
Precision Delivered: How Radiopharmaceuticals Are Reshaping Cancer Care
Radiopharmaceuticals, especially alpha‑emitting agents, are emerging as precision tools that deliver high‑LET radiation directly to cancer cells while sparing healthy tissue. Building on decades of beta‑emitter use, alpha therapies generate dense DNA damage and are less dependent on tumor oxygenation. Researchers are testing both systemic and intratumoral delivery to tackle hard‑to‑treat solid tumors such as pancreatic cancer and recurrent glioblastoma. Manufacturers are now focusing on simplifying production and distribution to move these agents from trials into routine oncology practice.

The Road Ahead—What’s Next for Host Cell Protein Analytics?
Advanced LC‑MS strategies are reshaping host cell protein (HCP) analytics, moving beyond the semi‑quantitative limits of traditional ELISA. Data‑independent acquisition (DIA) offers broad relative profiling, while stable‑isotope‑labeled (SIL) peptides provide absolute quantification for high‑risk HCPs. Integrating DIA, SIL, and ELISA...
Bispecific Antibodies Are Reshaping Multiple Myeloma Care: Prerna Mewawalla, MD
Bispecific antibodies are rapidly reshaping multiple myeloma treatment by delivering response rates up to 80% in earlier relapse settings, far surpassing the historic 30% seen with conventional therapies. These agents simultaneously bind a myeloma antigen such as BCMA, GPRC5D or...

Pfizer Presents Auristatin S ADC for GPNMB Tumors
Pfizer has unveiled PF-08046033, an antibody‑drug conjugate that links the potent cytotoxic agent auristatin S to an antibody targeting the transmembrane glycoprotein NMB (GPNMB). The ADC is designed to deliver the payload directly into GPNMB‑expressing tumors, potentially widening the therapeutic...

Pharma Industry Faces Long Haul to Get Return on Investment From AI
The pharmaceutical sector is confronting a prolonged timeline before artificial intelligence delivers a clear return on investment. While AI tools have accelerated early‑stage drug discovery, most companies still grapple with data integration, regulatory compliance, and the high cost of talent....

Modified CRISPR Tool Targets Down Syndrome Mutation
Researchers at Beth Israel Deaconess Medical Center and Harvard Medical School have engineered a modified CRISPR system that inserts the long non‑coding RNA XIST into one copy of chromosome 21, silencing the extra genetic material that causes Down syndrome. The new...

Pfizer Culls Early PD-L1 Asset After Series of Clinical Wins, Deals in Cancer
Pfizer terminated the Phase 1 study of its early‑stage PD‑L1 antibody‑drug conjugate PF‑08046037 after enrolling eight patients, citing strategic business reasons rather than safety or efficacy concerns. At the same time, the company announced a 47% reduction in disease‑progression risk for...

Arrowhead Pharmaceuticals Receives the CHMP’s Positive Opinion for Redemplo (Plozasiran) to Treat Familial Chylomicronemia Syndrome (FCS)
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) issued a positive opinion on Arrowhead Pharmaceuticals’ RNAi therapy Redemplo (plozasiran) as an adjunct to diet for adults with familial chylomicronemia syndrome (FCS). The recommendation follows the Phase III...
15 Companies You’ll See at NIBRT Careers in Biopharma 2026
On Saturday, April 25, the National Institute for Bioprocessing Research and Training (NIBRT) will host its 12th annual Careers in Biopharma event in Dublin’s O’Reilly Hall. The free‑registration fair will feature 15 leading biopharma companies—including AbbVie, Amgen, Eli Lilly, Pfizer and WuXi...

Japanese Pharma Companies Turning to CDMOs Earlier in Product Life Cycle
Japanese pharmaceutical firms are engaging contract development and manufacturing organizations (CDMOs) earlier in the product lifecycle, especially for peptide programs. Over the past 12‑24 months, demand for preclinical and early‑clinical peptide services has risen sharply, driven by venture‑backed biotech spinouts...

Lilly’s CAR-T Devotion; CBER’s Next Top Dog?
Eli Lilly announced the acquisition of Kelonia Therapeutics, an in‑vivo CAR‑T biotech, extending its push into next‑generation cell therapies. The deal follows Lilly’s recent milestone of becoming the first drugmaker to hit a $1 trillion market value, underscoring a diversification strategy that...

The Problem with Psychedelic Research
President Trump signed an executive order to speed the approval of psychedelic drugs for veterans with PTSD and depression, marking the first major regulatory push for these treatments. A new review of 24 studies compared psychedelics to open‑label antidepressants and...
Is Pharma Missing the Boat on Diagnostics?
Pharma giants are accelerating investments in diagnostics to support precision‑medicine strategies, highlighted by Roche's $595 million purchase of SAGA Diagnostics and Abbott's $21 billion acquisition of Exact Sciences. Venture capital for diagnostics lagged behind AI, with only $1.7 billion raised last year versus...

KT-621 Gets FDA Fast Track Designation for Eosinophilic Asthma
Kymera Therapeutics received FDA fast‑track designation for its oral STAT6 degrader KT‑621, targeting moderate‑to‑severe eosinophilic asthma and atopic dermatitis. The first‑in‑class molecule works intracellularly, offering a potential oral alternative to injectable biologics that block the IL‑4/IL‑13 pathway. Phase 2b trials are...
This Artificial Retina Doesn't Just Aim to Restore Sight—It Opens a Hidden Channel of Vision
Researchers at Yonsei University and the Institute for Basic Science have unveiled an implantable artificial retina that detects near‑infrared (NIR) light and converts it into electrical pulses to stimulate surviving retinal ganglion cells. The device combines a phototransistor array with...

Sanofi’s Tolebrutinib Gets CHMP Backing for Certain MS Patients Despite FDA Rejection
Sanofi’s BTK inhibitor tolebrutinib received a positive opinion from the European Medicines Agency’s CHMP for treating relapsing forms of multiple sclerosis, despite a complete response letter from the U.S. FDA in December. The CHMP recommendation paves the way for a...

CAR T-Cell Therapy May Prevent Progression of Smoldering Myeloma
A phase‑2 trial of ciltacabtagene autoleucel (cilta‑cel) in 20 high‑risk smoldering multiple myeloma patients achieved a 100% overall response rate, with all participants reaching minimal residual disease negativity within two months and no progression after a median 15.3‑month follow‑up. The...
New Scoring Tool Shows Radiation Can Reprogram Pancreatic Tumor Environment
Researchers at Fox Chase Cancer Center introduced the Harmonic Output of Stromal Traits Factor (HOST‑Factor), a composite scoring system that quantifies the functional state of the pancreatic tumor microenvironment. Using the tool, they showed that pulsed low‑dose‑rate (PLDR) chemoradiation reprograms...

BIO Is Expanding Its Work to Defend IP
The Biotechnology Innovation Organization (BIO) is scaling its intellectual‑property advocacy by creating an IP Task Force and a new Economic Growth, Innovation, and Intellectual Property Committee to steer strategy. BIO is actively lobbying on Capitol Hill, filing amicus briefs, and...
Cardiometabolic Trials: Using Expertise to Turn Complexity Into Robust Results
Nucleus Network’s Minneapolis site has emerged as a benchmark for early‑phase cardiometabolic trials, leveraging AI‑driven recruitment, rigorous PI oversight, and advanced imaging to deliver decision‑grade data. In the Rivus RIV‑HU6‑203 study the team screened 506 candidates, randomised 80 participants and...

Mabwell Initiates P-III Trial for 9MW2821 in TNBC
Mabwell has launched a Phase III trial of its Nectin‑4‑targeting antibody‑drug conjugate 9MW2821 in patients with locally advanced or metastatic triple‑negative breast cancer (TNBC). The study pits 9MW2821 against the investigator’s choice of chemotherapy in patients who have already received taxane‑based...
FDA Declines to Approve AbbVie's Botox Follow-Up
The FDA sent a complete response letter to AbbVie, rejecting its filing for TrenibotE, a rapid‑acting, shorter‑duration botulinum toxin intended as a follow‑up to Botox. The agency’s concerns focus on manufacturing data rather than safety or efficacy, and AbbVie says...
FDA Clears First Genetic Hearing Loss Gene Therapy
Regeneron became the first company to receive FDA clearance for a gene therapy targeting congenital hearing loss, approving its OTOF‑focused product Otarmeni. The therapy, an AAV‑delivered one‑shot infusion, will be offered free to U.S. patients, a rare move given typical...
CatalYm Doses First Patient in Phase II/III VINCIT Trial
CatalYm has begun dosing the first patient in its Phase II/III VINCIT trial, evaluating the anti‑GDF‑15 antibody visugromab for cancer‑associated cachexia. The double‑blind, placebo‑controlled study plans to enroll about 518 participants with advanced solid tumours such as NSCLC and colorectal cancer....

Nanoengineered Micellar Hydrogel with Controllable Strain‐Dependent Behavior for Brain Slice‐Like Tissue Patch Bioprinting
Researchers have created a nanoengineered chitosan micelle‑crosslinked hydrogel (CDP) that can be tuned to three distinct rheological states for extrusion‑based bioprinting. By balancing dynamic covalent crosslinks with shear‑induced micelle stacking, the CDP‑II formulation tolerates up to 200% strain while maintaining...

Is Stem Cell Therapy About to Transform Medicine and Reverse Ageing?
Stem cell therapy is re‑emerging as a credible route to tissue regeneration and age‑reversal after a decade of failed anti‑ageing bets. Researchers are now demonstrating partial cellular reprogramming that restores youthful function without erasing cell identity. Early‑stage human trials from...
Case Study: Data Sharing Through DECIPHER Supports Rare Disease Research and Clinical Care
The European Genomics Initiative’s DECIPHER platform now hosts data from the University of Bristol’s GenROC study, which has collected clinical and parent‑reported information on nearly 550 children with rare neurodevelopmental disorders. DECIPHER already contains genetic and phenotypic records for more...

Diagonal Therapeutics’ Innovative Clustering Antibodies for Vascular Diseases
Diagonal Therapeutics is advancing a pioneering platform of clustering antibodies designed to restore vascular receptor signaling, targeting the root cause of genetic vasculopathies. Its lead candidate, DIAG723, has earned FDA Orphan Drug Designation for hereditary hemorrhagic telangiectasia (HHT) and shows...

Johnson & Johnson Reports Clinical Findings on Imaavy (Nipocalimab) for Generalized Myasthenia Gravis (gMG) at AAN 2026
Johnson & Johnson presented Phase III Vivacity‑MG3 data on its anti‑IgG antibody, Imaavy (nipocalimab), in generalized myasthenia gravis (gMG). In the 24‑week post‑hoc analysis, patients receiving Imaavy plus standard of care were about four times more likely to achieve sustained...

Regeneron Reports the US FDA Accelerated Approval of Otarmeni (Lunsotogene Parvec-Cwha) in Genetic Hearing Loss
Regeneron’s Otarmeni (lunsotogene parvec‑cwha) received FDA accelerated approval for treating severe to profound sensorineural hearing loss caused by biallelic OTOF gene variants. The therapy, provided free of charge in the U.S., was evaluated in a Phase I/II CHORD trial of...

What Defines Equipment Readiness in Pharmaceutical Production
Equipment readiness is a critical pillar for pharmaceutical manufacturers, ensuring each batch meets strict quality and safety standards. Core elements include regular calibration, thorough sanitization, preventive maintenance, and meticulous documentation. Operator training and real‑time monitoring further safeguard compliance with regulatory...

India’s Praj Industries Opens AI-Enabled Precision Fermentation Lab, Signs Government MoU
Indian biotech firm Praj Industries has opened an AI‑enabled Advanced Precision Fermentation Lab at its Pune R&D centre, Praj Matrix. The facility focuses on large‑scale precision fermentation to boost efficiency, cut carbon emissions and reduce scale‑up risk for pharma, food,...

Opinion: Prasad’s FDA Exit Good for Rare Diseases but New CBER Head Must Repair Eroded Trust
Vinay Prasad’s exit from the FDA’s Center for Biologics Evaluation and Research (CBER) follows a Senate hearing that highlighted inconsistent approval pathways for rare‑disease therapies. Industry sponsors and patient advocates complained that the agency reversed previously negotiated trial designs, leaving patients...

'Sterility Failures' Prompt FDA to Threaten Radiopharmaceutical Producer with Disciplinary Action
The FDA issued a warning letter to the University of California San Francisco Radiopharmaceutical Facility after sterility testing uncovered Bacillus contamination in a PET‑imaging agent batch. The agency found the facility’s explanation—that the bacteria entered the test tube during analysis—insufficient...

Blood-Derived microRNA Signatures Associated with Hippocampal Structure and Atrophy Rate: Findings From the Rhineland Study
Researchers analyzed blood‑derived microRNA profiles from over 2,000 participants in the Rhineland Study to uncover associations with hippocampal structure and its longitudinal atrophy. Cross‑sectional analysis identified a set of miRNAs—including miR‑199a‑3p/199b‑3p, miR‑155‑5p, miR‑146a‑5p and miR‑505‑5p—linked to larger left hippocampal volume,...

New Bioreactor Turns Stem Cells Into an Immune-Cell Factory, Producing 40 Million Human Macrophages per Week
Researchers at Hannover Medical School have unveiled a medium‑scale bioreactor that converts induced pluripotent stem cells into human macrophages at commercial‑grade volumes. The system can harvest up to 40 million immune cells per bioreactor each week for up to ten weeks,...
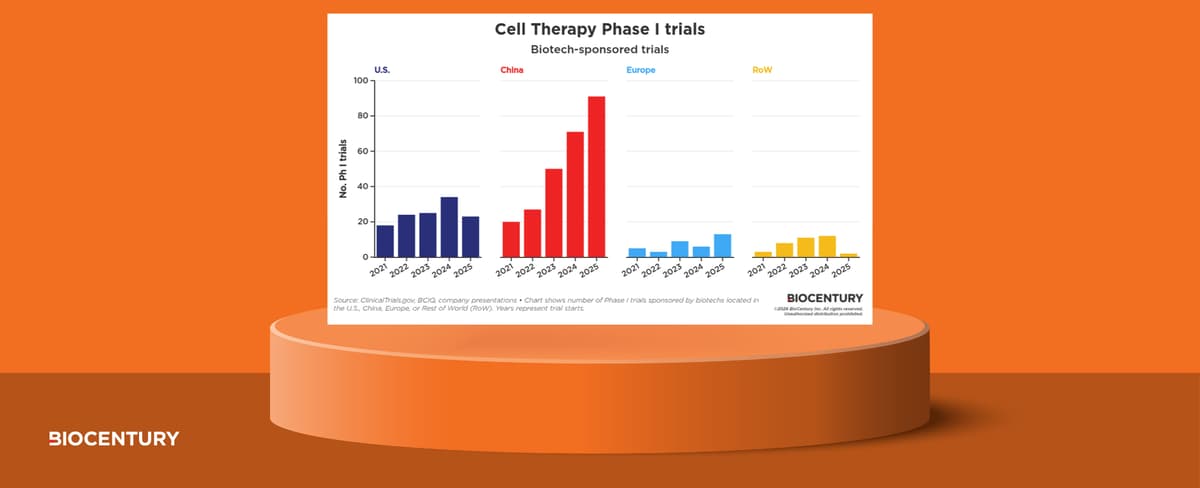
Cell Therapy Phase I Activity Accelerates on China Surge
BioCentury’s website now features a detailed Privacy Preference Center that categorizes cookies into strictly necessary, functional, marketing, advertising, and analytics groups. Users can toggle consent for each category, though disabling essential cookies may impair login and navigation. The policy clarifies...

These 'Good' Viruses Hold up a Booming Industry—AI Just Found a Faster Way to Track Them
Researchers at North Carolina State University combined electrochemical impedance spectroscopy with machine‑learning models to quantify viral vectors, eliminating the need for costly ELISA tagging. Six AI models accurately measured virus titers across five orders of magnitude, even with pH‑induced noise....
![[Comment] Alzheimer's Disease Immunotherapy and the Amyloid Hypothesis: When Aggregation Obscures Interpretation](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/614bb5987569a37ee2c14e1338f7df10.webp)
[Comment] Alzheimer's Disease Immunotherapy and the Amyloid Hypothesis: When Aggregation Obscures Interpretation
A Cochrane review released on April 16, 2026 pooled data from 17 randomized trials of amyloid‑beta‑targeting monoclonal antibodies, encompassing more than 20,000 participants with mild cognitive impairment or mild dementia due to Alzheimer’s disease. The analysis found little to no...
Designing Light-Controlled Chemistry with Custom Protein Pairs
Researchers at the University of California, Berkeley have engineered custom protein pairs that change conformation when illuminated with distinct wavelengths of light. By linking these light‑responsive proteins to catalytic domains, the team demonstrated precise, on‑demand activation of chemical reactions inside...

Sweet Protein: Pentasweet Breaks Ground on $76m Precision Fermentation Facility for Brazzein
Lithuanian biotech startup Pentasweet has broken ground on a €65 million ($76 million) precision‑fermentation facility in Vilnius that will produce commercial quantities of brazzein, a natural sweet protein up to 2,000 times sweeter than sugar. Phase I will establish core production capacity by early 2027,...
Increased Tumor Stiffness Accelerates Cancer Progression
Recent studies from Lund University demonstrate that increased stiffness of the tumor extracellular matrix directly drives cancer cell invasion through a β1‑integrin‑FAK‑Piezo1 mechanotransduction cascade. Using tunable 3D hydrogels, researchers showed that softening the matrix can reverse the invasive phenotype, but...

FLAG-Based Regimen Yields Robust Results in Acute Myeloid Leukemia Subtype
A Phase 2 trial at MD Anderson evaluated FLAG‑based chemotherapy combined with either gemtuzumab ozogamicin (FLAG‑GO) or idarubicin (FLAG‑IDA) in 219 newly diagnosed core‑binding factor AML patients. The FLAG‑GO arm delivered an 80% five‑year overall survival rate and a 67% relapse‑free survival...

ESCMID Global 2026: Zelicapavir Demonstrates Benefits for High-Risk Adults with RSV
At ESCMID Global 2026, Enanta Pharmaceuticals presented Phase IIb data for zelicapavir, an oral once‑daily N‑protein inhibitor, in high‑risk adults with RSV. The double‑blind study of 186 participants missed the primary endpoint but demonstrated faster symptom resolution—up to 7 days quicker in...

New Scoring Tool Reveals How Radiation Reprograms the Pancreatic Tumor Microenvironment
A novel scoring system developed by researchers quantifies how ionizing radiation reshapes the pancreatic tumor microenvironment. By integrating spatial transcriptomics, collagen imaging, and immune cell profiling, the tool identifies three distinct radiation‑induced phenotypes that correlate with patient survival and response...

Regeneron Will Offer Rare Disease Therapy Free As Part Of TrumpRx Deal
Regeneron announced it will provide its newly approved gene therapy for a rare pediatric hearing loss at no cost to U.S. patients under the TrumpRx program. The therapy, cleared by the FDA earlier this year, treats an X‑linked condition affecting...

Specialty Pharmacy's Critical Role as Advanced Therapies Move Into the Community: Fran Gregory, PharmD, MBA
Advanced therapies are set to expand dramatically, rising from 61 approved products today to roughly 180 by 2030. The growth includes cell and gene therapies, bispecific antibodies, and next‑generation monoclonal antibodies, many targeting high‑volume diseases such as cardiovascular and metabolic...
First Bispecific-ADC Combo Shows Strong Results in R/R Mantle Cell Lymphoma
A phase‑2 study published in Blood shows that combining the bispecific antibody mosunetuzumab with the antibody‑drug conjugate polatuzumab vedotin yields an 88.1% overall response rate in relapsed or refractory mantle‑cell lymphoma (MCL). The regimen produced a 78.6% complete response rate,...
AI Model Designs New Antibiotic for Staph Infections After Exploring 46 Billion Compounds
Researchers at McMaster University unveiled SyntheMol‑RL, a generative AI model that explored up to 46 billion virtual compounds and designed a novel, water‑soluble antibiotic named synthecin. The AI‑crafted drug demonstrated strong efficacy against drug‑resistant Staphylococcus aureus in mouse wound models when...

Pan-RAS Inhibitor ADCs Emerge at AACR 2026
At the 2026 American Association for Cancer Research meeting, four biotech companies unveiled antibody‑drug conjugates that deliver pan‑RAS inhibitors directly to tumor cells. Pan‑RAS inhibition has recently delivered unprecedented survival improvements in pancreatic and other RAS‑mutant cancers, but its clinical...