
The Stanford Professor Behind an FDA-Cleared Cardiac AI Wants $1 Billion for His Next Company
Stanford associate professor James Zou is reportedly raising about $100 million at a $1 billion target valuation for his new startup Human Intelligence, which will apply AI across the entire biomedical discovery pipeline. Zou’s portfolio includes the FDA‑cleared cardiac‑AI EchoNet, a Nature‑published Virtual Lab that designed novel nanobodies, and a Virtual Biotech framework that annotated 56,000 clinical trials. The raise comes amid a surge of AI‑in‑healthcare funding—$11 billion in Q1 2026 alone—and follows other Stanford spin‑outs that quickly reached unicorn status. Investors are betting on Zou’s breadth of AI‑biology research rather than existing revenue.

GLP-1 Drugs Linked to Cognitive Impairment, Though the Reason Why Probably Isn’t What You Expect
A retrospective analysis of nearly 65,000 type‑2 diabetes patients over ten years found that users of GLP‑1 agonists such as semaglutide experienced cognitive impairment at twice the rate of non‑users (2.6% vs 1.3%). Researchers attribute the higher incidence to a...

Oncolytics Could Re-Rate As Pelareorep Focuses On Registrational Paths
Oncolytics Biotech is refocusing its reovirus therapy Pelareorep on anal cancer and metastatic colorectal cancer, sharpening its commercial narrative. A colorectal cancer trial reported a striking 33% objective response rate, far exceeding the 6‑11% historical standard, prompting the FDA to...

A School-Based Vaccination Campaign with Trivalent Live Attenuated Intranasal Vaccine (tLAIV) During the 2024–2025 Influenza Season in Two Schools of...
A school‑based campaign offered the trivalent live attenuated intranasal influenza vaccine (tLAIV) to children aged 3‑11 in two Palermo schools during the 2024‑25 season. Of the 2,140 eligible students, 415 were vaccinated, yielding a 19.4 % adherence rate that varied by...
Clinical Trial Finds No Difference in Fluid Treatment Options for Pediatric Sepsis
A NIH‑backed trial involving more than 9,000 children across five countries compared balanced crystalloid fluid with standard 0.9% saline for pediatric septic shock. The study found no significant differences in mortality, persistent kidney dysfunction, or need for renal‑replacement therapy between...

Inhaled Treprostinil Improves FVC in IPF Phase 3 Trial: Steven D. Nathan, MD
A phase 3 double‑blind trial (TETON‑2) evaluated inhaled treprostinil in 539 idiopathic pulmonary fibrosis patients and demonstrated a statistically significant preservation of forced vital capacity versus placebo. At 52 weeks, the treprostinil arm showed a median FVC decline of –49.9 mL compared with...

Efficacy of a Combination of Fipronil and Permethrin in Reducing the Risk of Dirofilaria Immitis Transmission to Dogs in Field...
A field trial in northern Greece evaluated Frontline Tri‑Act®, a topical fipronil‑permethrin blend, for heartworm prevention in dogs. Twenty‑four heartworm‑naïve Beagles were split 1:1 between monthly IVP treatment and an untreated control over seven months of peak mosquito activity. At study...
![[Comment] Antibody-Based Malaria Prevention in an Intense Perennial Transmission Setting](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/a3263fcd8d733301ef10712d19f5c34a.webp)
[Comment] Antibody-Based Malaria Prevention in an Intense Perennial Transmission Setting
A recent phase‑2 trial of the monoclonal antibody L9LS in Kenyan children demonstrated high efficacy against Plasmodium falciparum in an intense, year‑round transmission setting. The study reported roughly 70% protection after a single dose, with a safety profile comparable to...

Inventiva Reloads C-Suite as It Looks to Readout
Inventiva S.A. announced three senior appointments as it prepares for a Phase III readout of lanifibranor in metabolic dysfunction‑associated steatohepatitis (MASH) in Q4 2026. Axel‑Sven Malkomes, former CureVac CFO, will become CFO, with incumbent Jean Volatier moving to EVP finance & corporate...
High-Resolution Imaging Shines Light on Nanoscale Nuclear Organization
Researchers at the Indian Institute of Science have upgraded DNA‑PAINT microscopy to tag up to 12 nuclear biomolecules at once, delivering 3–5 nm resolution. The revamped method visualizes nine targets in under four hours, a dramatic speed‑up from the hours‑per‑target pace...

Patent Analysis Is Increasingly Shaping AI-Driven Target and Drug Candidate Selection
A new review in Nature Reviews Drug Discovery outlines how AI is expanding target identification by merging multi‑omics, knowledge graphs, and foundation models. The authors argue that patentability, commercial tractability, and competitor analysis should be evaluated alongside druggability and safety...

Utilizing Wearable Technology to Characterize and Predict Post-Exertional Malaise Crashes Across Post-COVID Syndrome and Chronic Inflammatory Conditions: Study Protocol of...
A prospective observational study called U‑WaTCH will enroll 300 adults—100 with post‑COVID syndrome, 100 with inflammatory rheumatic diseases, and 100 healthy volunteers—to wear Apple Watch SE or compatible devices for up to 180 days. Continuous streams of heart‑rate variability, activity, sleep, environmental...

Bioprocessing Method Is a Critical Factor for IgM Oligomerization
Researchers compared fed‑batch and semi‑continuous perfusion bioprocesses for recombinant IgM production in CHO DG44 and CHO K1 cell lines, generating either pentameric (5IgM) or hexameric (6IgM) antibodies with or without a J‑chain. The perfusion approach, which continuously supplies fresh medium...
White House Announces MFN Agreement with Drug Manufacturer Regeneron
The White House unveiled a most‑favored‑nation (MFN) pricing agreement with Regeneron covering its blockbuster eye‑disease drug Eylea. The pact obligates Regeneron to extend to Medicare the lowest net price it offers any private payer, with annual price reviews built into...

FDA Fast-Tracks the Development of Psychedelic Medications Following President’s Executive Order
The FDA announced an accelerated regulatory pathway for psychedelic‑based therapies after the April 18 executive order targeting serious mental illness. It will prioritize serotonin‑2A agonists for treatment‑resistant depression, PTSD and substance‑use disorders, and issue national‑priority vouchers for psilocybin and methylone studies...

FDA Grants Three Priority Vouchers to Psychedelics
The FDA has granted three Commissioner's National Priority Vouchers to psychedelic‑focused firms—Compass Pathways, Transcend Therapeutics and the Usona Institute—following President Trump’s executive order to accelerate mental‑health treatments. All three companies already hold Breakthrough Therapy Designations, and the vouchers promise faster...

Tortugas Neuroscience Nabs $106M
Tortugas Neuroscience announced a $106 million seed and Series A financing round, one of the larger early‑stage biotech raises this year. The capital, led by Cure Ventures and co‑led by The Column Group and AN Venture Partners, will fund the completion of...

AI Learns to Predict Breast Cancer Risk From How Single Cells Respond to Pressure
Researchers at City of Hope and UC Berkeley unveiled a microfluidic platform, mechano‑NPS, that squeezes individual breast epithelial cells to gauge their mechanical response. By training a machine‑learning classifier called MechanoAge, they derived a "mechanical age" metric that correlates with...

The BioPharm Brief: Innovation, Gene Therapy Momentum, and Next-Gen ADCs Lead Today’s Headlines
The latest Ron Lanton Report shows biopharma innovation moving beyond Boston and San Francisco into emerging regions powered by academic collaborations, venture capital, and niche talent pools. The European Medicines Agency issued a positive opinion on Novartis’ intrathecal onasemnogene abeparvovec, expanding...

The Slow March of Clinical Biomarkers to Become Surrogate Endpoints
Clinicians are urging the FDA to recognize more clinical biomarkers as surrogate endpoints, hoping to accelerate drug approvals and reduce trial durations. Regulators remain cautious, approving only six biomarkers through the 2007 Biomarker Qualification Programme and often demanding definitive clinical...
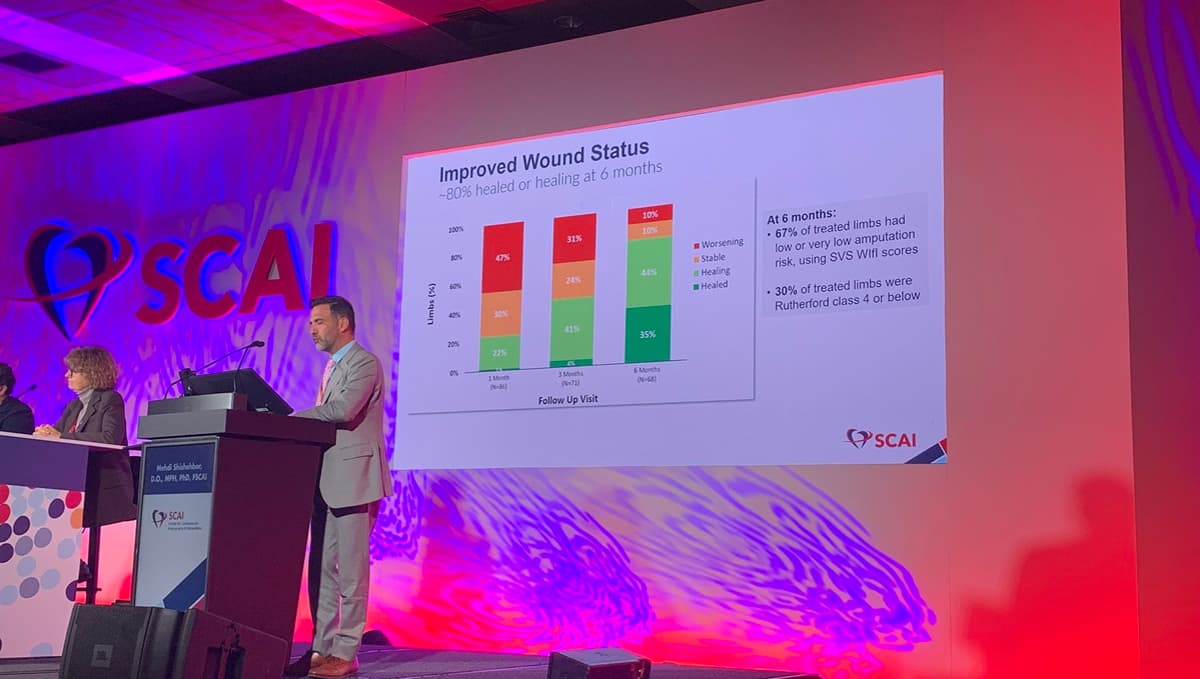
No-Option CLTI Patients Report Better QoL After Blood Flow Diversion Therapy: PROMISE III
The PROMISE III trial shows that transcatheter arterialization of the deep veins (TADV) using the LimFlow device markedly improves quality of life for no‑option chronic limb‑threatening ischemia (CLTI) patients. At six months, 80% of participants had healed or healing foot wounds...
Alphamab Oncology to Present Phase 1 Data on Bispecific ADC JSKN016 in HER2-Negative Breast Cancer at ASCO 2026
Alphamab Oncology will unveil Phase 1 results for its bispecific antibody‑drug conjugate JSKN016 at ASCO 2026, focusing on HER2‑negative breast cancer. The first‑in‑human trial in China demonstrated antitumor activity and a manageable safety profile in heavily pretreated patients, including triple‑negative cases. JSKN016...

One Biosciences Chooses Albany, NY, as Its U.S. Location
Paris‑based One Biosciences, backed by Institut Curie, announced its first U.S. hub in Albany, New York, where it will build a high‑complexity laboratory and computational analytics operation. The state’s Empire State Development agency will provide up to $525,000 in performance‑based...
Semaglutide NAION Risk Higher than Risk with SGLT2 Inhibitors
A VA‑based study published in JAMA Ophthalmology found that patients with type 2 diabetes taking semaglutide experienced more than double the risk of nonarteritic anterior ischemic optic neuropathy (NAION) compared with those on SGLT2 inhibitors. The analysis covered 102,361 veterans, with...

Policy Watch: FDA Moves to Implement Trump Order on Psychedelic Drugs
President Trump’s recent executive order tasked the FDA and ARPA‑H with accelerating psychedelic research. In response, the FDA issued Commissioner’s National Priority Vouchers to three firms—Compass Pathways, Transcend Therapeutics and the Usona Institute—fast‑tracking review of psilocybin and methylone candidates for...

STAT+: FDA to Speed up Review of Three Psychedelics as Mental Health Treatments
The FDA announced it will grant priority‑review vouchers to accelerate the evaluation of three psychedelic therapies—Compass Pathways’ psilocybin for treatment‑resistant depression, Usona Institute’s psilocybin for major depressive disorder, and Transcend Therapeutics’ MDMA‑like compound for PTSD. The move is part of...

Canadian Life Sciences Is at a “Generational Moment,” But Experts Disagree on Its Future
Canadian life sciences, contributing roughly 2% of GDP, is at a pivotal juncture as federal and provincial governments roll out new support mechanisms. BIOTECanada highlighted a federal task force, a Life Sciences Fund and BDC Capital’s $150 million CAD (~$110 million USD)...

Genomic Tool Untangles How Microbes Spread—Even when They Look Almost Identical
Researchers unveiled TRACS, a new genomic algorithm that pinpoints how microbes spread by detecting minute genetic differences. Published in Nature Microbiology, the tool successfully mapped transmission of SARS‑CoV‑2, Streptococcus pneumoniae, and Plasmodium falciparum across diverse cohorts. By distinguishing recent direct...

Protecting Innovation for Psychedelic Therapies Fast-Tracked Under New Executive Order
President Donald J. Trump signed an executive order that fast‑tracks FDA approval for breakthrough psychedelic therapies targeting serious mental illness. The order creates a Commissioner’s National Priority Voucher program, establishes pre‑approval patient access pathways, and earmarks $50 million in federal funding...

EU Biotech Act: The European Commission’s Landmark Proposal to Strengthen Biotechnology in Europe
On December 2025 the European Commission unveiled the European Biotech Act, a sweeping proposal aimed at cementing the EU’s position as a global biotech leader against the United States and China. The Act introduces fast‑track regulatory pathways, a strategic‑project framework...
Re: England Athletics Promotion of Medical Knee Procedure to Runners on Eve of London Marathon Is Condemned by Experts
England Athletics recently promoted Arthrosamid, a Class IIb implantable knee device, to runners ahead of the London Marathon. The product, marketed as a "drug," bypasses many safety tests because it carries a CE mark that allows manufacturer self‑certification. A UK...

FDA Awards Three Priority Review Vouchers For Psychedelics
On April 24, 2026 the FDA announced it will issue three priority review vouchers to companies developing psychedelic therapies. The vouchers cover two psilocybin programs targeting treatment‑resistant and major depressive disorder, and a methylone program for post‑traumatic stress disorder. The...

FDA Unveils Three Psychedelic-Focused Commissioner Vouchers
The U.S. Food and Drug Administration announced it has issued fast‑track “commissioner vouchers” to three psychedelic‑focused companies. The agency withheld the identities of the sponsors, a move that surprised investors and analysts. The vouchers are designed to expedite regulatory review,...

AI-Designed Drugs by a DeepMind Spinoff Are Headed to Human Trials
Isomorphic Labs, the DeepMind spinoff behind AlphaFold, announced that its AI‑designed drug candidates will soon enter human clinical trials. The company’s new IsoDDE engine claims to double the accuracy of AlphaFold 3 in predicting protein‑small‑molecule interactions. Partnerships with Eli Lilly and Novartis...

Learning, Predicting, and Interpreting Omics Data with Biologically Informed Models
Pablo Rodriguez‑Mier presented CORNETO, a unified optimization framework that fuses prior biological knowledge with high‑throughput omics data to infer context‑specific networks. The method was applied in the EU‑funded DECIDER project to pinpoint molecular mechanisms driving chemotherapy resistance in high‑grade serous...

Designer Baby Companies Are in Turmoil
Two high‑profile germline‑editing startups—Bootstrap Bio and Manhattan Genomics—have ceased operations within a year of launching. Bootstrap Bio folded after running out of capital and was further tarnished by the federal arrest of its chief science officer on child‑sex‑trafficking charges. Manhattan...

The Humility Of Bioscientists
Gene‑editing tools like CRISPR‑Cas9 are moving from rare‑disease therapies to agriculture and livestock, promising scalable health and climate benefits. Researchers such as Nobel laureate Jennifer Doudna and genome pioneer Craig Venter stress that the technology’s power outpaces our understanding of...

Postmarketing Requirements and Commitments: Reports
The FDA publishes annual reports in the Federal Register detailing the status of post‑marketing requirements (PMRs) and commitments (PMCs) for drugs and biologics, covering both CDER and CBER. These reports summarize data from internal databases and are updated quarterly on...

AAN 2026: Tavapadon Post-Hoc Analysis Strengthens Its D1/D5 Agonist Pitch
At the 2026 American Academy of Neurology meeting, AbbVie unveiled post‑hoc results from its Phase III TEMPO‑1 and TEMPO‑2 trials of tavapadon, a once‑daily oral D1/D5 partial agonist for early Parkinson’s disease. The analysis showed statistically significant improvements in seven of...
Reprogrammed Cardiomyocytes Soften the Blow in Heart Attack
A recent study published in the Journal of Molecular and Cellular Cardiology demonstrates that partial reprogramming of mouse cardiomyocytes with three Yamanaka factors (OCT4, SOX2, KLF4 – OSK) enables the cells to complete cytokinesis after a heart attack. By dismantling...
AI in Oncology Takes Off, Tackling HIV and Liver Disease, Pharma’s Recent Gains
At AACR 2026 in San Diego, AI-powered platforms took center stage, signaling a rapid shift toward data‑driven oncology. Parallel research revealed CRISPR screens that mapped CD4+ T‑cell genes that either promote or block HIV infection, while synthetic‑biology engineers demonstrated implantable...

AAN 2026: Head-to-Head Trial Shows Superiority of Qulipta for Migraine Prevention
AbbVie presented Phase IIIb TEMPLE trial data at AAN 2026, showing its oral gepant Qulipta outperforms topiramate in migraine prevention. Over 24 weeks, Qulipta had a 12.1% discontinuation rate versus 29.6% for topiramate and achieved a 73.7% responder rate (>50% reduction in...

Sanofi MS Drug Rejected in US Gets an Endorsement in Europe
Sanofi’s experimental multiple‑sclerosis drug tolebrutinib, marketed as Cenrifki, received a positive recommendation from the European Medicines Agency for secondary progressive MS, after the U.S. FDA rejected it earlier this year. The EMA’s endorsement paves the way for a final approval...
Drug Digest: Examining the Architecture of Next-Gen Biotherapeutic Modalities
Minaris Advanced Therapies’ chief commercial and technology officer, Dr. Eytan Abraham, discussed how multifunctional biotherapeutics are merging antibodies, ADCs and engineered cells to create more precise, personalized treatments. He highlighted multi‑targeting cell designs that improve specificity and reduce disease escape,...

Biogen Seeks Darzalex Rivalry in China for Multiple Myeloma with Felzartamab Deal
Biogen has secured exclusive rights to TJ Biopharma’s anti‑CD38 antibody felzartamab in Greater China for up to $850 million, including control of a pending biologics licence application for relapsed and refractory multiple myeloma. The drug will be manufactured domestically at TJ...

Novartis Pulls Pluvicto Prostate Cancer Label Expansion Filing in the EU
Novartis has pulled its European Medicines Agency (EMA) application to extend the label of Pluvicto, its lutetium‑177 PSMA‑targeted radioligand therapy, into earlier‑stage prostate cancer. The drug already received approval for this indication in the United States and the United Kingdom,...

Exclusive: UC Berkeley Startup Bets on Jumping Genes for GLP-1 Gene Therapy
A UC Berkeley spin‑out is developing a gene‑therapy platform that inserts a GLP‑1‑producing cassette into patients' cells using a jumping‑gene (transposon) system. The approach seeks to turn the body into its own continuous source of the hormone, eliminating the need...
AAN 2026: Long Half-Life and Long-Acting Injectable, What Pharmacists Should Know About RAP-219's Dosing Advantages for Focal Epilepsy
Rapport Therapeutics presented phase 2 data for RAP‑219 at the AAN 2026 meeting, showing 24% of drug‑resistant focal epilepsy patients achieved complete seizure freedom over an eight‑week period. The trial leveraged responsive neurostimulation (RNS) devices to capture objective electrographic seizure data,...
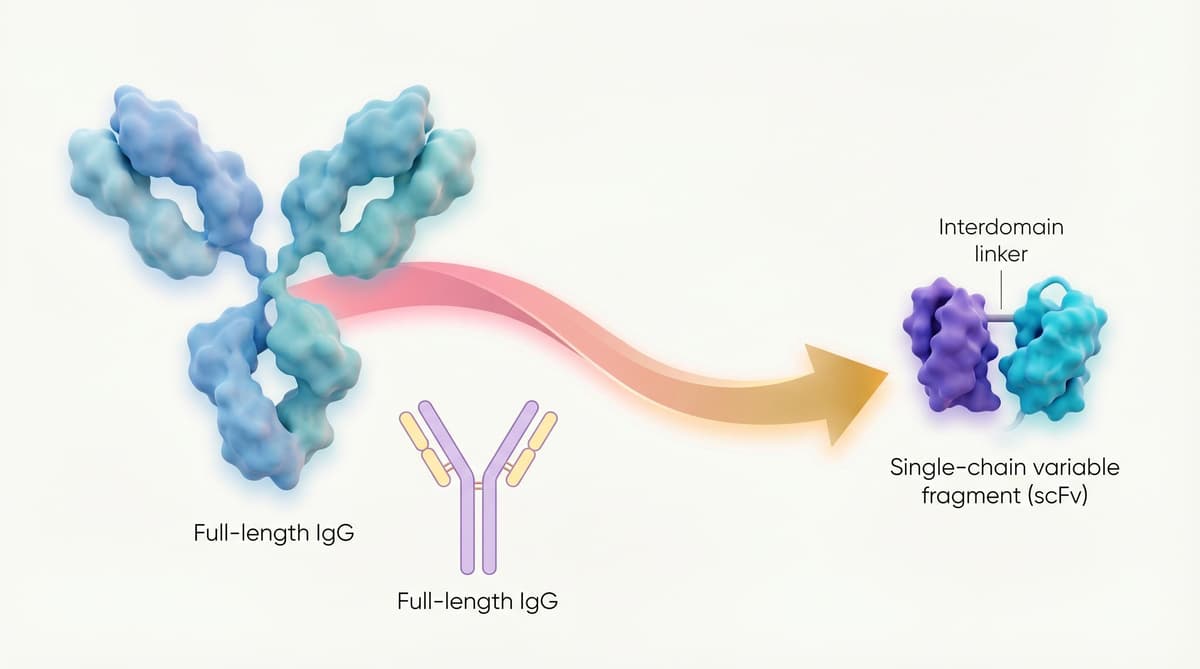
New Antibody Drugs Target Disease From Within
Researchers have used AI to redesign antibody binding fragments, creating more than 600 stable intracellular versions. By adjusting charge distribution, these fragments remain soluble inside cells and retain target specificity, enabling direct binding to disease‑driving proteins such as those implicated...

Regulatory Round-Up: Novartis, Sanofi and Arrowhead Secure CHMP Blessings
The European Committee for Medicinal Products (CHMP) issued positive opinions on three therapies: Sanofi's BTK inhibitor cenrifki for non‑relapsing secondary progressive multiple sclerosis, Arrowhead's siRNA drug redemplo for familial chylomicronemia syndrome, and Novartis' gene‑replacement therapy itvisma for spinal muscular atrophy....