
An Octopus Probe for High-Performance >1,300 Nm NIR-II Fluorescence Molecular Imaging of Cancer
Researchers at Stanford introduced the Octopus (OCTP) probe, a modular NIR‑II fluorescence agent that emits beyond 1,300 nm and targets the folate receptor. In pre‑clinical mouse studies, OCTP delivered markedly higher tumor‑to‑background ratios and brighter tumor signals than the FDA‑approved Cytalux probe. Its multi‑arm PEG architecture promotes rapid, sustained tumor uptake, swift renal clearance, and minimal off‑target toxicity. The technology promises more accurate intra‑operative margin detection, potentially reducing residual disease and preserving healthy tissue.

Enhanced Mechanical Strength and Controlled Degradation of PLA/ZnO Nanoparticle Composites for Biodegradable Implants
Researchers have engineered polylactic acid (PLA) composites reinforced with zinc oxide (ZnO) nanoparticles in two morphologies—nanospheres and nanorods—to create biodegradable implant materials. Tensile testing revealed that PLA/ZnO nanosphere composites reach 25.20 MPa, outperforming the nanorod variant at 22.98 MPa. Incorporating ZnO accelerated...
Vitamin D Boosts Breast Cancer Treatment Success by 79%
Researchers at São Paulo State University conducted a randomized trial of 80 women over 45 undergoing neoadjuvant chemotherapy for breast cancer. One half received 2,000 IU of vitamin D daily, while the other took a placebo. After six months, 43% of the...
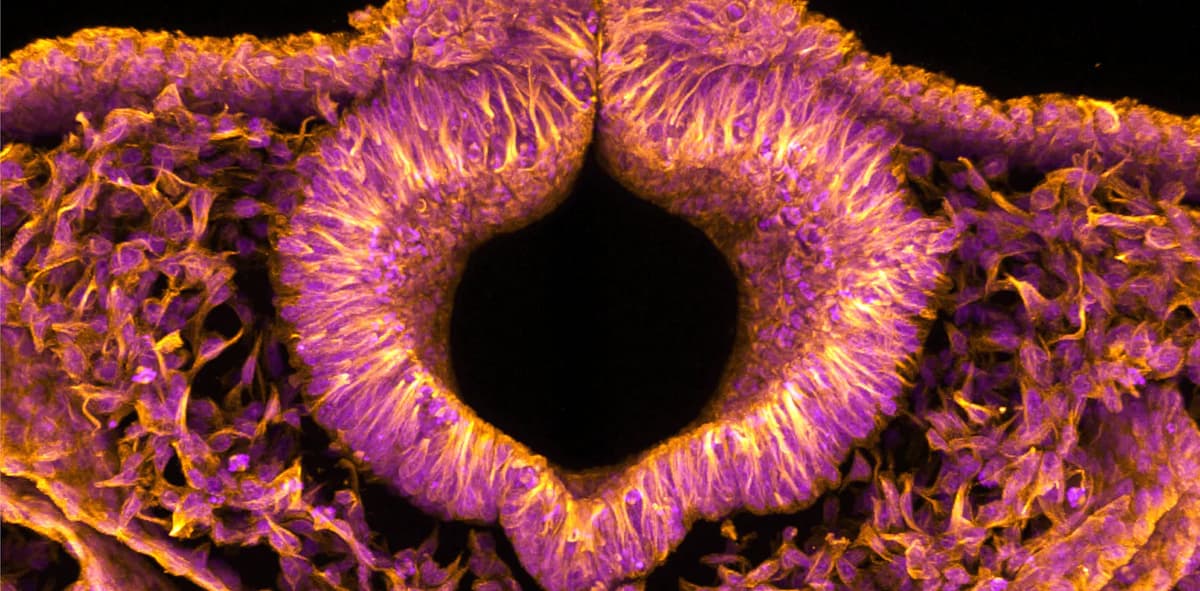
Fluorescent Quail Embryos Could Help Solve Serious Birth Defects in Humans
Researchers have engineered a fluorescent quail embryo that lights up cells during the first 72 hours of development, allowing live, high‑resolution imaging of neural tube formation. Using confocal microscopy, they tracked individual cell movements and discovered that loss of the...

How Big Is Organon’s Biosimilar Push for Sun Pharma?
Sun Pharma’s $11.75 billion acquisition of Organon propels the Indian drugmaker into the global biosimilars market, placing it among the top ten players. Organon’s biosimilar portfolio generated $691 million in 2025, about 11 % of its total revenue, with 45 % of that coming...

Drugging the Undruggable: Cancer's Slipperiest Targets Finally Meet Their Match
Researchers at the University of British Columbia and BC Cancer have unveiled a novel drug design strategy that tightly binds intrinsically disordered proteins, long deemed undruggable. The new compounds exhibit binding affinities up to a million times stronger than previous...
Bacteria-Resistant Coating on Catheters Reduces Infection and Need for Antibiotics
A clinical trial of Camstent's bacteria‑resistant polymer‑coated catheter showed a one‑third drop in catheter‑associated urinary tract infections (CAUTIs) and more than a 50 % reduction in antibiotic use versus standard catheters. Long‑term patients using the coated device reported zero symptomatic CAUTIs...

Nautilus Biotechnology Inc (NAUT) Q1 2026 Earnings Call Transcript
Nautilus Biotechnology reported a 23% drop in Q1 operating expenses to $15.4 million and ended the quarter with $156.1 million in cash, extending its runway into 2027. Management guided to modest $0.5 million revenue in 2026, primarily from grant‑funded early access customers, while...

CDER Proposes to Withdraw Approval of TAVNEOS
The FDA’s Center for Drug Evaluation and Research has proposed withdrawing approval of TAVNEOS (avacopan) after uncovering data manipulation in the pivotal trial and a lack of demonstrated efficacy for severe ANCA‑associated vasculitis. The agency also flagged new safety signals,...

China Races to Build Record Biobank to Rival US Drugs Research
China is constructing a national biobank that will house blood and DNA samples from 33,000 children, targeting brain‑disease research. The initiative, led by the Chinese Institute for Brain Research, seeks to create a data‑rich platform that rivals the United States’...

The BioPharm Brief: FDA Advances Autoimmune Pipeline with New Delivery Options and Priority Review
The FDA cleared a subcutaneous autoinjector for anifrolumab, allowing at‑home treatment of moderate‑to‑severe systemic lupus erythematosus. It also granted Priority Review to nipocalimab for warm autoimmune hemolytic anemia, a rare blood disorder, accelerating its path to market. AbbVie submitted a...

April 30, 2026: Meeting of the Oncologic Drugs Advisory Committee Meeting Announcement - 04/30/2026
The FDA’s Oncologic Drugs Advisory Committee will meet on April 30, 2026 to evaluate two AstraZeneca submissions: NDA 220359 for camizestrant tablets combined with CDK4/6 inhibitors in ESR1‑mutated hormone‑receptor‑positive breast cancer, and sNDA 218197/S‑004 for Truqap (capivasertib) plus abiraterone in PTEN‑deficient metastatic hormone‑sensitive prostate...

Intellia’s Data Reveal Tees Up FDA Filing for CRISPR-Based In Vivo Gene-Editing Med
Intellia Therapeutics filed a rolling FDA submission for lonvo‑z, its CRISPR‑based in‑vivo gene‑editing therapy for hereditary angioedema (HAE). In a placebo‑controlled Phase 3 trial of 80 patients, a single infusion cut swelling attacks by 87% versus placebo and left 62% of...

FDA Grants Breakthrough Designation for Efimosfermin for MASH
GSK’s investigational liver drug efimosfermin received FDA breakthrough therapy designation for metabolic dysfunction‑associated steatohepatitis (MASH). A phase‑2 trial showed 45.2% of patients achieved at least one‑stage fibrosis improvement versus 20.6% on placebo, and 67.7% attained MASH resolution compared with 29.4%...
STAT+: Erasca Touts Strong, Though Preliminary, Results in Trial of Pancreatic and Lung Cancer Therapy
Erasca announced that its oral RAS‑targeting drug ERAS‑0015 produced tumor shrinkage in 40% of patients with advanced pancreatic cancer and 62% of those with advanced non‑small‑cell lung cancer. The early‑stage data, gathered from trials in the United States and China,...

A Treatment for Pre-Eclampsia May Be on the Horizon
Researchers have unveiled a blood‑filtering therapy that shows promise in treating pre‑eclampsia, a life‑threatening pregnancy complication. Early‑stage trials reported significant reductions in maternal blood pressure and improved organ function without harming the fetus. The approach works by extracorporeally removing circulating...

Pencil Beam Laser Could Help Researchers Design Brain-Targeted Therapies
MIT researchers have demonstrated that laser light can self‑organize into a tightly focused "pencil beam," enabling a new bioimaging modality that is both faster and high‑resolution. In proof‑of‑concept experiments the team captured three‑dimensional images of the human blood‑brain barrier 25...
Serif Biomedicines Raises $50 Million
Flagship Pioneering announced the creation of Serif Biomedicines, a new biotech focused on Modified DNA therapeutics. The company emerges after five years of platform development. Flagship pledged an initial $50 million to fund the launch. The venture positions Serif to pursue...

FDA May Be Poised To Collaborate With Paradigm On AI Model
The FDA is reportedly preparing an announcement on real‑world evidence (RWE) that could involve a partnership with Paradigm on its new SPIRAL artificial‑intelligence model. SPIRAL is designed to generate RWE for post‑approval and late‑stage clinical studies, leveraging large‑scale health‑data sources....

Autism Genetics Linked to Reduced Brain Cell Fiber Density
Researchers analyzed brain imaging and genetic data from over 30,000 UK Biobank adults and nearly 5,000 ABCD children, finding that higher polygenic scores for autism consistently correlate with lower neurite density across the cortex and major white‑matter tracts. The association...

Randomized Radical Reaction Leads to Selective Cyclizations
A new “radical sampling” strategy reported in JACS (2026) enables selective formation of six‑membered nitrogen heterocycles such as piperidines and morpholines from simple aldehyde and amine precursors. The method uses a light‑activated catalyst to generate radicals that compete between rapid...

Withdrawn | Cancer Accelerated Approvals
The FDA has withdrawn 31 cancer drugs that were granted accelerated approval, spanning indications from lung and breast cancer to rare lymphomas. The most recent removal was Trodelvy for metastatic urothelial carcinoma on November 22, 2024. Withdrawals typically follow failed...

Exercise, Ibuprofen Reduce Cancer-Related Cognitive Impairment
A phase‑2 randomized trial of 86 chemotherapy patients found that a six‑week home‑based exercise program markedly improved cognitive performance, cutting Trail Making Test times by over 21 seconds compared with placebo. Low‑dose ibuprofen (200 mg twice daily) also yielded modest gains,...
A Brain Implant for Depression Is About to Be Tested in Humans
Motif Neurotech received FDA approval to begin a first‑in‑human study of its blueberry‑sized brain implant designed to treat severe depression. The device sits just above the dura and delivers wireless electrical stimulation to the central executive network, aiming to restore...

Kerry Facility Expansion Helps Meet Demand for Lactose-Free Dairy
Kerry announced a major expansion of its Carrigaline, Ireland biotechnology plant, boosting industrial‑scale production of lactase enzymes. The upgraded facility now supports processing of over 2 million tonnes of milk each year, helping dairy producers meet soaring demand for lactose‑free and...

How Does Integrated Fluid Management Enable Flexible Bioproduction Across Modalities?
At INTERPHEX 2026, Avantor senior vice president Jerry Keybl highlighted how integrated fluid management underpins scalable bioproduction across cell, gene and other therapeutic modalities. He argued that consistent, well‑characterized fluid paths from early development through commercial scale reduce variability and...
Veradermics Soars on Positive Data for Baldness Treatment
Veradermics announced that its experimental oral minoxidil pill, VDPHL01, achieved the primary endpoint in a Phase 3 trial, delivering a 30‑33 hair‑per‑cm² increase over six months versus placebo. The drug was well tolerated, with side‑effect rates matching placebo and no cardiac...

Pam Marrone Targets Resistant Weeds with Cocktails of Microbial Metabolites as Bioherbicide Space Heats Up
Invasive Species Corporation (ISC), led by Pam Marrone, is developing bioherbicides that combine multiple microbial metabolites to tackle glyphosate‑resistant weeds. The company has advanced two lead candidates from greenhouse tests to extensive field trials across the Midwest and Southeast in...

Lilly to Acquire Ajax Therapeutics
Eli Lilly announced a definitive agreement to acquire Ajax Therapeutics for up to $2.3 billion in cash. Ajax’s lead candidate, AJ1-11095, is a first‑in‑class Type II JAK2 inhibitor currently in a Phase 1 study (AJX‑101) for myelofibrosis patients who have failed Type I JAK2...
Compass Shares Crash as ‘Confounding’ Survival Data Raise Approvability Questions for Bispecific
Compass Therapeutics reported that its bispecific antibody tovecimig failed to meet the secondary overall survival (OS) endpoint in the Phase 2/3 COMPANION‑002 trial, though it achieved a 56% reduction in progression risk, extending progression‑free survival (PFS) to 4.7 months versus 2.6...

University of Southampton and UCB to Develop Digital Antibodies with AI
The University of Southampton and biopharma firm UCB have launched the Digital Antibody Research Collaboration (DARC), a three‑year AI‑driven platform for in‑silico therapeutic antibody design. DARC aims to compress the typical ten‑to‑twelve‑year development timeline by automating molecule modeling, testing and...

This Dangerous Pregnancy Complication Is Common. A New Treatment Might Help
A novel blood‑filter that removes excess soluble Flt‑1 reduced the protein by roughly 17% in a pilot study of 16 women with early‑onset preeclampsia. The intervention modestly lowered blood pressure and proteinuria, allowing pregnancies to extend a median of 10...

ESCMID Global 2026: Pritelivir Excels in Immunocompromised Refractory HSV Patients
Aicuris presented Phase III data for its oral helicase‑primase inhibitor pritelivir at ESCMID Global 2026. In the PRIOH‑1 trial of 101 immunocompromised adults with acyclovir‑refractory HSV, pritelivir achieved significantly higher lesion‑healing rates than investigator‑chosen IV or topical therapies. The drug also...

Missouri Analytical Laboratories Inc - 615319 - 10/09/2024
The U.S. Food and Drug Administration issued a closeout letter to Missouri Analytical Laboratories Inc., confirming that the company’s corrective actions have addressed the violations cited in a 2021 warning letter. The FDA noted that while the immediate issues appear...

Lilly to Buy Startup Ajax in Bid for a Better JAK Drug
Eli Lilly announced it will acquire New York‑based biotech Ajax Therapeutics for up to $2.3 billion. The deal centers on Ajax’s experimental JAK inhibitor AJ1‑11095, which binds an inactive JAK2 conformation to overcome resistance seen with existing therapies for myelofibrosis and...

FDA Grand Rounds – Anti-Biofilm Technologies for Enhancing the Safety of Medical Device Surfaces - 05/29/2025
On May 29, 2025, the FDA hosted a Grand Rounds webcast on anti‑biofilm technologies for medical device surfaces, presented by Dr. Jayaleka J. Amarasinghe, a microbiologist at the agency’s Winchester Engineering and Analytical Center. The session highlighted two emerging strategies...

Bacterial Defense System Builds DNA in Unexpected New Way to Stop Viruses
Scientists at Stanford have identified a bacterial antiphage system called DRT3 that synthesizes double‑stranded DNA with a precise GT/AC repeat pattern. The system relies on two reverse transcriptases: Drt3a copies an RNA template, while Drt3b builds its complementary strand using...

Fathom Therapeutics Scores $47 Million Series A
Fathom Therapeutics, previously known as Atommap Corp., announced an oversubscribed $47 million Series A round. The financing was led by Sutter Hill Ventures with participation from Chemistry, Alexandria Venture Investments, and NY Ventures. Fathom leverages quantum chemistry and artificial intelligence to design...

A Single Dose of Psilocybin Outperforms Nicotine Patches for Quitting Smoking
A Johns Hopkins pilot trial found that a single, weight‑adjusted dose of psilocybin combined with cognitive‑behavioral counseling helped 40% of smokers remain abstinent for six months, far surpassing the 10% quit rate achieved with standard nicotine patches. The psychedelic group...
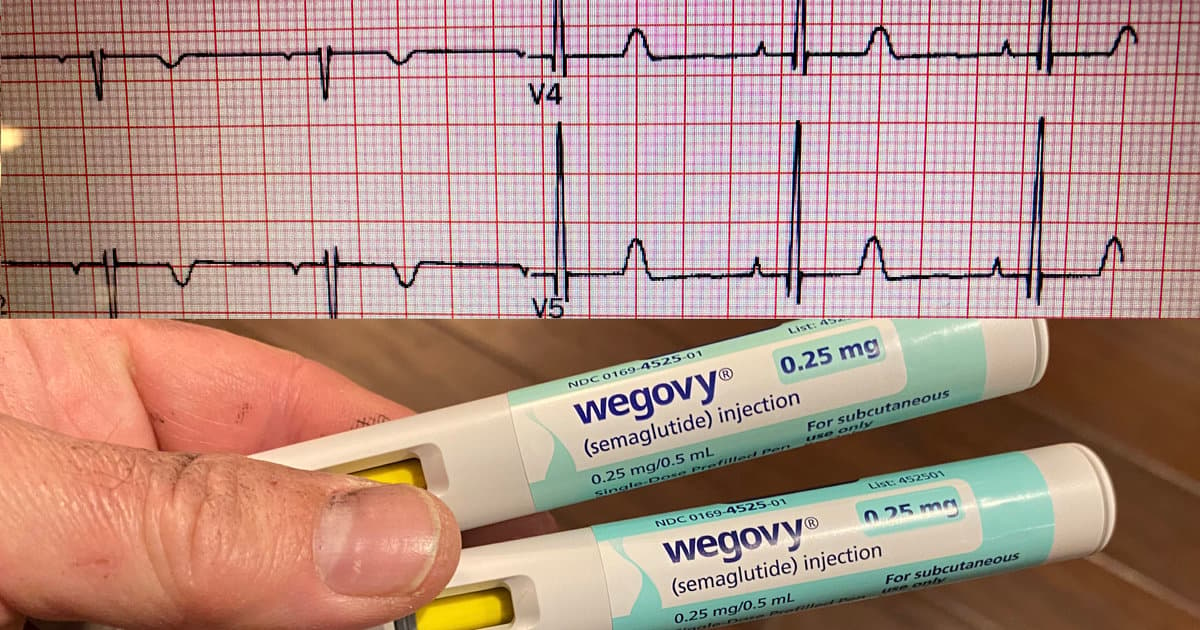
Multiple GLP-1 Drugs Linked to Lower AFib Risk
A retrospective analysis of 13,034 patients who started GLP‑1 receptor agonist therapy between 2020 and 2024 found a significant reduction in atrial fibrillation (AFib) incidence compared with a propensity‑matched cohort of over 385,000 untreated individuals. The benefit persisted regardless of...

Cellular Rejuvenation Has the Potential to Reverse Aging
Researchers have identified a natural cellular rejuvenation process that resets embryonic cells to a youthful state within two weeks, effectively erasing parental age markers. Over the past two decades, labs have revived skin cells from 90‑year‑olds and rejuvenated diseased mice,...

FDA Clears Subcutaneous Anifrolumab Autoinjector for Moderate to Severe SLE
The FDA has cleared AstraZeneca’s subcutaneous anifrolumab autoinjector (Saphnelo) for adults with moderate to severe systemic lupus erythematosus (SLE) on standard therapy. The new formulation mirrors the approved intravenous product but allows weekly self‑administration at home. Approval rests on the...
FDA Priority Review Advances Nipocalimab for Adults With Warm Autoimmune Hemolytic Anemia
The FDA has granted priority review to Johnson & Johnson’s nipocalimab‑aahu (IMAAVY) for adults with warm autoimmune hemolytic anemia (wAIHA), a rare disease lacking any approved U.S. therapy. The designation compresses the review clock to roughly six months, marking the...

STAT+: Astellas Retries XLMTM Gene Therapy After Deaths
Astellas Pharma announced it will restart its next‑generation gene therapy trial for X‑linked myotubular myopathy (XLMTM) after pausing the program following two patient deaths. Meanwhile, Intellia Therapeutics reported its one‑time CRISPR treatment lonvo‑z reduced hereditary angioedema attacks by 87% in...

STAT+: Pharmalittle: We’re Reading About a Lilly Deal, an Intellia CRISPR Rare-Disease Treatment, and More
Intellia Therapeutics reported that a single dose of its CRISPR‑based therapy lonvo‑z dramatically reduced swelling attacks in hereditary angioedema patients during a Phase 3 trial, positioning it for a rolling FDA submission as the potential second approved CRISPR drug. The treatment...
FDA Review Sought for Subcutaneous Risankizumab Induction in Crohn Disease
AbbVie has filed a supplemental NDA with the FDA to add subcutaneous (SC) risankizumab induction for adults with moderate‑to‑severe Crohn disease. The request relies on the phase 3 AFFIRM trial, which enrolled 289 patients—65% of whom had failed prior advanced therapies—and...

FDA Clears Tandem Control-IQ+ for Type 1 Diabetes During Pregnancy: First AID System to Earn Indication
The FDA has cleared Tandem Diabetes Care’s Control‑IQ+ system for use in pregnancies complicated by type 1 diabetes, making it the first automated insulin‑delivery (AID) platform with a pregnancy indication in the United States. The clearance covers both the t:slim X2 pump...

Gilead’s Pipeline Strategy in 2026: A Broadening Focus Beyond HIV
Gilead Sciences is accelerating a diversification push in 2026, adding $12.6 billion of oncology and immunology assets to a portfolio still dominated by HIV drugs. The company completed three major acquisitions—Arcellx for $7.8 billion, Tubulis for $3.15 billion, and Ouro Medicines for $1.67 billion—bolstering...
CureVac Takes Moderna to Court over mRNA Vaccine Patents
CureVac has filed a lawsuit against Moderna alleging infringement of its mRNA‑stabilisation and delivery patents that underpin Spikevax. The case, backed by BioNTech after its 2025 acquisition of CureVac, seeks a share of the multibillion‑dollar revenues from COVID‑19 mRNA vaccines....

FDA’s Need for Speed Could Strain Small Biotechs. Here’s How They Can Keep Up.
The FDA is accelerating drug approvals through the National Priority Voucher pilot, which can shrink review timelines from ten months to as little as two, and a new plausible mechanism pathway for niche therapies lacking large trial data. While the...