor (EGFR) in solid tumours. The funding will accelerate pre‑clinical and early‑clinical programs aimed at delivering more selective immunotherapies. Meanwhile,...

CoCoGraph AI Model Generates Molecules that Comply with Rules of Chemistry
Researchers at Universitat Rovira i Virgili have unveiled CoCoGraph, an AI diffusion model that generates synthetic molecules while strictly adhering to fundamental chemical rules. By progressively disordering and reconstructing real molecules, the system ensures valid bond counts and produces chemically...

Light-Controlled Gene Expression Platform Reportedly Doubles Standard Fed-Batch Manufacturing Performance
Prolific Machines announced a 21 g/L monoclonal antibody titer after a 15‑day intensified fed‑batch CHO run, more than double the typical sub‑10 g/L industry benchmark. The company’s optogenetic platform uses light to dynamically regulate gene expression, giving manufacturers real‑time control over protein...

Dark Proteome Yields 1,785 New Microproteins that Could Reshape Disease Research
Scientists have identified 1,785 microproteins hidden in the human dark proteome, a roughly 10% increase to the known protein catalog. These tiny proteins, 65% under 50 amino acids, were uncovered by mining 95,520 mass‑spectrometry experiments and 3.7 billion spectra. The team...

A New Kind of CRISPR Could Treat Viral Infection and Cancer by Shredding Sick Cells' DNA
Researchers have engineered a novel CRISPR protein, Cas12a2, that acts as a molecular shredder, destroying DNA in cells that express a specific RNA trigger. In vitro, Cas12a2 cut the growth of KRAS‑mutant lung‑cancer cells by 50% and eliminated over 90%...

J&J’s Ottava Robot Meets Goals in Gastric Bypass Study
Johnson & Johnson reported that its investigational Ottava robotic system met safety and efficacy endpoints in a 30‑patient gastric bypass study, achieving an average 30‑pound weight loss within 30 days and no device‑related adverse events. All procedures were completed robotically...
IV Ketamine Shows Rapid Benefits for Suicide Risk, Depression in Major Depressive Episodes
A new meta‑analysis of 26 randomized trials involving 1,166 patients shows that intravenous ketamine rapidly alleviates both suicidal ideation and depressive symptoms in major depressive episodes. Single infusions cut suicidal scores by an SMD of –0.69 within 24 hours and depressive...

A Low-Cost Rotavirus Test Could Save Childrens’ Lives in Nigeria
Researchers at Obafemi Awolowo University have created a low‑cost, point‑of‑care rotavirus test that works without electricity or specialized training. The nanobead‑based kit showed 88% sensitivity in pilot hospitals, beating the standard ELISA test’s 60% sensitivity. Rotavirus accounts for nearly half...

The VESALIUS-CV Trial
In the VESALIUS-CV trial, researchers evaluated the impact of adding the PCSK9 inhibitor evolocumab to standard lipid‑lowering therapy in patients at high cardiovascular risk who had never experienced a myocardial infarction or stroke. Over a median follow‑up of 3.2 years,...
Cyclin E1 and CCNE1 Shift in Ovarian Cancer Post-PARP
The British Journal of Cancer study shows high‑grade serous ovarian carcinoma tumors develop resistance to PARP inhibitors by up‑regulating Cyclin E1 and amplifying the CCNE1 gene. Researchers compared pre‑ and post‑treatment samples using FISH and IHC, finding a significant rise in...

Novel Psychedelic Compound 25C-NBF Shows Rapid Antidepressant Effects without Addictive Traits
Researchers reported that the synthetic psychedelic 25C‑NBF rapidly induces dendritic growth and reverses depressive behaviors in male rodents after a single dose. The compound binds tightly to the serotonin 5‑HT2A receptor while showing minimal activity at 5‑HT2B, reducing heart‑valve risk....

Scientists Create "Living Plastic" That Can Self-Destruct on Command
Scientists at Shenzhen Institutes of Advanced Technology have engineered a "living plastic" that embeds dormant Bacillus subtilis spores. The spores stay inactive during normal use but can be triggered to awaken and produce two enzymes that sequentially break down polycaprolactone,...

Decoding Proteomic Changes in Pediatric Brain Injury
Researchers Khalifah and Guerguerian published a comprehensive proteomic analysis of severe pediatric traumatic brain injury in *Pediatric Research*. Using high‑throughput mass spectrometry, they mapped thousands of proteins in brain tissue and cerebrospinal fluid across acute, sub‑acute, and chronic phases. The...

STAT+: Novo Nordisk’s Less-Bad News on Its Wegovy Pill Boosts Earnings and Share Price
Novo Nordisk reported first‑quarter results that showed a modest improvement in its outlook for the obesity drug Wegovy. The company narrowed its adjusted sales decline forecast to 4%‑12% for the year, up from a prior 5%‑13% range, and aligned its...

BIOTECanada Responds to CDA-AMC Guidance for Incorporating Impacts on Informal Caregivers and Productivity Outcomes in Economic Evaluations.
BIOTECanada, together with Innovative Medicines Canada, submitted a formal response to the Canadian Drug Agency’s draft guidance on incorporating informal caregiver health‑related quality of life and productivity outcomes into economic evaluations. The association recommends expanding the societal perspective to all...
Sustaining Science: Preserving Knowledge Amid Big Data
The bioengineering community is publishing a wave of data‑intensive studies that span proteomics, photonics, plasma actuation, epigenetics, and oncology. Each article, released on May 6, 2026, demonstrates how massive datasets are unlocking new scientific insights. At the same time, the field grapples...

Top Biotech Deals in April 2026
April 2026 marked a flurry of blockbuster biotech mergers, highlighted by Sun Pharma’s $11.75 billion acquisition of Organon, giving it a foothold in women’s health across 140 countries. Eli Lilly emerged as the most active buyer, spending roughly $11.5 billion on four...

Nobel Laureate Jules Hoffmann Heads Comprehensive Review Celebrating 40 Years of Toll-Like Receptor Research
A new review in *Immunity & Inflammation*, led by Nobel laureate Jules Hoffmann and collaborators, chronicles 40 years of Toll‑like receptor (TLR) research. It traces the field from the discovery of the Drosophila Toll gene to the mapping of the...

Early DNA Methylation Links to Infant Respiratory Infections
Scientists published an epigenome‑wide association study in Pediatric Research linking DNA methylation at the TRIM6 and TTC23 gene promoters in newborns to respiratory infection risk during the first year of life. The prospective analysis of peripheral blood samples showed that...

BriaCell Therapeutics Reports US FDA IND Clearance to Initiate P-I/II Trial of Bria-BRES+ in Breast Cancer
BriaCell Therapeutics announced that the U.S. Food and Drug Administration has granted IND clearance to launch a combined Phase I/II study of its Bria‑BRES+ therapy in patients with metastatic breast cancer. Preclinical results presented at AACR indicate Bria‑BRES+ stimulates both...
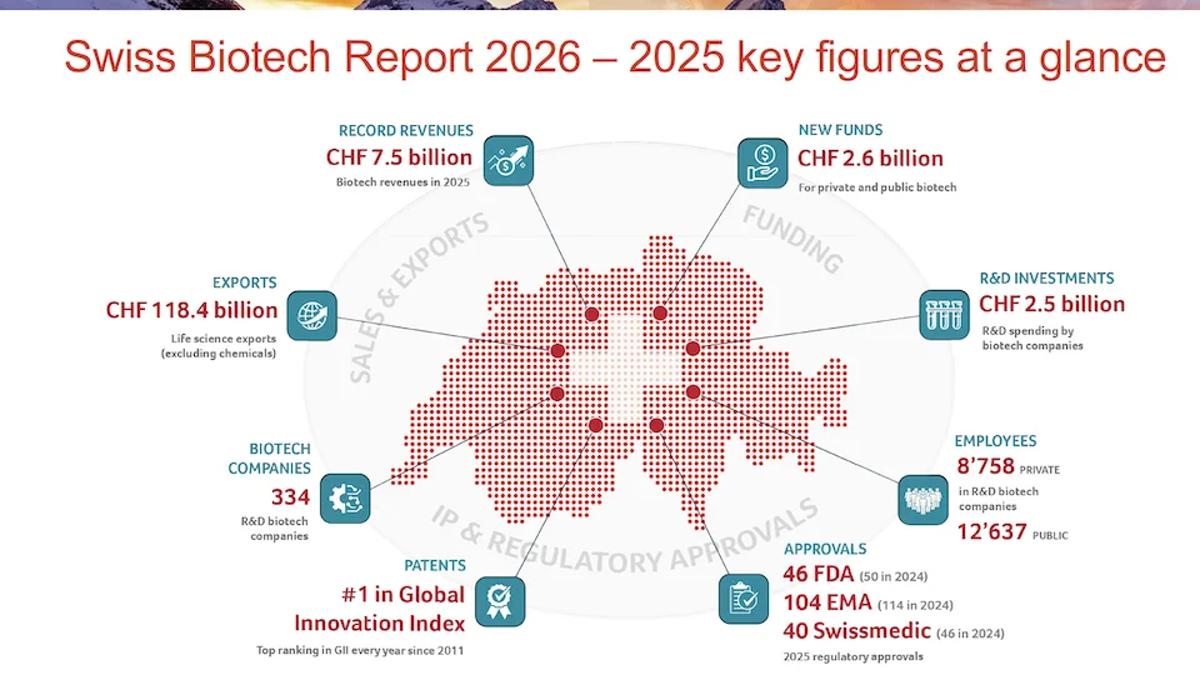
Switzerland’s Biotech Sector Defies Tough Markets with Record Revenue
Switzerland’s biotech sector posted a record CHF 7.5 billion ($8.3 billion) in revenue for 2025, up from CHF 7.2 billion the year before, driven by more market‑ready products and strong CDMO demand. Capital inflows rose 2.1% to CHF 2.6 billion ($2.9 billion), with privately financed firms leading the...

FDA Approves Selpercatinib for Medullary Thyroid Cancer with a RET Mutation
The FDA granted traditional approval on September 27, 2024, for Eli Lilly’s selpercatinib (Retevmo) to treat adult and pediatric patients two years and older with advanced or metastatic medullary thyroid cancer (MTC) harboring a RET mutation. The decision follows accelerated approvals...
MiR-205a Suppresses CDH11, Halting Chondrocyte Growth
Researchers have identified that microRNA miR‑205a suppresses the expression of cadherin‑11 (CDH11) in chondrocytes, disrupting the Wnt/β‑catenin signaling pathway essential for cartilage formation. Experimental overexpression of miR‑205a reduced CDH11 levels and halted chondrocyte differentiation, while inhibition of miR‑205a restored CDH11...

SCIENTURE Announces Third Patent Grant for ARBLI™ (Losartan Potassium) Oral Suspension, Strengthening Long-Term Market Exclusivity Through 2041
Scienture Holdings announced that the U.S. Patent and Trademark Office granted a third patent (U.S. Patent No. 12,605,365) for its ARBLI™ losartan potassium oral suspension. The new patent, issued April 21, 2026, extends exclusive protection through October 7, 2041, reinforcing the company’s IP moat. ARBLI™...

Novelty Nobility Expands AGC Biologics Deal to Take Product Candidate Through GMP Manufacturing
Korea‑based Novelty Nobility has expanded its contract with CDMO AGC Biologics to move its bispecific antibody NN4101 through process development and GMP manufacturing at AGC’s Chiba, Japan facility. Cell‑line development was completed in Copenhagen and will be transferred to Chiba...
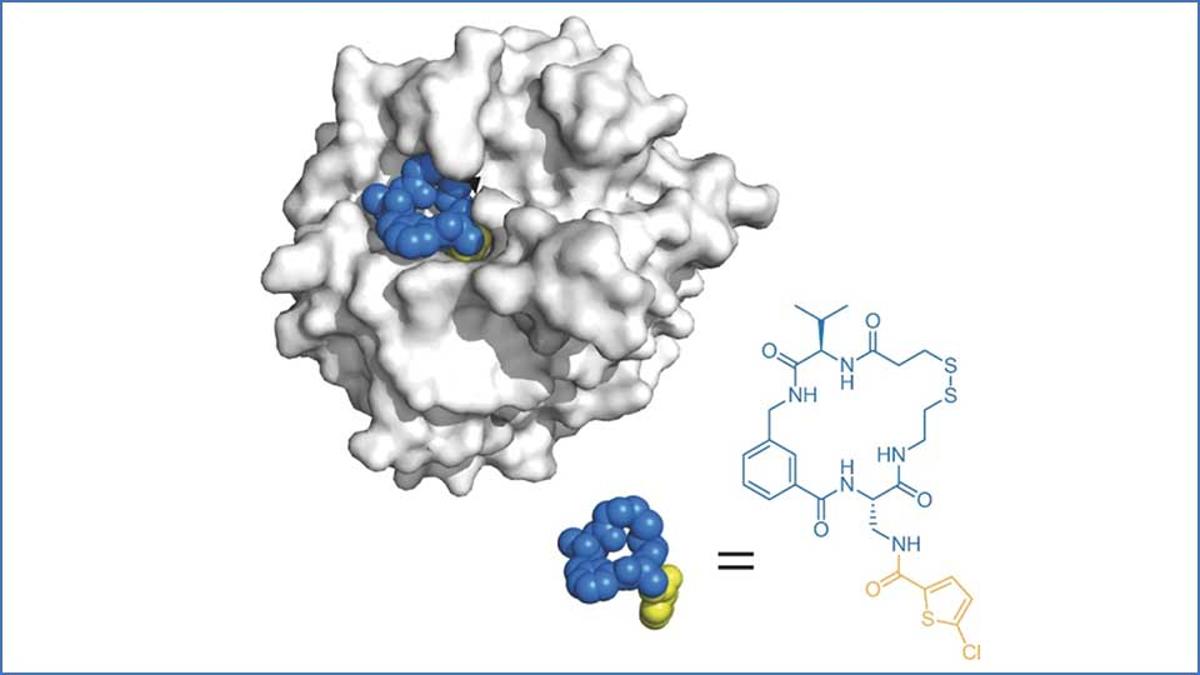
Macrocycles: Big Is the New Beautiful
Macrocycles are gaining traction as a hybrid drug modality that combines the high specificity of biologics with the oral accessibility of small molecules. Advances in synthetic chemistry, AI‑driven design, and DNA‑encoded libraries now enable rapid exploration of billions of candidates,...

Backed by USD 4.3M Public Funding, Spermotile Seeks Asia Partners for AI Fertility Innovation
Spermotile, a medtech startup developing an AI‑driven sperm‑selection platform, has secured roughly $4.3 million in public funding from the EU and Norway. The company is showcasing its technology at Echelon Singapore 2026 to court investors, manufacturing partners, and CROs across Southeast...

Alebund Pharmaceuticals Completes Patient Enrollment in P-III (RESPOND-2) Trial of AP301 for Hyperphosphatemia
Alebund Pharmaceuticals announced that patient enrollment is complete for its global Phase III RESPOND‑2 trial of AP301, a novel therapy for hyperphosphatemia in dialysis patients. The study enrolled 282 participants—138 in the United States and 144 in China—exceeding the planned 264....
DDW 2026: Key Readouts From the Conference
Digestive Disease Week 2026 in Chicago showcased several pivotal GI‑focused readouts. AbbVie reported real‑world Skyrizi data showing steroid use fell from 34% to 7% after 52 weeks and 77% of Crohn’s patients reported improved quality of life. Johnson & Johnson’s...

4basebio Announces Lease of an Innovation Hub and Manufacturing Facility in Cambridge, UK
4basebio PLC announced a lease for a 26,500 sq ft innovation hub and manufacturing facility in Cambridge, UK, slated to open in late summer 2026. The site will feature 7,500 sq ft of specialized laboratory space with 15 labs, advanced biosafety equipment, and a...
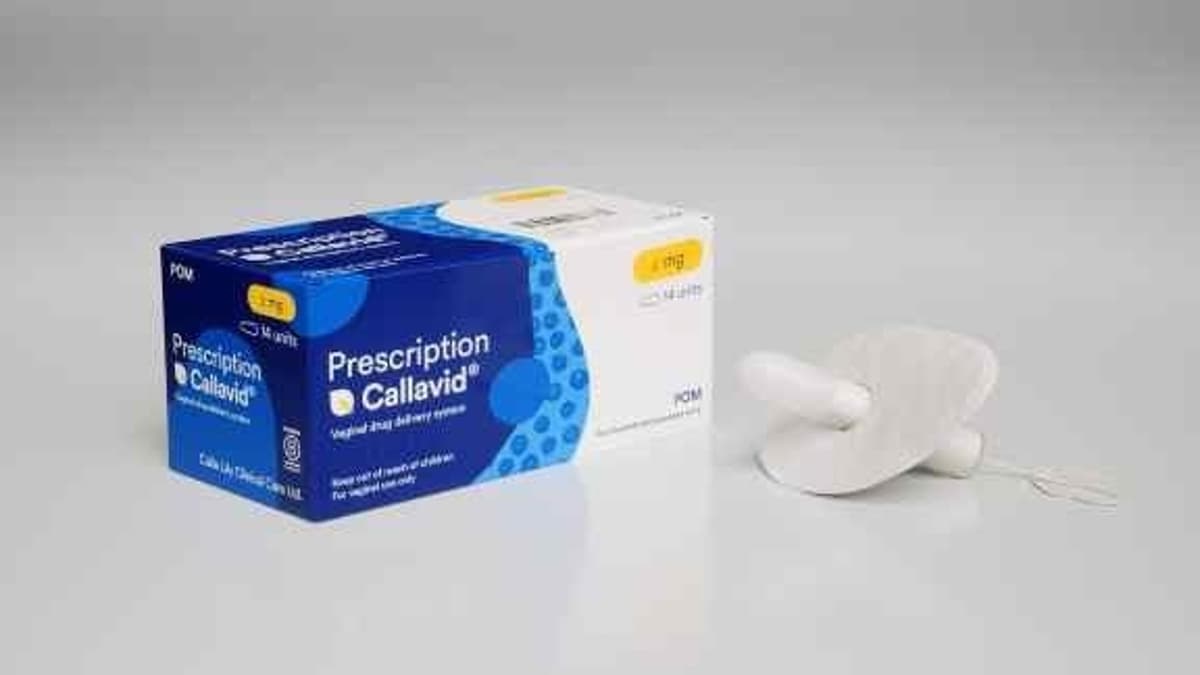
Calla Lily Clinical Care Doses First Patients in Clinical Trial for Intravaginal Drug Delivery Platform for Threatened Miscarriage
Calla Lily Clinical Care has begun dosing the first participants in the FREEDOM clinical trial, testing its 400 mg intravaginal progesterone product Callavid. The NIHR‑funded study targets women with luteal phase insufficiency, a condition linked to threatened miscarriage and infertility. Callavid’s...

CellCentric Raises $220M to Get Multiple Myeloma Pill to Market
CellCentric, a 22‑year‑old transatlantic biotech, closed a $220 million Series D round to accelerate development of its oral multiple myeloma drug. The capital will fund the completion of Phase III trials, scale manufacturing, and prepare regulatory filings. The therapy is designed for patients...
Pfizer, Lilly, More Report Q1, FDA Names Acting CBER Director and an ALS Awakening
Pfizer posted first‑quarter earnings that topped consensus estimates, yet analysts deemed the beat insufficient given higher expectations for the New York‑based giant. Eli Lilly delivered a striking 56% revenue surge, though the company noted that price reductions on several products capped its...

Bayer to Buy Perfuse Therapeutics for $300M Upfront
Bayer announced a $300 million upfront payment to acquire ophthalmology biotech Perfuse Therapeutics, securing its mid‑stage program aimed at the leading causes of blindness. The transaction represents Bayer’s first pharmaceutical merger‑and‑acquisition in several years, signaling a renewed focus on specialty eye...

Signadori Bio Raises €11M to Advance Tumour Platform
Signadori Bio announced an €11 million ($11.9 million) seed extension to fund its in‑vivo monocyte engineering platform for solid tumours. The Series A round, led by Taiho Ventures with Sofinnova and Invivo Partners, will support lead candidate nomination, platform refinement, and team expansion...

Novo Nordisk's Q1 in Four Words: Pills, Pricing, Payments and Pressure
Novo Nordisk’s first‑quarter performance centered on its obesity drug Wegovy, which now serves roughly one million patients in the United States. The company is navigating heightened pricing pressure as insurers push for larger rebates while regulators scrutinize the drug’s cost....

Sickle Cell Disease After Casgevy: Seven Companies to Watch in 2026
Casgevy’s $2.2 million, ex vivo CRISPR cure proved sickle cell disease can be edited at its genetic root, but its complex manufacturing and conditioning limit broad access. In response, a wave of innovators is targeting simpler, safer, and more scalable solutions—from Beam’s...

Cytospire Therapeutics Secures £61m to Advance Cancer Treatment
Cytospire Therapeutics, a UK biotech, closed a £61 million (≈$77 million) Series A round to accelerate its multispecific engager antibody platform. The capital will fund the development of its lead candidate, CYT X300, aimed at EGFR‑positive solid tumours such as colorectal, head‑and‑neck and non‑small‑cell...