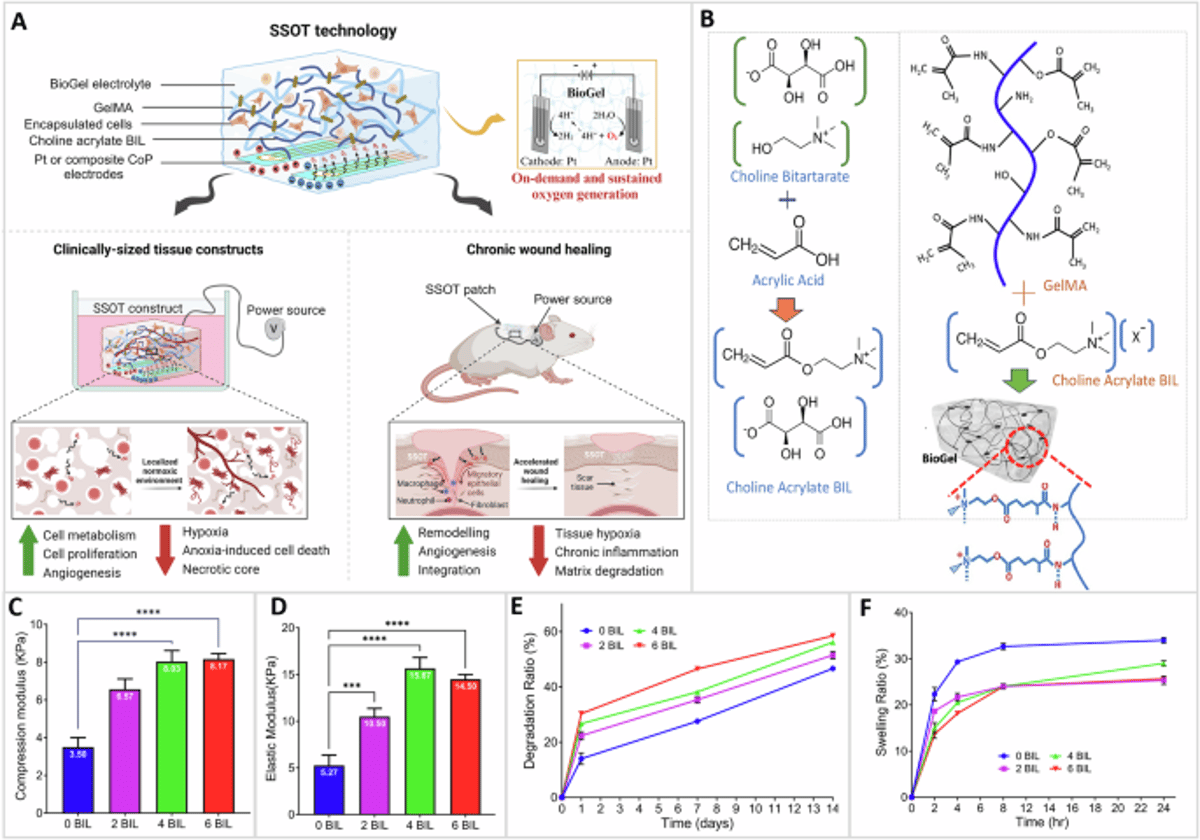
New Bioengineered Patch Makes Its Own Oxygen to Heal Wounds and Grow Tissue
Researchers at UC Riverside and Rowan University unveiled a self‑oxygenating tissue patch, the Smart Self‑Oxygenating Tissue (SSOT) system, that creates oxygen on‑demand via low‑voltage electrolysis in a conductive hydrogel called BioGel. The BioGel incorporates a choline‑based ionic liquid, boosting stiffness and slowing enzymatic degradation while serving as the electrolyte for the reaction. In vitro, the patch lifted cell survival under severe hypoxia to roughly 75%, and in diabetic mouse models it accelerated wound closure within 33 days with minimal immune reaction. The study demonstrates a viable route to sustain thick engineered tissues until vascularization occurs.

Replimune Cries Foul on Regulatory Flexibility. But Many Americans Want a Stricter FDA
The FDA rejected Replimune’s RP1 melanoma combination therapy twice, citing patient‑population heterogeneity that it says undermines efficacy interpretation. The biotech’s CEO decried the agency’s lack of regulatory flexibility, while a Politico poll revealed most Americans prefer a slower, more rigorous...

Piramal Pharma Solutions Partners with Ajinomoto Bio-Pharma Services to Advance ADC Development and Manufacturing
Piramal Pharma Solutions and Ajinomoto Bio‑Pharma Services have signed a strategic collaboration to accelerate antibody‑drug conjugate (ADC) development and manufacturing. Piramal will refer its customers to Ajinomoto’s AJICAP platform for site‑specific ADC conjugation, while Ajinomoto will direct clients to Piramal...

Kailera Raises $625M IPO in Biotech Record
Kailera Therapeutics completed a $625 million IPO, establishing a new biotech record and eclipsing Moderna’s 2018 $604 million debut. The offering, led by Goldman Sachs, JPMorgan and Morgan Stanley, attracted strong institutional demand and priced above expectations. Proceeds will fund Kailera’s gene‑editing...

Next Stage of Growth with a New CEO
Resyca BV, a Dutch specialist in soft‑mist inhalation drug delivery, has appointed Deborah Jones as its new chief executive. Jones brings over two decades of senior leadership experience, most recently overseeing business strategy for Proveris Scientific across EMEA and India....

Lilly’s Tirzepatide Sheds Lean Muscle Harder than Novo’s Semaglutide, Study Suggests
A new, pending‑peer‑review study compares Eli Lilly’s tirzepatide with Novo Nordisk’s semaglutide, confirming tirzepatide delivers greater overall weight loss but also leads to a larger reduction in lean body mass. Researchers used dual‑energy X‑ray absorptiometry to quantify fat‑free mass loss, finding up...

Helsinki’s Algorithmiq Wins €1.7 Million Prize for Quantum-Enabled Light-Sensitive Cancer Drug Discovery
Algorithmiq, a Helsinki‑based quantum software firm, secured the €1.7 million ($2 million) Wellcome Leap prize for demonstrating a practical quantum advantage in drug discovery. The team built an end‑to‑end quantum‑classical workflow that ran on up to 100 qubits to simulate a photosensitiser...

How Clinical Trials Validate New Peptide Therapies
Clinical trials are essential for turning promising peptide molecules into reliable therapeutics by confirming that their effects are consistent, measurable, and reproducible across diverse populations. The article outlines how preclinical research defines mechanisms and safety, while Phase I focuses on human...

This New Test Could Be a Breakthrough in UTI Treatment
Researchers evaluated an experimental rapid susceptibility test, RMD AST, on 352 urine samples and found it matched standard laboratory cultures 96.95% of the time. The assay delivers results in six hours, a dramatic cut from the typical 48‑72‑hour culture period. Faster,...

Multiple Myeloma Drug Blenrep Backed for Wider NHS Use
The UK’s health technology regulator NICE has broadened the approved use of GSK’s BCMA‑targeting drug Blenrep, allowing it to be combined with Takeda’s Velcade and dexamethasone for a larger second‑line multiple myeloma population. The new guidance lifts the restriction that...

Lilly’s Obesity Pill Foundayo Gets 1,390 Prescriptions in Debut Week
Eli Lilly's newly launched oral obesity drug Foundayo filled 1,390 prescriptions in the United States during its debut week ending April 10, according to IQVIA data. By comparison, Novo Nordisk's oral Wegovy recorded 3,071 prescriptions in its first four days...

This Sam Altman-Backed $1.8 Billion Startup Bets AI Can Get Drugs Through Clinical Trials Faster
Formation Bio, backed by Sam Altman and top VCs, has raised $615 million at a $1.8 billion valuation to use AI for faster, cheaper clinical trials. The New York‑based firm plans to acquire a portfolio of about ten early‑stage drug candidates, many stalled...

3 Ways to Invest in the Growing GLP-1 Weight Loss Market
The global GLP‑1 receptor agonist market is set to nearly triple, reaching roughly $185 billion by 2033 with a 12.4% compound annual growth rate. Investors can tap the surge through a direct play in Structure Therapeutics, whose aleniglipron candidate posted a...
Labcorp and CHOP Launch Pediatric Diagnostics Partnership to Expand Advanced Testing and Drive Clinical Lab Growth
Labcorp and the Children’s Hospital of Philadelphia (CHOP) have formed a strategic partnership to accelerate development and commercialization of pediatric‑specific diagnostics. The collaboration will combine CHOP’s research expertise with Labcorp’s nationwide testing network to bring new molecular and genetic assays...

Karnataka Approves AI Centre of Excellence in Bengalurus Electronics City
The Karnataka government approved a four‑year AI‑Biotechnology Centre of Excellence at the Institute of Bioinformatics and Applied Biotechnology in Bengaluru’s Electronics City, allocating roughly $2.4 million. The project partners the institute with the Centre for Cellular and Molecular Platforms (C‑CAMP) and...
The Innovators Working to Make in Vivo Cell Therapy a Reality
First‑in‑human trials of in‑vivo CAR‑T therapies are now underway, delivering therapeutic genes directly inside patients via viral or lipid‑nanoparticle vectors. Big‑pharma interest is evident after AstraZeneca’s $1 bn purchase of EsoBiotec and Eli Lilly’s $2.4 bn acquisition of Orna Therapeutics, despite limited clinical...

Anavex Withdraws EU Application for Alzheimer’s Drug Blarcamesine
Anavex Life Sciences has withdrawn its European marketing authorisation application for blarcamesine, a small‑molecule therapy aimed at early Alzheimer’s disease, after the EMA’s Committee for Medicinal Products for Human Use concluded in December 2025 that the drug’s benefits did not...

Antarka Unveils ANKros-CPD, a DNA Repair Active for Skin Longevity
Antarka introduced ANKros‑CPD, a biotechnology‑derived enzyme that repairs UV‑induced DNA lesions in skin, at in‑cosmetics Global in Paris. The active is a stabilised CPD‑photolyase sourced from Antarctic microorganisms, enabling topical use. By targeting cyclobutane pyrimidine dimers, it addresses DNA damage,...

Bavarian Nordic Reports Swissmedic’s Approval of Vimkunya to Prevent Chikungunya
Swissmedic has granted approval to Bavarian Nordic’s Vimkunya, a single‑dose, virus‑like particle vaccine for chikungunya, targeting individuals 12 years and older. The vaccine is designed to elicit protective immunity as early as one week after injection. Bavarian Nordic has also...

South Korea’s Healthcare Boom Creates New Billionaires
South Korea’s healthcare sector is rapidly generating wealth, propelling several pharma executives onto the nation’s richest list. Sam Chun Dang Pharm’s weight‑loss partnership with Japan’s Daiichi Sankyo lifted chairman Yoon Dae‑in to a $5.9 billion net worth, while ABL Bio and Voronoi secured multibillion‑dollar deals that...

Argobio: The Venture Model Building Europe’s Next Biotech Champions
Argobio, led by Thierry Laugel, raised roughly $54.5 million in 2021 to launch a venture‑builder focused on European biotech. The model has already spun out three companies—Enodia, Laigo Bio and Elkedonia—that together secured over $46.9 million in seed funding for platforms in protein degradation...

Radiopharm Completes Final Patient Dosing of RAD 101 for Diagnosis of Brain Metastases in the US
Radiopharm Theranostics announced the final patient dosing in its U.S. Phase IIb trial of the 18F‑RAD101 PET imaging agent for recurrent brain metastases. The study enrolled 30 patients and interim results demonstrated 90% concordance with MRI, meeting the predefined efficacy...

NTU's AI Chip Detects Disease Biomarkers in 20 Minutes and More Briefs
Researchers at Singapore’s NTU unveiled an AI‑enabled nanophotonic chip that detects microRNA disease biomarkers in about 20 minutes, bypassing traditional PCR methods. South Korea’s Neurophet raised $21.6 million to expand its AI brain‑imaging platform for Alzheimer’s and other neuro‑conditions, targeting the...

Stockholm’s BioLamina Secures €20 Million EIB Loan to Scale Cell Therapies for Chronic Diseases
Swedish biotech scale‑up BioLamina secured a €20 million (≈$22 million) venture‑debt loan from the European Investment Bank to accelerate its laminin‑based cell‑culture platform. The financing will fund expansion of manufacturing capacity, broaden the product portfolio and reinforce the company’s technology base for...

'Self-Regulating' Wound Patch Developed in South Korea
Researchers at KAIST unveiled a self‑regulating wound‑healing patch that merges a 630‑nm organic LED with a ROS‑triggered drug delivery system. The OLED emits uniform light to stimulate cell regeneration while nanocarriers release Centella asiatica extract in proportion to the generated...

Merck’s PD-1/VEGF Data Star in Stacked Lineup of AACR ‘26 Data Reveals
Merck will unveil early clinical data on MK‑2010, a PD‑1/VEGF bispecific antibody it licensed from LaNova for $588 million, at the AACR 2026 meeting. The readout will test Merck’s ability to compete with ivonescimab and other emerging bispecifics from Pfizer/BioNTech and BMS....
Cough Drops From Several Brands Being Recalled, FDA Says
On March 20, the FDA initiated a Class II recall of 15 cough‑drop products sold under five private‑label brands after an inspection of the Chinese manufacturer Xiamen Kang Zhongyuan Biotechnology revealed unspecified concerns. The affected items, many bearing lot 20241030 and expiring...

Stem Cell Editing Programs the Immune System to Make Own Therapeutic Proteins
Researchers at Rockefeller University used CRISPR to edit hematopoietic stem and progenitor cells (HSPCs), programming them to produce therapeutic antibodies or other proteins after vaccination. In mice, as few as 7,000 edited HSPCs generated durable, high‑titer antibody responses that protected...

Biotech Insights - Spring 2026
The FDA’s post‑approval change framework now spotlights GLP‑1 products, exemplified by Wegovy’s tablet approval, and mandates reporting based on the change’s impact—major and moderate changes require supplemental NDAs, while minor changes go in the annual report. In parallel, the Federal...

Will Retatrutide Help Me Lose Weight or Look ‘Shredded’?
Retatrutide, an experimental triple‑hormone peptide, has shown more than 20% body‑weight loss in a 48‑week clinical trial, outperforming existing GLP‑1 drugs like Ozempic and Wegovy. Researchers say it works by modulating GLP‑1, GIP and glucagon pathways to suppress appetite and...

Obesity Treatment Firm Kailera Upsizes IPO to Raise $625 Million
Kailera Therapeutics Inc., a clinical‑stage biotech specializing in obesity treatments, announced an upsized initial public offering. The company sold roughly 39 million shares at $16 each, raising $625 million, the largest U.S. biotech listing since 2021. The offering was priced at the...
From Lockdown to the Lab: Researcher Develops 'Decoy Molecule' To Slow Down Coronavirus
During the COVID‑19 lockdown, Ph.D. candidate Koen Rijpkema engineered decoy molecules that bind tightly to the coronavirus Mac1 enzyme, which normally dampens immune signaling. By mimicking the enzyme’s natural substrate, the decoys keep Mac1 occupied, allowing the immune system to detect...
Menstrual Cycle Reshapes Nearly 200 Blood Proteins, Offering a Broader View of Women's Health
A team at Aarhus University mapped the blood proteome across the menstrual cycle, identifying nearly 200 proteins that fluctuate systematically. The study, published in Nature Medicine, reveals that these changes affect immune, hormonal, and metabolic pathways far more than previously...

Funding the Future of European Biotech
In a BioSpace Insights “Denatured” podcast, host Jennifer C. Smith‑Parker talks with Edoardo Negroni of AurorA‑TT and Naveed Siddiqi of Novo Holdings about Europe’s world‑class biotech science and the venture ecosystem needed to commercialize it. The guests argue that Europe’s research...

High-Precision Human Immune Aging Clock Identifies RUNX1 as Key Target for T Cell Senescence
Researchers from the Chinese Academy of Sciences unveiled a high‑precision Human Immune Aging Clock (HIAC) that leverages single‑cell multi‑omics to predict immune age with a 5.66‑year mean absolute error. The clock identifies T cells as the most sensitive cellular indicator...
Weight Loss, Obesity Drugs Bring Potential New MASLD, MASH Treatment Strategies
A new review in *Diabetes, Obesity and Metabolism* shows that GLP‑1, GIP and glucagon‑based drugs, originally approved for obesity and diabetes, also improve liver outcomes in metabolic dysfunction‑associated steatotic liver disease (MASLD) and its progressive form MASH. A recent meta‑analysis...

They Froze a Brain to −196°C. Then Brought It ‘Back to Life’ in a Groundbreaking New Study.
Researchers at the University Hospital Erlangen demonstrated that mouse hippocampal tissue can survive vitrification at –196 °C and resume normal neuronal activity after rewarming. The study, published in PNAS, showed structural integrity and functional synaptic signaling in brain slices, with modest...
Designing Implants that Don’t Scar the Brain
A new study systematically compared stiff silicon electrodes with flexible polyimide probes for intracortical neural implants. The researchers found that material choice dominates tissue response: polyimide probes trigger far less scarring and inflammation than silicon, while probe thickness or wireless...

Kennedy: 90% Of FDA Reviewers Are Using AI For Faster Drug Approvals
U.S. Health and Human Services Secretary Robert F. Kennedy Jr. told the House Ways & Means Committee that more than 90% of FDA reviewers are now using artificial‑intelligence tools to speed drug approvals. The AI applications are also being rolled...

Spotlight On: Biosimilar Litigations - April 2026
The April 2026 Spotlight On: Biosimilar Litigations memo outlines which patent disputes are tracked in the sector. It clarifies that lawsuits between biosimilar applicants or manufacturers and reference‑product sponsors are included, while conflicts solely among reference sponsors or non‑practicing entities are...

UK Says It Has Hit Target on Commercial Trial Set-Up Times
The UK’s National Institute for Health and Care Research (NIHR) reported that average clinical‑trial set‑up time fell to 122 days in the six months to March, beating its 150‑day target set for March 2026. The reduction follows a suite of reforms,...

Staging, ctDNA, and the Art of Personalizing Metastatic Breast Cancer Therapy: Hayley Knollman, MD
Hayley M. Knollman, MD, highlighted how estrogen‑receptor‑positive metastatic breast cancer still relies on conventional staging—blood work, imaging, and tissue biopsies—while emerging HER2‑low categories gain relevance only after disease spreads. She noted that circulating tumor DNA (ctDNA) and broad genomic panels are now...

OpenAI Debuts GPT-Rosalind, a New Limited Access Model for Life Sciences, and Broader Codex Plugin on Github
OpenAI unveiled GPT‑Rosalind, a domain‑specific reasoning model built to accelerate life‑science research, alongside a Codex plugin that links the model to over 50 public multi‑omics databases. The model demonstrated top‑tier performance on benchmarks such as BixBench and LABBench2, surpassing GPT‑5.4...

For Regrowing Human Limbs, This Salamander Gene Could Hold the Key
Scientists identified SP6 and SP8 as conserved genes that drive limb regeneration in axolotls, zebrafish and mice, and demonstrated that a viral gene‑therapy delivering FGF8 can partially rescue digit regrowth in mice lacking these genes. The work, published in PNAS,...

FDA Signals Potential Expansion of Testosterone Therapy to Treat Low Libido in Idiopathic Hypogonadism
The FDA announced it will entertain supplemental new drug applications to add low libido in men with idiopathic hypogonadism as an approved indication for existing testosterone replacement therapy (TRT) products. The move follows a December 2025 expert panel review of...

Clinical Trial of a Prion Disease Drug Candidate Begins Enrolling Participants
Broad Institute and UMass Chan have launched the first human trial of a prion disease therapy, a divalent small interfering RNA designed to silence the prion protein gene. The phase 1 PRiSM study will enroll 15 symptomatic patients to assess...
RSV Vaccines Work to Prevent Hospitalization
Recent clinical data show that newly approved respiratory syncytial virus (RSV) vaccines dramatically cut hospital admissions. In infants, the vaccine lowered hospitalization risk by roughly 70%, while older adults experienced a 50% reduction in severe cases. The FDA has accelerated...
Roche to Start Phase III Trial to Broaden Access to Elevidys in Duchenne Muscular Dystrophy
Roche announced a global, pivotal Phase III trial of Elevidys, its gene‑therapy for Duchenne muscular dystrophy, targeting roughly 100 early‑ambulatory boys. The 72‑week, placebo‑controlled study will assess change in time‑to‑rise‑from‑floor velocity as the primary efficacy endpoint. Results are intended to bolster...

Study Data Technical Conformance Guide - Technical Specifications Document
The FDA has issued the final Study Data Technical Conformance Guide (Docket FDA-2014-D-0092), outlining technical specifications for electronic study data submissions. The guidance clarifies the agency’s expectations but remains non‑binding, allowing sponsors to use alternative approaches that meet regulatory requirements....

Financial Transparency and Efficiency of the Prescription Drug User Fee Act, Biosimilar User Fee Act, and Generic Drug User Fee...
The FDA announced a public meeting on June 23, 2026 to discuss financial transparency and efficiency of the Prescription Drug User Fee Act (PDUFA VII), Biosimilar User Fee Act (BsUFA III) and Generic Drug User Fee Act (GDUFA III). The session will present five‑year financial...