
Roche to Acquire PathAI for ~$1.05B
Roche has signed a definitive agreement to acquire AI‑driven diagnostics firm PathAI for an estimated $1.05 billion, comprising $750 million upfront and up to $300 million in milestones. The deal, slated to close in the second half of 2026, builds on a partnership that began in 2021 and expanded in 2024 to co‑develop AI‑enabled companion diagnostic algorithms. By integrating PathAI’s platform, Roche aims to accelerate automation in digital pathology and deepen its precision‑medicine portfolio. The acquisition also positions Roche to offer AI‑enhanced support for clinical trials and translational research.

Scientists Discover Why Ozempic Works Better for some People
GLP‑1 drugs like Ozempic improve blood sugar and weight loss, but response varies. A Japanese study of 92 type‑2 diabetes patients found that those who overeat in response to external cues (appearance or smell) achieved greater weight loss and glucose...

What’s Next for IVF
Advances in IVF are moving beyond traditional lab techniques toward AI, robotics, and novel embryo‑delivery devices. Researchers at the Carlos Simon Foundation have built a “Transfer Direct” system that injects embryos into the uterine lining, while AI platforms such as...

Halozyme and Oruka Sign Agreement for Hypercon Technology
Halozyme’s Hypercon technology has entered an exclusive global licensing agreement with Oruka Therapeutics to develop ORKA-001, a biologic targeting the IL‑23p19 subunit for psoriasis and related inflammatory diseases. The deal provides Halozyme with an upfront cash payment, future milestone fees,...

Global Drugmakers Eye More China Biotech Deals After Record Year
Global pharmaceutical firms are intensifying their pursuit of Chinese biotech partners after 2025 set a record $135.7 billion in out‑licensing deals, nearly three times the 2024 total. Executives from Johnson & Johnson and Merck highlighted plans to expand on‑the‑ground resources and...
Fibrosis, IPF and the Search for Better Therapies
The BioSpace "Denatured" podcast episode spotlights the persistent challenges of idiopathic pulmonary fibrosis (IPF) and broader fibrotic diseases. Host Jennifer C. Smith‑Parker interviews Georg Vo Beiske, CEO of Tribune Therapeutics, and Jonas Hallén, CMO of Calluna Pharma, to dissect current treatment gaps and...
Macrophage‐Inspired Nanomedicines: Harnessing Innate Biology for Precision Therapy
The review outlines macrophage‑inspired nanomedicines that exploit three delivery platforms—whole macrophage carriers, macrophage‑membrane‑coated nanoparticles, and macrophage‑derived exosomes. By leveraging the cells’ natural homing, barrier‑penetrating and immunomodulatory traits, these systems aim to improve targeting of cancers, atherosclerosis and other inflammatory diseases....

Increased Capital Boosts Biotech R&D Job Postings, Employment
Biotech R&D job postings on BioSpace jumped 21% year‑over‑year in April, marking the longest month‑over‑month rise since mid‑2024. A CBRE report shows biotech employment reached a record 295,600 in February after five consecutive months of growth, the longest streak since...

Artera Secures FDA Clearance for AI-Based Breast Cancer Risk Tool
The U.S. Food and Drug Administration granted clearance to ArteraAI Breast, an AI‑driven digital pathology tool that combines digitized histopathology slides with clinical data to generate a risk score for distant metastasis in early‑stage hormone‑receptor‑positive/HER2‑negative breast cancer. The system stratifies...

WA Biotech Duo Takes Aim at Diabetes Beyond Ozempic
Australian biotech duo ProGenis Pharmaceuticals and Syngenis Labs are developing RNA‑based antisense therapies that target insulin resistance, a root cause of type‑2 diabetes, to complement GLP‑1 drugs. Syngenis is building Australia’s first GMP‑grade synthetic DNA/RNA manufacturing facility, expected to be...

PYC Therapeutics Advances PKD Program with Phase 1b Multiple Ascending Dose Study Initiation
PYC Therapeutics has dosed the first patient in a Phase 1b multiple‑ascending‑dose (MAD) study of its PKD candidate PYC‑003, targeting safety, tolerability and early efficacy signals such as urinary PC1 protein, total kidney volume and eGFR. Results from the earlier Phase 1a...
The Peptide Problem: Hype Is Outrunning the Evidence
Health Canada has warned Canadians against buying and injecting unauthorized peptide products such as BPC‑157, CJC‑1295, ipamorelin, TB‑500 and retatrutide, and has already seized several shipments. Influencers on Instagram and TikTok are promoting these compounds for anti‑aging, weight loss and...

Akebia Therapeutics Inc (AKBA) Q1 2026 Earnings Call Transcript
Akebia Therapeutics reported 2025 total revenue of $236.2 million, a 47% increase year‑over‑year, driven by the commercial launch of its oral HIF‑PH inhibitor Vafseo and higher sales of the phosphate binder Auryxia. Vafseo now reaches 290,000 dialysis patients across 24 organizations,...

Monte Rosa Therapeutics Inc (GLUE) Q1 2026 Earnings Call Transcript
Monte Rosa Therapeutics reported Q1 2026 worldwide revenue of $141.2 million, a 14.3% year‑over‑year increase, driven primarily by strong U.S. sales of its AtriClip and Cryosphere product lines. Adjusted EBITDA rose to $17 million, nearly doubling the prior‑year quarter, while gross margin...
Ironwood Pharmaceuticals Inc (IRWD) Q1 2026 Earnings Call Transcript
Ironwood Pharmaceuticals reported 2025 LINZESS U.S. net sales of $865 million, matching guidance despite a 6% price erosion tied to Medicare Part D changes. Prescription demand grew 11% for the second year, maintaining a 45% market share in the IBS‑C and chronic...

There Is No Vaccine for Deadly Hantavirus: What that Means for Future Outbreaks
An outbreak of Andes hantavirus on the cruise ship MV Hondius left three confirmed cases and three deaths, highlighting the deadly potential of the rodent‑borne virus. The World Health Organization confirmed the strain has no approved treatments or vaccines. Virologist...

NewAmsterdam Pharma Company NV (NAMS) Q1 2026 Earnings Call Transcript
NewAmsterdam Pharma reported Q1 2026 net revenue of $45.1 million, up from $42 million a year earlier, while cash rose to $308 million and the company remains debt‑free. U.S. VASCEPA captured 48% of the IPE market, a 6‑point gain, and branded prescriptions grew...

Lantheus Holdings Inc (LNTH) Q1 2026 Earnings Call Transcript
Lantheus Holdings reported Q1 2026 revenue of $377.3 million, a 1.2% year‑over‑year increase and 8.6% when excluding divested SPEC revenue. Oncology sales from PYLARIFY fell 6.5% despite a 5.8% volume rise, while cardiology product DEFINITY grew 6.8% and retained over 80%...

PTC Therapeutics Inc (PTCT) Q1 2026 Earnings Call Transcript
PTC Therapeutics reported record Q1 2026 product revenue of $226 million, driven by a $125 million Sephience launch that grew 36% quarter‑over‑quarter. The company lifted full‑year product revenue guidance to $750‑$850 million and total revenue to $1.08‑$1.18 billion, citing strong U.S. uptake, the first...

INmune Bio Inc (INMB) Q1 2026 Earnings Call Transcript
INmune Bio reported Q1 2026 results showing a cash balance of $21.4 million, sufficient to sustain operations through Q1 2027, and a narrowed net loss of $5.4 million. The company highlighted significant progress on its CORDStrom platform, including approval of the U.K. Pediatric Investigation...

Guardant Health Inc (GH) Q1 2026 Earnings Call Transcript
Guardant Health reported Q1 2026 revenue of $302 million, a 48% year‑over‑year increase and the fastest annual growth in five years. Oncology revenue rose 36% to $205 million, driven by 86,000 tests—a 47% volume jump—while its Shield screening test generated $42 million from...

Zymeworks Inc (ZYME) Q1 2026 Earnings Call Transcript
Zymeworks reported a PDUFA deadline of August 25, 2026 for its zanidatumab HER2‑positive gastroesophageal adenocarcinoma (GEA) indication, with a supplemental BLA filed in China. The company stands to receive up to $265 million in milestone payments contingent on U.S. and Chinese...
Delcath Systems Inc (DCTH) Q1 2026 Earnings Call Transcript
Delcath Systems reported Q1 2026 revenue of $25 million, a 26% increase year‑over‑year, driven primarily by $23.3 million from its HEPZATO kit. The company recorded a net loss of $1.1 million but posted adjusted EBITDA of $3.4 million and reaffirmed a 2026 revenue target...
Absci Corp (ABSI) Q1 2026 Earnings Call Transcript
Absci Corp (ABSI) reported Q1 2026 revenue of $200 K while R&D expenses rose to $19.3 M as the company advances its lead prolactin‑receptor antibody ABS‑201. The Phase 1/2a androgenic alopecia trial is on track, with safety data due next month and interim...

Harmony Biosciences Holdings Inc (HRMY) Q1 2026 Earnings Call Transcript
Harmony Biosciences reported record Q1 product revenue of $243.8 million, a 21% increase year‑over‑year, driven by sustained demand for its Wakix sleep‑wake therapy. The company added roughly 400 patients per quarter, reaching about 8,500 on Wakix, and reaffirmed 2026 revenue guidance...

Lexicon Pharmaceuticals Inc (LXRX) Q1 2026 Earnings Call Transcript
Lexicon Pharmaceuticals reported Q1 2026 revenue of $5.5 million, largely from a $4.3 million Novo Nordisk licensing deal and INPEFA sales. The company cut its quarterly net loss to $15.5 million and reduced annual operating expenses by $129.5 million, boosting cash to $125 million after...

Crinetics Pharmaceuticals Inc (CRNX) Q1 2026 Earnings Call Transcript
Crinetics reported $10.3 million net product revenue from Palsonify in Q1 2026, driven by 232 new patient enrollments and a prescriber base that grew to 263 unique physicians. Treatment‑naive patients rose to 15 % of enrollments, indicating expanding first‑line use. Reimbursement coverage...

Arrowhead Pharmaceuticals Inc (ARWR) Q2 2026 Earnings Call Transcript
Arrowhead Pharmaceuticals reported accelerating uptake of its newly launched triglyceride‑lowering therapy REDEMPLO, surpassing 400 prescriptions with a 40% surge in the last month. The company announced a price reduction to $45,000 per patient per year and secured payer policies that...

Scholar Rock Holding Corp (SRRK) Q1 2026 Earnings Call Transcript
Scholar Rock Holding Corp reported $368 million in cash and secured a $550 million debt facility, providing ample liquidity for 2026 initiatives. The FDA completed a reinspection of the Catalent Indiana site with no further requests, clearing the path for an imminent...

Organogenesis Holdings Inc (ORGO) Q1 2026 Earnings Call Transcript
Organogenesis Holdings reported a 58% drop in Q1 net product revenue to $36.3 million, driven primarily by a 63% plunge in advanced wound‑care sales after CMS reimbursement policy changes created clinician confusion. The company recorded a GAAP operating loss of...
SIGA Technologies Inc (SIGA) Q1 2026 Earnings Call Transcript
SIGA Technologies reported Q1 2026 revenue of roughly $6 million, a pretax loss of $5 million and a cash balance of $146 million with no debt. The company announced a $13 million oral TPOXX order for an Asia‑Pacific customer in Q2 and sealed an...

Insmed Inc (INSM) Q1 2026 Earnings Call Transcript
Insmed reported that its newly launched Brinsupri generated $144.6 million in its first full quarter and now projects at least $1 billion in U.S. revenue for 2026. The company highlighted strong early adoption, with over 11,500 new patients in 2025, 90%+ payer...

Science Spotlight: New Prime Editing Tools for Large DNA Insertions
BioCentury’s website employs a tiered cookie framework that classifies cookies into strictly necessary, functional, marketing, advertising, and analytics groups. Strictly necessary cookies power core services such as authentication, registration, and user‑preference management, while functional cookies enhance site personalization. Marketing and...

From Discovery to GMP: Building Scalable Cell Therapy Manufacturing
Genetic Engineering & Biotechnology News and ElevateBio released an eBook titled “From Discovery to GMP: Building Scalable Cell Therapy Manufacturing.” It argues that the next growth phase for cell and gene therapies hinges on integrating therapeutic design with manufacturing to...

Membrane Complex Aids Rock-Eating Microbes in Converting Carbon Dioxide to Biomass
Researchers at the Universities of Potsdam and Marburg have detailed the structure of the DAB2 membrane complex in the sulfur bacterium Halothiobacillus neapolitanus. The complex enables lithoautotrophic microbes to convert CO₂ directly into bicarbonate (HCO₃⁻) using the cell’s membrane potential,...

An Essential Framework to Navigating Technical Challenges in Contemporary OSD Development
Oral solid dosage (OSD) development faces mounting technical hurdles as biopharma pipelines become richer in poorly soluble compounds, with 70‑90% of new small‑molecule candidates classified as BCS Class II or IV. Early‑risk identification—through comprehensive physicochemical profiling, excipient compatibility testing, and Quality...

The BioPharm Brief: Precision, Immunotherapy, Expansion
Madrigal Pharmaceuticals is licensing Arrowhead’s RNA‑interference candidate ARO‑PNPLA3 to broaden its precision‑medicine pipeline for metabolic dysfunction‑associated steatohepatitis (MASH). LTZ Therapeutics secured $38 million to push forward myeloid‑engager immunotherapies that activate innate immune cells against cancer. Amgen and Eli Lilly announced major U.S....

Death-Defying Protein Found in Tardigrades Preserves Synthetic Cells
Researchers at the University of Michigan and the University of Chicago have shown that the tardigrade‑derived protein CAHS12 can protect synthetic cells from dehydration. By embedding CAHS12 into lipid‑bound vesicles, the engineered cells survived drying and regained protein‑making activity after...

DNA-Guided CRISPR Flips Gene Editing Script, Opening a New Path for Precise Diagnosis and Antivirals
Researchers at Hong Kong University of Science and Technology have created the first DNA‑guided CRISPR‑Cas12a system that can programmatically target and cleave RNA. The new platform, called SLEUTH, combines the DNA‑guided enzyme with isothermal amplification to achieve attomolar‑level detection of...

What’s New in Clinical Trial Innovation
The FDA’s Center for Clinical Trial Innovation (C3TI) publishes a newsletter that highlights new developments, opportunities, and initiatives in clinical‑trial innovation. Readers can subscribe via a public.govdelivery.com form to receive updates directly in their inbox. The site also provides free...
The Human Genome Encodes for a New Category of Molecule
Scientists have identified a previously unrecognized class of molecules encoded within the human genome, arising from short open reading frames once deemed non‑coding. These micro‑proteins, often called micropeptides, exhibit distinct biochemical activity and appear to regulate cellular pathways. Early experimental...

These Companies Help Parents Try to Pick Their Babies' Traits. Experts Are Wary
Companies such as Herasight, Orchid Health and Nucleus Genomics now offer polygenic embryo screening that estimates disease risk and predicts traits like height, BMI, longevity and IQ. The service relies on polygenic risk scores derived from DNA samples of parents...
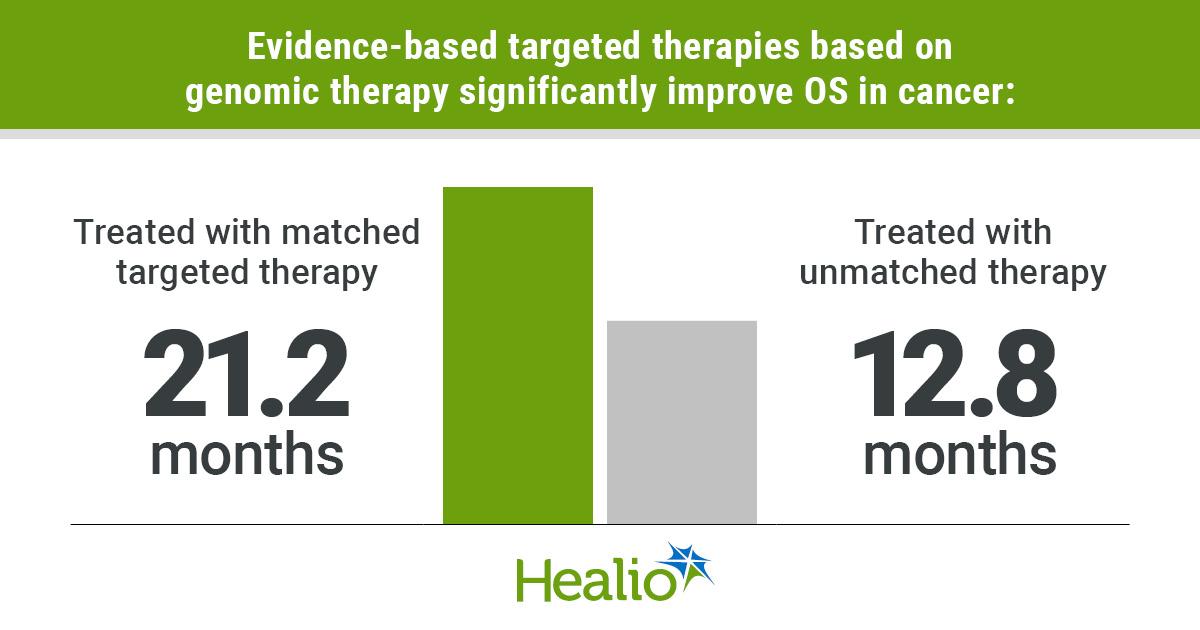
Treating Cancer Based on Mutation Alone Does Not Improve Survival
A large Australian study of 3,383 advanced‑cancer patients found that targeted therapies approved for a specific mutation within the same tumor type improved overall survival by roughly 40%, while using a drug solely because of a shared mutation across different...

OECD Blog Item Explores Why Biotech Start-Ups Lag in Europe
The OECD’s May 6 blog highlights that Europe continues to fall behind the United States and Asia in biotech start‑up formation, patenting, and venture‑capital funding. It points to the European Commission’s December 2025 Biotech Act, which introduces regulatory sandboxes, pre‑submission consultations, a...

Bayer Sees Opportunity With $300M Acquisition of Eye Drug Biotech
Bayer announced a $300 million upfront acquisition of Perfuse Therapeutics, a clinical‑stage biotech developing PER‑001, an endothelin‑receptor blocker delivered via a six‑month intravitreal implant. Phase 2 trials showed significant vision improvement in glaucoma and diabetic retinopathy, positioning the drug as a potential...

HOPE Supports Tailored Approach to BP After Stroke Thrombectomy
The HOPE trial, presented at the European Stroke Organisation Conference 2026, tested a reperfusion‑guided blood‑pressure strategy after endovascular thrombectomy for acute ischemic stroke. Patients whose post‑procedure mTICI score was 2b were targeted to a systolic BP of 140‑160 mm Hg, while those...
Brolucizumab Superior in Preserving Visual Acuity in Proliferative Diabetic Retinopathy
A phase‑3 CONDOR trial of 689 adults with proliferative diabetic retinopathy (PDR) showed that intravitreal brolucizumab outperformed panretinal laser photocoagulation (PRP) in preserving visual acuity over 54 weeks. The brolucizumab arm gained a mean BCVA change of +0.2 letters versus...

Light without Electricity? Glowing Algae Could Make It Possible
University of Colorado Boulder researchers discovered that acidic (pH 4) or basic (pH 10) solutions can sustain the bioluminescence of Pyrocystis lunula algae for up to 25 minutes. By embedding the algae in a water‑based hydrogel and 3D‑printing it into shapes, they...

Clinical Trials Day
Clinical Trials Day on May 20 commemorates James Lind’s 1747 scurvy experiment, widely regarded as the first randomized clinical trial. The observance highlights the enduring role of trials in generating the safety and efficacy data that underpin FDA drug approvals. FDA’s...

BsUFA IV: Fiscal Years 2028-2032
The FDA announced the kickoff of the fourth Biosimilar User Fee Act (BsUFA IV) reauthorization, covering fiscal years 2028‑2032. A public meeting was held on December 3, 2025 to launch the process, following the 2022 law that reauthorized BsUFA II. BsUFA III expires in September 2027,...