
Inhibrx Says Combo Therapy Shrank More Tumors than Merck's Keytruda Alone
San Diego‑based biotech Inhibrx reported that its experimental antibody INBRX‑106, when paired with Merck’s immunotherapy Keytruda, produced a higher rate of tumor shrinkage than Keytruda alone in patients with metastatic head and neck squamous cell carcinoma. The early‑stage study enrolled heavily pre‑treated patients and showed an improved objective response without adding significant toxicity. Inhibrx said the data support advancing the combination into a larger Phase 2 trial later this year. The company plans to file additional regulatory filings based on these results.
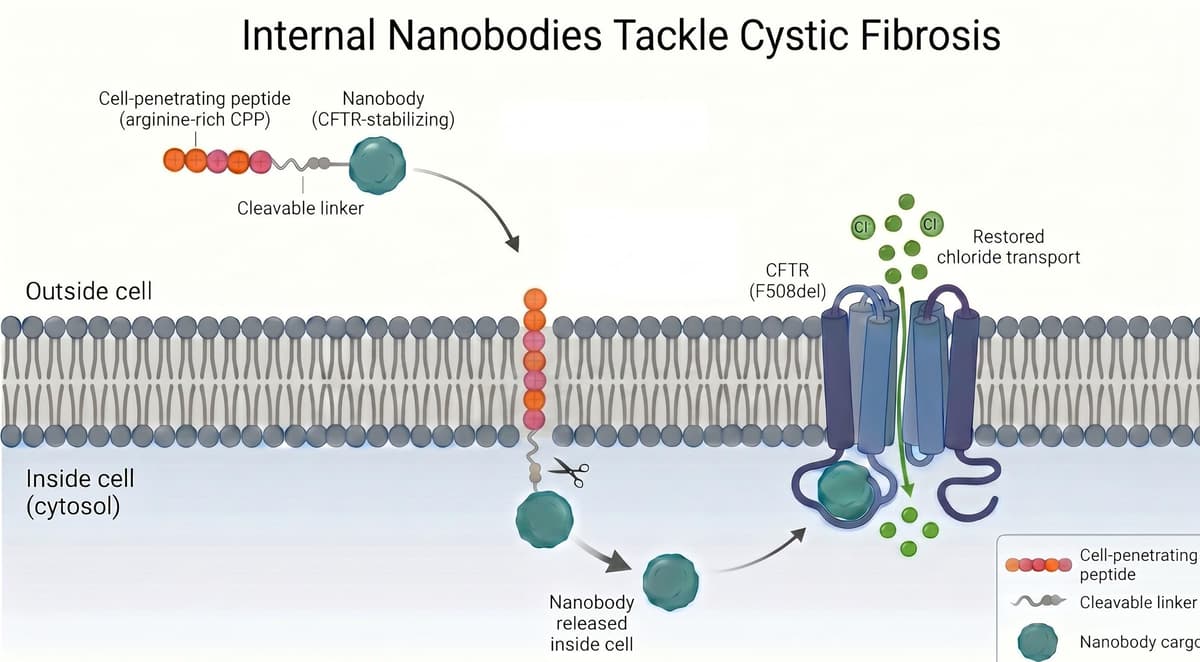
Internal Nanobodies Tackle Cystic Fibrosis
Researchers have engineered a cell‑penetrating nanobody that enters airway cells and stabilizes the misfolded CFTR protein responsible for cystic fibrosis. By fusing the nanobody to a ten‑arginine peptide, the hybrid molecule crosses the cell membrane and restores up to 90%...
Ocrevus Slows Disability Progression in Advanced PPMS, Trial Finds
A Phase 3 ORATORIO‑HAND trial involving more than 1,000 adults with primary progressive multiple sclerosis (PPMS) showed that Ocrevus (ocrelizumab) significantly slows disability progression. Over a median follow‑up of nearly three years, the drug reduced the risk of confirmed disability...

US Government Spends Hundreds of Millions on Biotech Pilot Plants as National Security Priority
The U.S. government is pouring hundreds of millions of dollars into BioMADE, a public‑private consortium aimed at scaling bio‑manufacturing for food, defense and industrial applications. Since its 2021 launch, BioMADE has secured $87 million from the Department of Defense (DoD), $450 million...
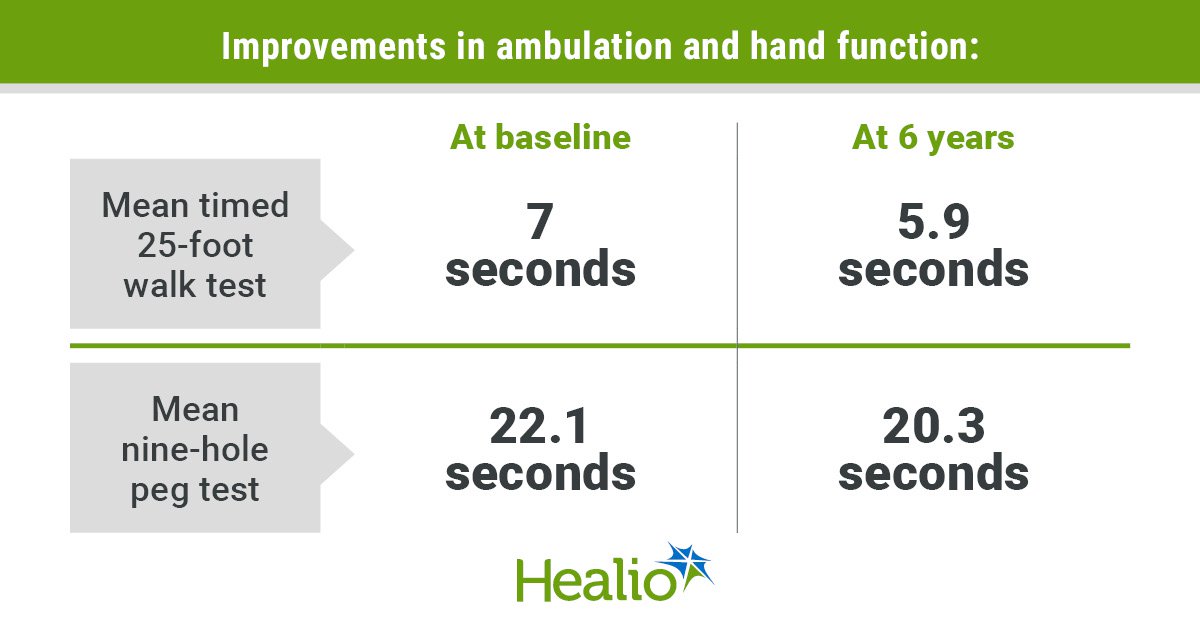
Ocrelizumab Preserves Ambulation, Hand Function in MS
A six‑year analysis of the ENSEMBLE trial shows that early‑stage relapsing‑remitting multiple sclerosis patients treated with ocrelizumab largely maintained functional ability. 86.1% preserved normal ambulation and 93% kept normal hand dexterity throughout the study, while 34% of those with baseline...

Braveheart Bio's Hengrui-Licensed Cardiac Drug Scores Second Clinical Win
Braveheart Bio announced that its heart‑muscle therapy, licensed from China’s Jiangsu Hengrui Pharmaceuticals, achieved its primary endpoint in a mid‑stage (Phase 2) trial for heart failure with reduced ejection fraction. The study reported an 8% absolute improvement in left‑ventricular ejection fraction...

Drug Repurposing
The FDA has issued a request for public comment on drug repurposing, seeking input from patients, clinicians, and researchers on priority disease areas and candidate drugs. The initiative builds on existing programs such as Project Renewal, which updates oncology drug...

The Next Frontier for Hantavirus: Finding Vaccines and Treatments
A hantavirus outbreak on a cruise ship has killed three people and sickened several others, marking the first documented person‑to‑person transmission of the disease. The strain’s heightened transmissibility has revived interest in vaccine and therapeutic candidates that were previously shelved...

QP Essentials: Reducing Risk and Delays in EU and UK Trials
Clinical trial sponsors often encounter delays when shipping supplies to the EU and UK because Qualified Persons must certify each batch. Catalent’s webinar outlines the core QP responsibilities, highlights differences between EU and UK regulations, and offers practical steps such...
FDA Grants Fast Track Designation to Zai Lab’s DLL3-Targeting ADC for epNECs
Zai Lab’s DLL3‑targeting antibody‑drug conjugate zocilurtatug pelitecan received FDA Fast Track designation for extrapulmonary neuroendocrine carcinomas (epNECs). Early‑stage data showed a 38.2% objective response rate in heavily pre‑treated patients, indicating meaningful antitumor activity. The designation promises more frequent FDA interactions,...
Study Compares Two Antibiotics in Treating Severe Hemorrhagic Bacterial Pneumonia
Researchers at Osaka Metropolitan University compared cefiderocol (CFDC) and levofloxacin (LVFX) in a mouse model of severe hemorrhagic pneumonia caused by multidrug‑resistant Stenotrophomonas maltophilia. Both drugs significantly increased survival and reduced bacterial loads in lungs and heart relative to untreated...
HIV-1 Strains Reveal Varied Paths to Antibody Escape
Scientists have mapped how HIV‑1 strains evade broadly neutralizing antibodies (bnAbs) through multiple, strain‑specific pathways. Using genomic sequencing, cryo‑EM and X‑ray crystallography, the team showed that the virus remodels its envelope glycoprotein (Env) via glycan shield alterations, loop mutations and...

Inside CSL’s Strategy to Simplify a Biotech Giant
CSL Limited is entering a strategic simplification phase, announced in August 2025, that will see its vaccine arm Seqirus spun off as a separate ASX‑listed company by the end of 2026 and a restructuring program that could cut up to 15% of...
Novo Nordisk microRNA Drug Fluffs Its Lines in Heart Failure
Novo Nordisk’s microRNA‑targeting drug CDR132L failed to demonstrate a statistically significant improvement in left ventricular end‑systolic volume index in the phase 2 HF‑REVERT trial. The study enrolled 280 post‑myocardial‑infarction patients with an ejection fraction of 45% or lower and elevated NT‑proBNP...
FUJIFILM Biosciences and NextCell Launch New Commercial Platform Comprised of RUO Stromal Cells and Cell Culture Media
FUJIFILM Biosciences and NextCell Pharma have launched a global commercial platform that pairs FUJIFILM’s PRIME‑XV MSC Expansion XSFM medium with NextCell’s research‑use‑only umbilical‑cord mesenchymal stromal cell product, NextCell‑Cord RUO. The bundle offers a standardized, scalable solution for MSC research, addressing...

Novo Hands Over Parkinson’s Therapy to Zuckerberg-Backed Startup
Novo Nordisk agreed to transfer its experimental Parkinson’s disease therapy to Cellular Intelligence, an AI startup backed by Mark Zuckerberg, to accelerate development. Cellular Intelligence will assume responsibility for clinical‑trial execution, while Novo retains rights to milestone payments and royalties...
GSK Enlists Local Aid for Chinese Rollout of Hep B Drug
GlaxoSmithKline (GSK) has signed a five‑and‑a‑half‑year agreement with Sino Biopharm’s subsidiary Chia Tai Tianqing Pharma (CTTQ) to import, distribute and promote its investigational hepatitis B antisense drug bepirovirsen across more than 5,000 Chinese medical centres, pending NMPA approval. The partnership gives...

Makary’s Reported FDA Removal Could Be 'Broad Positive' For Biopharma
President Donald Trump reportedly agreed to remove FDA Commissioner Marty Makary, a move confirmed by the Wall Street Journal and echoed by anonymous sources. The announcement follows the recent departure of CBER director Vinay Prasad and adds to a pattern of...

PulseSight Therapeutics Reports P-I (PST-611-CT1) Trial Data on PST-611 in Dry AMD/Geographic Atrophy
PulseSight Therapeutics presented Phase I (PST-611-CT1) trial data for PST-611 in dry age‑related macular degeneration (geographic atrophy) at ARVO 2026. The study enrolled six patients across two dose cohorts and demonstrated favorable safety, tolerability, stable best‑corrected visual acuity, and no...

Deal Finally Unlocks NHS Access to ITF's Duchenne Drug
ITF Pharma’s oral HDAC inhibitor Duvyzat has secured NICE endorsement for ambulatory Duchenne muscular dystrophy patients, unlocking access for more than 500 NHS England cases. The drug will be funded immediately through the Innovative Medicines Fund while NHS England arranges...

European Regulators Greenlight Fractyl Health's Clinical Test of GLP-1 Gene Therapy
European regulators have granted Fractyl Health permission to begin the first human trial of a GLP‑1 gene‑therapy candidate. The Phase 1 study will test a single‑dose AAV‑based vector designed to produce continuous GLP‑1, aiming to replace daily injections for type 2 diabetes...

Quality CDMO, Inc. - 665961 - 05/01/2026
The U.S. Food and Drug Administration issued a closeout letter to Quality CDMO, Inc., confirming that the firm’s corrective actions have addressed the violations cited in the September 11, 2024 warning letter. While the FDA acknowledges the remediation, it emphasizes that compliance...

CRISPRi Screening Identifies Fungal-Specific Drug Targets
Researchers adapted a pooled CRISPR interference (CRISPRi) platform for Candida albicans, allowing high‑throughput repression of 130 essential, fungal‑specific genes. The screen revealed dosage‑sensitive vulnerabilities across ten infection‑relevant stress conditions. Testing two drug‑resistant clinical isolates showed most hits are conserved, indicating...

ABEC Expands Process Sciences Support for Biomanufacturing
ABEC announced the expansion of its Process Sciences group to provide biopharmaceutical manufacturers with end‑to‑end support across the bioprocess lifecycle. The team combines chemical and bio‑engineering expertise with advanced modeling, experimental testing, and custom equipment design, covering everything from early...
Lipid Profiles in Amygdala: Sporadic vs GBA Parkinson’s
On May 11, 2026 a cluster of research releases highlighted breakthroughs across biomedicine and technology. Rapid nanopore sequencing was deployed to map malaria transmission across Africa, while a new study linked ACAD8 deficiency to cardiac hypertrophy through histone modifications. Parallel...

20 Future Scottish HealthTech and MedTech Leaders
Scotland’s health‑tech and med‑tech ecosystem is shifting from academic clusters to a global industrial powerhouse, propelled by the Life Sciences Strategy for Scotland 2035. The sector already generated over £10 billion (≈$13 bn) by early 2021, beating its interim £8 billion target four...

A Single Infusion Could Suppress H.I.V. for Years, Study Suggests
Scientists presented early data showing that a single infusion of genetically engineered immune cells can drive HIV to undetectable levels in patients. In a small study, two participants received the therapy, with one maintaining viral suppression for almost two years....

When Reliability Becomes Patient Care in Nuclear Medicine
Nuclear medicine is evolving from a purely diagnostic tool to a therapeutic platform through radioligand therapy and theranostics, where the same molecular target guides both imaging and treatment. Because radiopharmaceuticals decay continuously, manufacturing, release, and distribution must function as a...

LanzaTech & DTU to Open Biofoundry to Turn Carbon Emissions Into High-Value Products
U.S. synthetic‑biology firm LanzaTech has signed a two‑year agreement with Denmark’s Technical University (DTU) Bright hub to launch an AI‑powered C1 biofoundry. The facility will use engineered microbes to convert methane, carbon monoxide and carbon dioxide from industrial waste streams...

Precision in Motion: Decoding the Critical Operations Behind the Logistics of Next-Generation Cell and Gene Therapies
Cell and gene therapies (CGTs) are transforming personalized medicine but demand ultra‑precise logistics because they are patient‑specific, highly sensitive, and often viable for only minutes to hours. Traditional freight cannot meet the sub‑30‑minute delivery windows, cryogenic temperature requirements, and real‑time...

The Capacity Crunch in Biopharma Is a Location Problem
Biopharma manufacturers are hitting a capacity crunch as demand for cell and gene therapies outpaces the construction of new GMP‑ready plants, which can take years. Companies are therefore prioritizing locations where compliant facilities, utilities and cold‑chain logistics already exist. Middlesex...

Day One Concludes Patient Enrolment for Tovorafenib’s FIREFLY-2 Trial
Day One Biopharmaceuticals announced that the Phase III FIREFLY‑2 trial of tovorafenib has finished enrolling roughly 400 pediatric low‑grade glioma patients across 140 sites worldwide. The open‑label, randomized study pits oral tovorafenib against four standard chemotherapy regimens as a first‑line option...
STAT+: Medicare Is Spending Far Less than Expected on New Alzheimer’s Drugs
Medicare’s uptake of the newly approved Alzheimer’s therapies Leqembi and Kisunla is far lower than projected, prompting the agency to forecast minimal spending through 2026‑2027. Earlier estimates had anticipated billions in annual costs, but real‑world adoption has stalled. The drugs...

STAT+: Five Years After Disaster, a Rare Disease Community Gets New Chance at Treatment
Astellas Pharma has re‑initiated its gene‑therapy trial for X‑linked myotubular myopathy (XLMTM) five years after a previous study was halted by safety concerns. Early observations from the first participant, Joshua "JJ" Gonzalez, indicate a dramatic reduction in airway suctioning, suggesting...

Henlius Receives US FDA IND Clearance for HLX05-N (Biosimilar, Erbitux)
Henlius announced that the U.S. Food and Drug Administration granted Investigational New Drug (IND) clearance for HLX05‑N, its biosimilar version of Erbitux (cetuximab), aimed at treating metastatic colorectal cancer. The IND follows extensive analytical and non‑clinical studies that demonstrated high...

Nuclera Launches Antibody Triage Service to Advance AI-Driven Antibody Discovery
Nuclera, a biotech firm specializing in protein production, announced a new antibody‑screening service designed to accelerate AI‑driven antibody discovery. The offering uses a 96‑plex cell‑free expression platform and surface plasmon resonance to triage large in‑silico‑generated libraries, delivering early binding data...

Bologna’s Cellply Secures €7.15 Million to Develop Tools for Faster Immunotherapy and Cell Therapy Development
Italian deep‑tech startup Cellply has closed a €7.15 million ($7.8 million) funding round led by ENEA Tech e Biomedical. The capital will fund global rollout of its VivaCyte® single‑cell analysis platform and development of next‑generation ATMP quality‑control tools. The round also saw...
BMS’ Sotyktu (Deucravacitinib) Wins EC Approval to Treat Adults with Active Psoriatic Arthritis (PsA)
The European Commission has approved Bristol‑Myers‑Squibb’s oral drug Sotyktu (deucravacitinib) for adults with active psoriatic arthritis, either alone or with methotrexate. The decision follows positive Phase III data from the POETYK PsA‑1 and POETYK PsA‑2 trials, which enrolled over 1,200 patients and met...

Patient Perspectives on Gene Therapies and Gene Editing for Familial Cardiomyopathies
Early‑phase trials are testing gene replacement and editing for hypertrophic and dilated cardiomyopathies, yet patient viewpoints remain understudied. Interviews with 21 adults (average age 57, 57% female) revealed four decision drivers: perceived disease severity, quality‑of‑life impact, treatment safety and delivery,...

CEAS Collective: Bridging Biotech Innovation and Consumer Wellness Through Direct-to-Patient Delivery
CEAS Collective is positioning itself at the intersection of biotech and consumer wellness by offering science‑driven products through a direct‑to‑patient delivery model. The company emphasizes rigorous quality control, sourcing botanicals with verified purity, and a digital logistics platform that bypasses...
Advances in RSV Vaccine Research and Development
Respiratory syncytial virus (RSV) remains a leading cause of acute lower‑respiratory infections, accounting for roughly 33 million cases and over 3 million hospitalizations each year, especially in children under five and older adults. Recent advances focus on stabilizing the prefusion F (preF)...

Hong Kong Outlines Strategy to Strengthen Global Health Innovation and Clinical Trials
Hong Kong announced a comprehensive strategy to become a regional hub for health innovation, focusing on expanding clinical trial capacity, regulatory reform, and research commercialization. The government unveiled the Greater Bay Area International Clinical Trial Institute, a “one institute, one...

Merck, Amgen Double Down on Bad Cholesterol to Vanquish Number 1 Killer
Merck’s oral PCSK9 inhibitor enlicitide cut LDL‑C by 64.6% in an eight‑week Phase III trial, outperforming other oral non‑statin drugs. Updated ACC/AHA lipid guidelines now require LDL‑C < 55 mg/dL for ASCVD patients, leaving roughly 70% of statin users above target. The tighter goals...

Published FDA Rejections Point to Manufacturing, Data Gaps as Key Stumbling Blocks
The FDA’s new policy to publish complete response letters (CRLs) has made over 350 rejection letters publicly available, revealing that more than half of drug rejections stem from manufacturing problems and 41% from product‑quality issues. Analysts say the transparency lets...

BeOne Medicines Recognized as "Outstanding Global Oncology Company of the Year" At HKCT Business Awards 2026
BeOne Medicines was named "Outstanding Global Oncology Company of the Year" at the 2026 HKCT Business Awards, recognizing its rapid R&D progress and manufacturing capabilities. The Swiss‑based firm now operates in more than 45 markets, employs over 1,200 oncology R&D...

Hong Kong Highlights Innovation and Greater Bay Area Collaboration at Hospital Authority Convention 2026
Hong Kong officials used the Hospital Authority Convention 2026 to unveil a suite of health‑innovation policies, including faster drug approvals, a new regulatory centre, and expanded AI use in hospitals. A Greater Bay Area Clinical Trial Collaboration Platform will allow...
StockWatch: EnGene Shares Crater on Declines in Complete Response Rates to Bladder Cancer Therapy
enGene (ENGN) shares plunged 83% after Phase II LEGEND trial data showed its gene therapy detalimogene achieved a 43% complete response at six months, far below the 62% benchmark previously reported. The updated data also revealed a 13.3% twelve‑month response and...

Certara Inc (CERT) Q1 2026 Earnings Call Transcript
Certara reported a record Q1 2021 with revenue of $66.7 million, up 16% year‑over‑year, and adjusted EBITDA rising 20% to $23.9 million. Bookings surged 34% YoY, driven by strong demand for both software and services, while services bookings jumped 39%. The company...
Heron Therapeutics Inc (HRTX) Q1 2026 Earnings Call Transcript
Heron Therapeutics reported $155 million in 2025 net product revenue and adjusted EBITDA of $14.7 million, surpassing its guidance range. Acute‑care sales surged, with ZYNRELEF revenue up 48% YoY and the franchise growing 57% in Q4, driven by the CrossLink IGNITE program, a...

Exagen Inc (XGN) Q1 2026 Earnings Call Transcript
Exagen reported Q1 2022 revenue of $10.4 million, driven by a record 30,903 AVISE CTD/Lupus tests and 2,175 ordering providers. The company secured a new Medicare PLA code (0312U) for AVISE Lupus, boosting reimbursement from $295 to $1,085 per test. Gross...