
Motif Neurotech Receives FDA IDE Approval to Initiate RESONATE Trial of Motif XCS System in Treatment-Resistant Depression
Motif Neurotech has secured FDA Investigational Device Exemption (IDE) to launch the RESONATE early feasibility study of its Motif XCS System in patients with treatment‑resistant depression who have failed at least two medications. The trial will monitor 12‑month safety, symptom reduction, quality of life, anxiety and cognitive function using standard clinical scales. The XCS device is a small, wirelessly powered implant placed over a validated depression target via a roughly 20‑minute outpatient procedure, allowing continuous brain‑signal monitoring to tailor therapy. Successful outcomes could position the system as a first‑in‑class therapeutic brain‑computer interface for psychiatric care.
GSK/Tesaro Claim Denied in Jemperli Lawsuit with AnaptysBio
A Delaware court rejected GSK and its Tesaro unit's attempt to terminate the 2014 licensing agreement with AnaptysBio, preserving the existing royalty rate for the PD‑1 cancer drug Jemperli. The ruling affirms that AnaptysBio has not breached the contract, while...
STAT+: Oruka’s Long-Acting Psoriasis Therapy Posts Strong Results in Mid-Stage Study
Oruka Therapeutics reported that its long‑acting injectable, ORKA‑001, achieved complete skin clearance in 63% of plaque‑psoriasis patients during a mid‑stage trial. The data suggest the drug can be administered just once per year, a stark contrast to current biologics that...

STAT+: Veradermics’ Hair Loss Drug Succeeds in Late-Stage Trial
Veradermics announced that its oral hair loss drug VDPHL01 met primary endpoints in a Phase III trial. Over six months, participants taking the pill grew 30‑33 hairs per square centimeter versus seven in the placebo group. Patient‑reported improvement reached 79‑86%, and...
Zepbound’s and Ozempic’s Greatest Benefit May Be Their Anti-Inflammatory Power
GLP‑1 drugs such as Ozempic and Zepbound are gaining recognition for anti‑inflammatory effects that go beyond weight loss and glucose control. Clinical data show semaglutide reduces C‑reactive protein by about 40% independent of weight loss and improves liver inflammation in...

AstraZeneca Reports FDA Approval of Saphnelo for Systemic Lupus Erythematosus (SLE)
The U.S. Food and Drug Administration has granted approval for AstraZeneca’s Saphnelo autoinjector (anifrolumab, 120 mg weekly) for adult patients with systemic lupus erythematosus (SLE) when used alongside standard of care. The decision follows the Phase III TULIP‑SC trial, which enrolled 367...

Novartis Secures Pair of Regulatory Wins for Skin Disease and Malaria Treatments
Novartis won two regulatory milestones: the European Commission approved its oral BTK inhibitor Rhapsido for adults with chronic spontaneous urticaria who have failed antihistamines, and the World Health Organization granted prequalification to Coartem Baby, an artemether‑lumefantrine formulation for infants weighing 2‑5 kg....
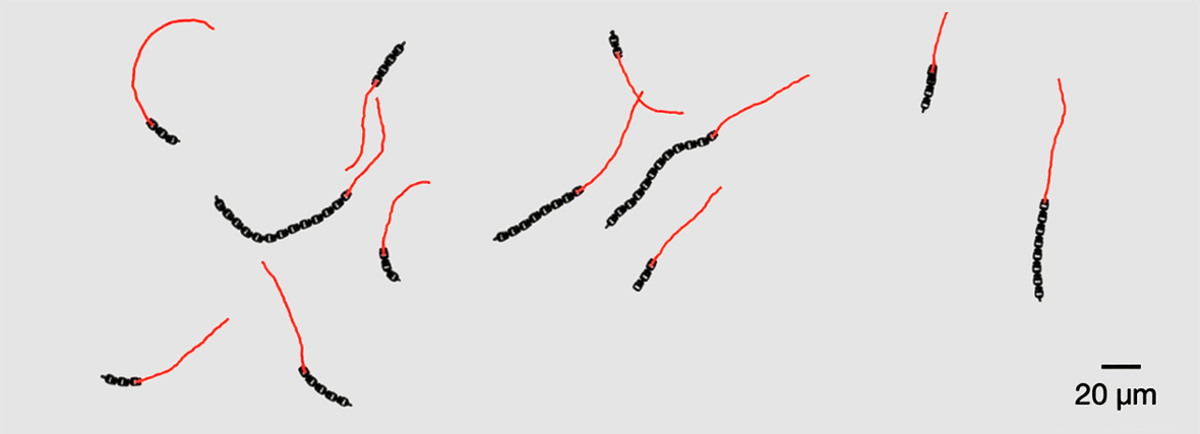
Leiden’s Sensor-Free Microrobots Move Like Living Organisms
Researchers at Leiden University have created soft, chain‑like microrobots that move and adapt without sensors, software, or external control. Each 5 µm segment is linked by 0.5 µm joints and powered solely by an electric field, allowing the robot’s shape to dictate...

New NIHR-Funded TRC for Parkinson’s Disease
The National Institute for Health and Care Research (NIHR) has launched the Parkinson’s disease Translational Research Collaboration (PD‑TRC), the first of eight UK TRCs dedicated to Parkinson’s. Backed by NIHR and four major charities, the hub links 17 centres of...

Lilly Falls on Slower Start for Foundayo versus Wegovy
Eli Lilly’s oral GLP‑1 agonist Foundayo launched in early April with modest uptake, recording 1,390 prescriptions in week 1 and 3,707 in week 2, far below Novo Nordisk’s oral Wegovy, which logged over 3,000 fills in its first days and 18,410 in the second...
UK Biotech Could Be Bound for Recovery, BIA Report Reveals
UK biotech venture financing rebounded in Q1 2026, with total equity raised climbing 18% to £552 m ($746 m). Venture‑capital inflows also rose 17% to £516 m ($699 m), and 25 companies secured funding, up from 15 a year earlier. While public‑market activity stayed flat—no...

BrioHealth Secures FDA Approval to Launch BrioVAD System Trial
BrioHealth Solutions received conditional FDA approval to launch the Brio4Kids trial, testing its BrioVAD left ventricular assist device in children with advanced heart failure. Enrollment in the U.S. study is slated for mid‑2026, with initial data expected in the fourth...

Kyowa Kirin and Kura Initiate Phase II Trial of Ziftomenib for AML
Kyowa Kirin and Kura Oncology have opened a Japanese Phase II registrational study of the oral menin inhibitor ziftomenib in adults with relapsed or refractory NPM1‑mutated acute myeloid leukaemia. The single‑arm, open‑label trial will measure a composite complete remission rate (CR + CRh) as...
Review Questions Benefits of Anti-Amyloid Alzheimer’s Drugs
A Cochrane review of 17 clinical trials involving 20,342 patients with early Alzheimer’s disease found that anti‑amyloid drugs provide no clinically meaningful benefit on cognitive decline or dementia severity. The analysis also highlighted an increased risk of brain swelling and...

Sanofi Reports the CHMP Positive Opinion for Cenrifki (Tolebrutinib) to Treat Non-Relapsing SPMS
Sanofi’s oral BTK inhibitor Cenrifki (tolebrutinib) received a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) for treating secondary progressive multiple sclerosis (SPMS) without relapses in the past two years. The recommendation is...

AI in Single-Cell Analysis: Solving the Interpretation Gap
Single‑cell omics drives drug discovery but interpreting cell‑state annotations remains a bottleneck. Nygen Analytics introduced CyteType, an AI‑augmented platform that adds a traceable interpretation layer to existing pipelines, converting raw clusters into biologically meaningful labels. By combining marker‑gene analysis, literature...

Invest in the Future of Cancer Diagnostics and Treatment
Cancer incidence is rising as populations age and obesity increases, prompting a shift toward proactive diagnostics. Researchers are advancing liquid‑biopsy technologies, especially multi‑omics MCED tests, to detect tumors earlier and with greater specificity. Simultaneously, AI‑enhanced imaging is slashing MRI scan...

Exclusive Human Milk Diet Benefits Very Low Birth Weight Infants
A phase III randomized controlled trial in Japan demonstrated that an exclusive human milk diet markedly improves growth velocity and reduces serious complications in very low birth weight (VLBW) infants compared with mixed feeding regimens. The study eliminated bovine‑based protein fortifiers,...

Novartis’ Itvisma Receives the CHMP Positive Opinion for Spinal Muscular Atrophy
Novartis’ gene‑replacement therapy Itvisma (onasemnogene abeparvovec) received a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) for treating patients aged two years and older with 5q spinal muscular atrophy (SMA). The recommendation is...

Evonik: €80m for Biopharma CDMO Capacity in Slovakia
Evonik Industries is allocating roughly €80 million (about $87 million) to expand its fermentation plant in Slovenská Ľupča, Slovakia. The investment adds downstream capacity for pharmaceutical active ingredients and creates around 50 new jobs. The site, already a biotech hub producing spider‑silk protein...

Turn Health Data Into Impact: Browns University's Online Master in Biostatistics & Health Data Science
Brown University has launched an online Master’s in Biostatistics with a concentration in Health Data Science, delivered through its School of Public Health and School of Professional Studies. The 20‑month, five‑semester program comprises nine courses and a capstone project that...

In the Land of the Unblind: Are Psychedelics Really Better than Antidepressants?
Recent meta‑analysis comparing psychedelic‑assisted therapy (PAT) with open‑label antidepressant trials finds no clinically important difference in depression outcomes. While early PAT studies suggested larger effects, the analysis shows that functional unblinding limits any advantage, and open‑label antidepressants marginally outperform blinded...

Secretome-Mediated Antimicrobial and Immunomodulatory Activity of Lactobacillus Johnsonii Against Multidrug-Resistant Enteroaggregative Escherichia Coli
Researchers evaluated Lactobacillus johnsonii as a probiotic against a multidrug‑resistant enteroaggregative E. coli (EAEC) strain. The bacterium showed strong gastrointestinal tolerance, high auto‑aggregation (80 % at 4 h), and secretome‑driven inhibition of EAEC growth and biofilm formation, surpassing gentamicin. It also reduced...
Astrocytic Connexin 43 Hemichannel Dysregulation Drives Prefrontal Circuit Dysfunction and Schizophrenia-Like Behaviors
Researchers found that connexin 43 (Cx43) protein is significantly elevated in the prefrontal cortex of individuals with schizophrenia and in mice treated with the NMDA‑antagonist MK801. The increase is selective for Cx43 and boosts astrocytic hemichannel opening without altering gap‑junction coupling,...

Probiotics Extend Life of Apparel and Footwear
The Lab, a biotech fashion‑care brand, launched the "Take Care of the Things You Love" campaign to promote probiotic‑based cleaning products that protect and extend the life of sneakers, hats, apparel, denim and accessories. The range uses beneficial bacteria to...

Eli Lilly and Company (LLY): Among the Best Stocks to Buy While the Market Is Down
Eli Lilly announced a strategic acquisition of Kelonia Therapeutics for up to $7 billion, with $3.25 billion paid upfront and up to $3.75 billion in milestones. Kelonia’s in‑body CAR‑T platform, led by Phase 1 candidate KLN‑1010, expands Lilly’s oncology pipeline beyond its dominant weight‑loss franchise....

StockWatch: Trump Order Lifts Psychedelic Drug Shares
President Donald Trump signed Executive Order 14401, directing the FDA to accelerate research and grant Commissioner’s National Priority Vouchers (CNPVs) to breakthrough psychedelic therapies. The order also mandates the HHS to allocate at least $50 million through ARPA‑H to partner with...
Novartis Antimalarial Clears Key WHO Review to Get the Combo Drug to Babies
Novartis’s Coartem Baby, the first antimalarial specifically designed for infants, has cleared the World Health Organization’s pre‑qualification review, allowing public‑sector and donor agencies to procure the drug in regions lacking robust regulatory systems. The formulation, a liquid‑dissolvable, cherry‑flavored version of artemether‑lumefantrine,...

Team Finds Surprising Food Source for Tumors
University of Rochester researchers discovered that cancer cells, particularly breast tumors, consume the antioxidant glutathione as a primary fuel source. Analysis of tumor fluid revealed abundant glutathione, and preclinical experiments showed that inhibiting its uptake slows tumor growth. The team...

Oruka Therapeutics to Host Conference Call to Report Week 16 Data for ORKA-001 From the Ongoing EVERLAST-A Trial on April...
Oruka Therapeutics (Nasdaq: ORKA) announced it will present Week 16 data from its EVERLAST‑A Phase 2a trial of ORKA‑001, an investigational antibody for moderate‑to‑severe plaque psoriasis. The results will be released on Monday, April 27, 2026, during an 8:00 a.m. ET conference call and webcast. ORKA‑001...
Popular Weight-Loss Drug May Cut Heart Attack Risk by 54%, New Studies Find
Recent studies show tirzepatide, the GLP‑1/GIP agonist behind Zepbound and Mounjaro, slashes major cardiovascular events by 54% in patients after angioplasty and cuts complications by roughly 30% after aortic valve replacement. The benefits persisted for at least a year, translating...
Discovery of a Novel Vulnerability in Aggressive Lymphoma Could Change Future Therapy
Researchers at the University of Cologne’s Center for Molecular Medicine have identified the protein cFLIP as a critical driver of resistance in diffuse large B‑cell lymphoma (DLBCL), especially the ABC subtype. By overexpressing cFLIP, lymphoma cells block both intrinsic and...
‘Science Fiction’: How Life-Saving Organs Are Being Kept Alive Outside the Body
Organ shortages have driven a shift from static cold storage to active preservation methods. Normothermic machine perfusion (NMP) keeps kidneys and livers metabolically active in a nutrient‑rich, oxygenated circuit, extending viable time outside the body. An Australian first double transplant...

Letter to the Editor: Standard Chemoradiotherapy with Concurrent and Adjuvant Camrelizumab in Patients with High Risk Nasopharyngeal Carcinoma: Multicentre, Randomised,...
A phase‑3 BMJ trial showed that adding the PD‑1 inhibitor camrelizumab to concurrent chemoradiotherapy and 17 cycles of adjuvant maintenance extended progression‑free survival in high‑risk nasopharyngeal carcinoma, but overall survival did not improve significantly (HR 0.59, P = 0.19) after a median 39.9‑month...
Fluorescent Probe Lights up Centrioles and Cilia in Living Cells Across Species
Scientists at EPFL have unveiled CenSpark, a fluorescent probe that selectively labels centrioles and cilia in living cells. The probe binds to the unique microtubule architecture of these organelles, enabling super‑resolution and live‑cell imaging across a spectrum of species, from...

Assessing the Roche Acquisition of SAGA Diagnostics and the Future of Molecular Residual Disease Monitoring
Roche has agreed to acquire Sweden‑based SAGA Diagnostics for up to $595 million, adding the ultra‑sensitive Pathlight™ MRD platform to its Foundation Medicine portfolio. Pathlight tracks tumor‑specific structural variants in circulating DNA, achieving sub‑one‑part‑per‑million detection limits and a 13.7‑month lead time...

New Cytometer Measures Cell Stiffness to Improve Disease Diagnosis
Researchers at Brown University and NIST introduced a mechanophenotyping cytometer that gauges cell stiffness via time‑of‑flight measurements in microfluidic channels. The device can analyze 60–100 cells per second, dramatically outpacing atomic force microscopy’s one‑cell‑per‑30‑seconds rate. By linking travel time to...

Scientists Transform Wool Into Bone Repair Material
Scientists at King’s College London have shown that keratin extracted from wool can act as a biodegradable scaffold for bone regeneration. In rat skull‑defect models, the wool‑based membranes guided new bone growth that was more organized and structurally similar to...

Natural Compound Obakulactone Shows Therapeutic Potential for Rheumatoid Arthritis
Researchers have identified obakulactone, a natural tetracyclic triterpenoid from Phellodendri cortex, as a promising therapeutic for rheumatoid arthritis (RA). In a CFA‑induced rat model, oral dosing (50‑200 mg·kg⁻¹·d⁻¹) over 21 days markedly reduced joint swelling, restored cartilage integrity, and modulated immune...

Rotavirus Cases in Children Are Rising, but a Highly Effective Vaccine Has Slashed Hospitalizations
Rotavirus infections in U.S. children are climbing earlier this season, with test positivity reaching nearly 8% in early 2026. Since the oral vaccine’s introduction in 2006, hospitalizations have fallen 80% and emergency‑room visits 57%, underscoring its effectiveness. However, vaccination coverage...

Platelet-to-HDL Ratio Linked to Eosinophils in Pediatric Asthma
A new study in Pediatric Research reveals a positive correlation between the platelet‑to‑HDL cholesterol ratio (PHR) and blood eosinophil counts in children with asthma. Analyzing a well‑characterized pediatric cohort, researchers found that higher PHR values align with elevated eosinophils, indicating...
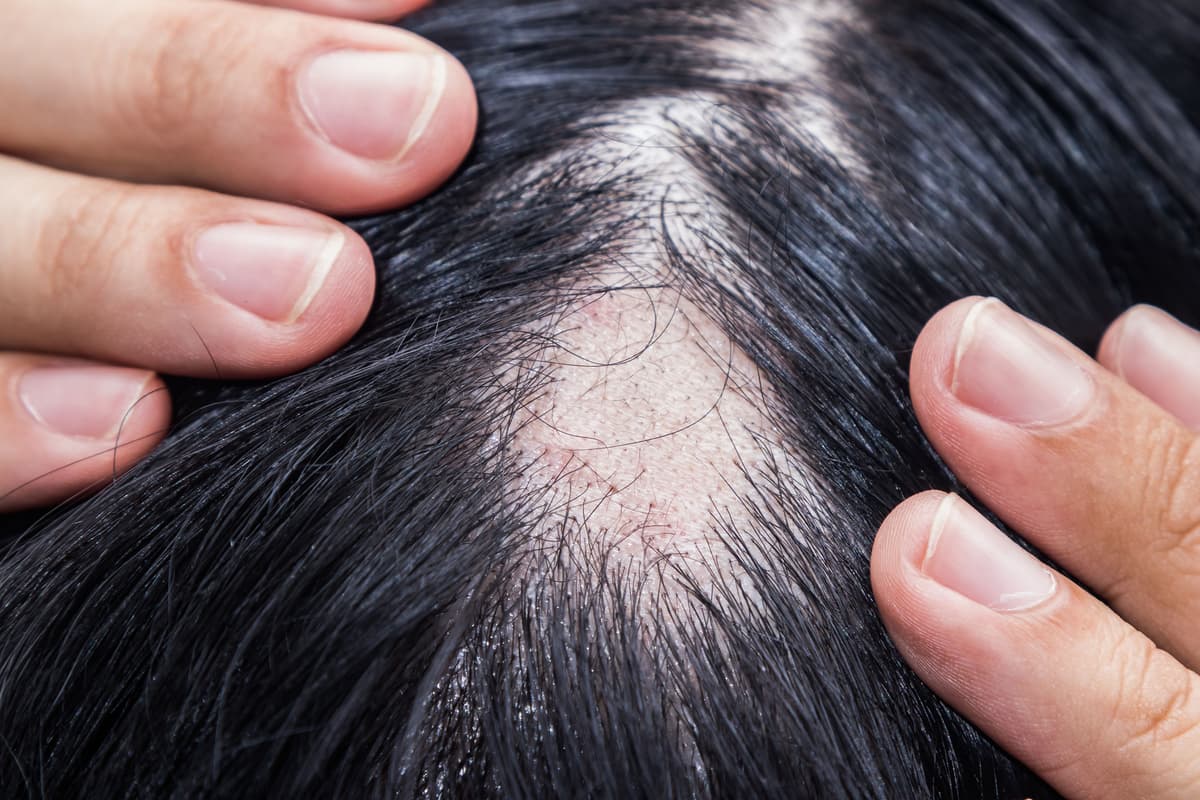
Deuruxolitinib Demonstrates Consistent Efficacy, Early Hair Regrowth in Severe Alopecia Areata
Deuruxolitinib (Leqselvi), an oral JAK1/JAK2 inhibitor from Sun Pharma, demonstrated robust efficacy in two pooled Phase 3 trials (THRIVE‑AA1 and THRIVE‑AA2) involving 867 adults with severe alopecia areata. At 24 weeks, 31% of treated patients achieved a SALT score of 20...
MASH Cirrhosis Trials Lack Consistent End Points
A new systematic review of phase 2 and 3 trials for metabolic dysfunction‑associated steatohepatitis (MASH) cirrhosis finds that endpoint selection is highly variable, with most studies relying on histologic improvement and few incorporating patient‑centered outcomes. The analysis identified only nine eligible...
CAR Therapies Could Offer New HBV, HIV Treatments
A new systematic review in Frontiers in Medicine evaluates 43 studies of virus‑directed CAR‑T and CAR‑NK therapies for chronic hepatitis B and HIV. Preclinical data show significant reductions in HIV p24 antigen, HBV surface antigen, and viral DNA, while early...

New Trial Prevents Cognitive Decline in Older Cancer Patients
A multicentric randomized controlled trial in India, called GOCog, tested a culturally tailored multidomain intervention to prevent chemotherapy‑induced cognitive decline in patients aged 60 and older. The program combined cognitive training, physical activity, nutrition guidance, and psycho‑educational support, and was...
KMT2C/D Loss Creates Targetable Cancer Weaknesses
Researchers have identified that loss of the histone‑modifying genes KMT2C or KMT2D creates exploitable weaknesses in cancer cells. Using a genome‑wide synthetic‑lethal screen, they uncovered a set of 12 drug candidates that selectively kill KMT2C/D‑deficient tumors. In mouse xenograft models,...

Four Independent Studies Validate The Blight Tolerance Of Darling American Chestnut Trees
SilvaBio and academic partners released data from four independent field studies confirming that American chestnut trees engineered with the OxO transgene develop 30%‑81% smaller blight cankers than wild‑type trees. Trials at Purdue, the University of New England and SUNY‑ESF showed...
PE/PPE Proteins Drive Tuberculosis Drug Resistance
Researchers have identified the PE/PPE protein families as key drivers of drug resistance in Mycobacterium tuberculosis. Genetic analyses reveal that specific PE/PPE variants up‑regulate efflux pumps and alter cell‑wall permeability, reducing the efficacy of first‑line antibiotics such as isoniazid and...

'Eventually, It Becomes You': Inventors of New 'Living' Knee Replacement Describe Why This Tech Is Desperately Needed and How It...
Columbia University and the University of Missouri are developing NOVAKnee, a 3D‑printed, biodegradable knee implant seeded with stem‑cell‑derived bone and cartilage. The scaffold is designed to dissolve as new tissue forms, potentially offering a longer‑lasting solution than metal‑plastic prostheses that...

Repeated Doses of Psilocybin Show Promise for Treating Obsessive-Compulsive Disorder
A randomized clinical trial found that weekly high‑dose psilocybin significantly reduced obsessive‑compulsive symptoms in treatment‑resistant patients. Fifteen adults received up to four doses over eight weeks, with 73 % achieving at least a 35 % drop in Yale‑Brown scores and 40 % attaining...