
Finland’s Algorithmiq Takes Top Spot in $50M Q4Bio Challenge with Quantum Drug Simulation Milestone
Finland’s Algorithmiq won the $2 million prize in Wellcome Leap’s $50 million Q4Bio challenge, becoming the first to demonstrate an end‑to‑end quantum‑classical workflow for drug simulation on real hardware. Using up to 100 qubits on IBM’s quantum computer, the team modeled the activation pathway of a photodynamic‑therapy drug currently in Phase II trials. Backed by IBM, Microsoft and Cleveland Clinic, the win positions Algorithmiq at the forefront of quantum‑enabled drug discovery. The funding will accelerate commercialization and expand AI‑integrated pipelines.

Theradaptive Receives US FDA Approval to Initiate P-III Trial of OsteoAdapt SP for Spinal Fusion Procedures
Theradaptive has secured U.S. FDA approval to launch the pivotal OASIS Phase III trial evaluating its OsteoAdapt SP device as a bone‑graft alternative in transforaminal, anterior, and lateral lumbar interbody fusion (TLIF, ALIF, LLIF) procedures. The trial follows earlier studies involving...
AstraZeneca’s Breast Cancer Drug Fails to Earn Backing of FDA Advisory Committee
The FDA’s advisory committee voted against recommending AstraZeneca’s oral SERD camizestrant for HR⁺/HER2‑ metastatic breast cancer patients with an ESR1 mutation, citing concerns over the Phase 3 SERENA‑6 trial design. The study switched patients to camizestrant at the point of mutation...

Lilly's Double-Beat Widens the GLP-1 Gap—And a New Pill Could Make It Permanent
Eli Lilly posted a strong Q1 2026, with revenue up 56% YoY to almost $20 billion, driven by $12.8 billion in GLP‑1 sales from Mounjaro and Zepbound. The FDA also approved Foundayo, the first oral GLP‑1 agonist for obesity that can be taken without...

BIO on the American Road Tours Gene Therapy Hub in Ohio
BIO President John F. Crowley toured Ohio on April 28, spotlighting the state’s emerging gene‑therapy hub. Researchers at Nationwide Children’s Hospital have delivered two of the FDA’s first eight approved gene therapies for Duchenne muscular dystrophy and spinal muscular atrophy type 1....
Corcept Ties ALS Drug to Improved 2-Year Survival as Phase 3 Start Date Nears
Corcept Therapeutics reported that its experimental ALS drug dazucorilant reduced the two‑year risk of death by roughly 87% in a Phase 2 extension study, despite missing its primary motor‑function endpoint. The survival benefit was most pronounced at the 300 mg dose, though...

Seaport, Hemab Price IPOs, While Avalyn Soars in Nasdaq Debut
Three biotech companies priced their initial public offerings between Thursday and Friday, collectively raising more than $850 million. Seaport Therapeutics secured $225 million at $20 per share, while Hemab Therapeutics attracted $210 million at $18 per share. Avalyn Pharma led the pack with...

MDMA-Assisted Therapy for Depression: A Promising but Early First Step
A small open‑label proof‑of‑principle study examined MDMA‑assisted therapy in 12 adults with moderate‑to‑severe major depressive disorder. Participants received two MDMA dosing sessions spaced a month apart together with nine psychotherapy sessions. At two months, 75% of participants achieved remission and...

Cue Biopharma Licenses Ascendant-221 From Ascendant Health Sciences in a ~$691.5M Deal
Cue Biopharma announced an exclusive license with Ascendant Health Sciences for Ascendant-221, an anti‑IgE monoclonal antibody aimed at allergic diseases. The agreement grants Cue worldwide rights, except Greater China, in exchange for a $15 million upfront payment and up to $676.5 million...

Approaches to Reducing Toxicity and Side Effects in Cell and Gene Therapy
Cell and gene therapies are expanding rapidly, with the market projected to exceed $9 billion in 2025 and grow over 15% annually through 2035. Safety remains a hurdle, prompting multiple strategies to curb cytokine release syndrome (CRS) and related toxicities. Companies...

FDA's ODAC Delivers One Loss, One Win for AstraZeneca
The FDA’s Oncologic Drugs Advisory Committee (ODAC) voted 6‑3 against AstraZeneca’s oral SERD camizestrant in combination with CDK‑4/6 inhibitors for first‑line HR‑positive, HER2‑negative breast cancer with ESR1 mutations, citing a lack of overall survival benefit despite a 56% progression‑free survival...

FDA Taps Szarama as Acting CBER Director as Prasad’s Tumultuous Tenure Ends
Vinay Prasad is leaving his role as head of the FDA’s Center for Biologics Evaluation and Research (CBER) after a contentious tenure that included the Elevidys gene‑therapy shutdown and multiple rare‑disease therapy rejections. The agency has named his deputy, biophysicist Katherine Szarama,...
Gene Editing at Scale, Clinic Seeks Generalizable Therapies
Integrated DNA Technologies helped deliver a CRISPR therapy that rescued baby KJ Muldoon from a fatal urea‑cycle disorder, proving gene editing can correct a single disease‑causing mutation. The success highlights the field’s next hurdle: scaling personalized edits for disorders with...

Smarter AAVs Drive Gene Therapy’s Next Chapter
Gene therapy’s growth is hampered by AAV manufacturing bottlenecks, safety concerns, and high costs, prompting a wave of innovations across bioprocessing, analytics, and vector design. Companies like Thermo Fisher, PackGene, Catalent, and Asimov are deploying design‑space modeling, high‑throughput purification, and...

What’s Next in the Evolution of Standards for Biologics Development
The United States Pharmacopeia (USP) is redefining how documentary standards support biologics, moving from product‑specific monographs toward a hybrid model that blends platform‑based chapters, emerging standards, and analytical reference materials. This shift addresses the growing complexity of monoclonal antibodies, ADCs,...
Identifying the Ages when Alzheimer’s Biomarkers Sharply Change
A Mayo Clinic Study of Aging analysis identified specific ages when Alzheimer’s disease biomarkers change sharply, using breakpoint regression on plasma proteins, PET imaging, hippocampal volume and cognition across 45‑90‑year‑olds. The most consistent inflection points clustered between 62 and 71...

The Cost of Inefficiencies in Clinical Trials
Clinical trials are hampered by fragmented technology, leading to roughly 45% higher costs compared with unified platforms. Integration fees, duplicate data entry, and managing multiple vendors add significant expense and delay timelines. Unified data‑management solutions such as CRScube claim 50‑60%...

How One Startup Turned Extinction Into a Multi-Billion-Dollar Science Movement
Serial entrepreneur Ben Lamm’s Colossal Biosciences, a Texas‑based de‑extinction startup, announced progress reviving the extinct Blue Buck, adding another mammal to its portfolio that already includes woolly mammoths, dire wolves and the Tasmanian tiger. The effort now spans nearly every...

How Saudi Arabia Is Developing as a Middle East Hub for Clinical Trials
Saudi Arabia is positioning itself as the Middle East’s primary hub for clinical trials, backed by robust state investment and the Vision 2030 Health Sector Transformation Programme. Healthcare spending is forecast to reach $61.05 million in 2026, while pharmaceutical sales are...
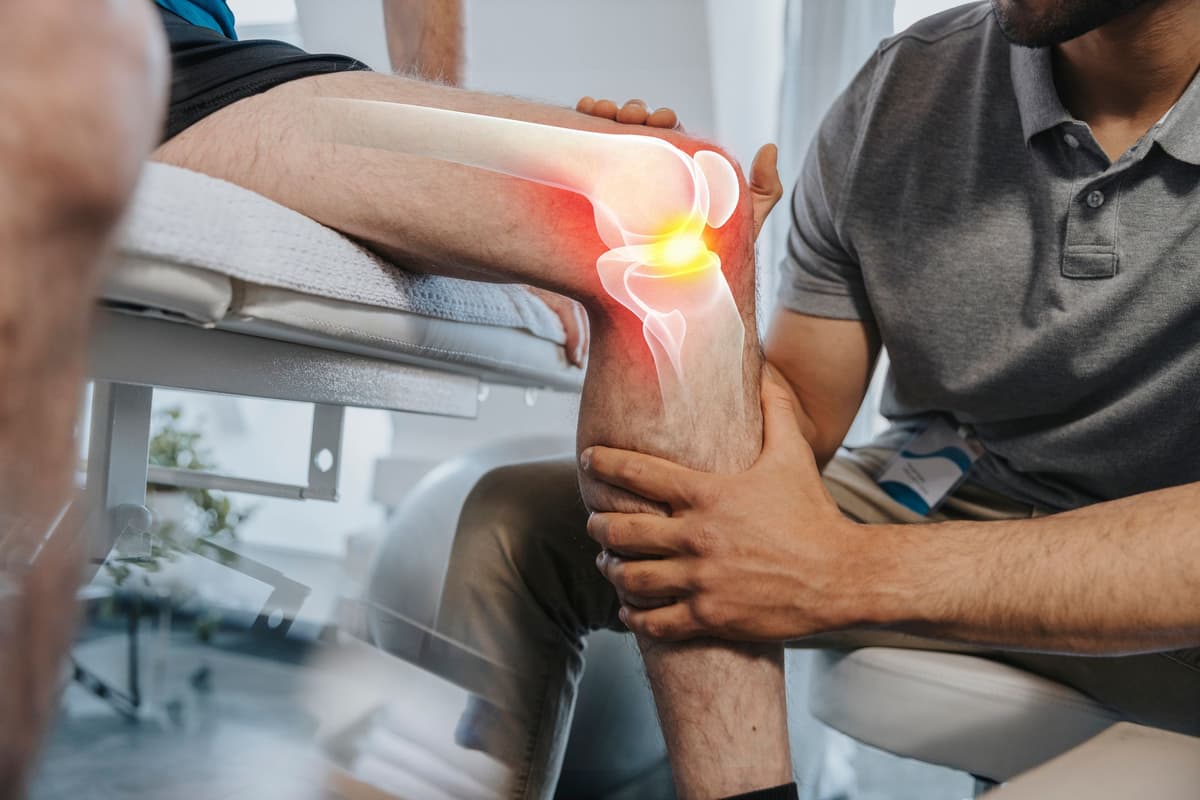
This Treatment Could Reverse Osteoarthritis Joint Damage With a Single Injection
Researchers at the University of Colorado Boulder have secured a $33.5 million ARPA‑H grant to develop a regenerative osteoarthritis therapy that could reverse joint damage with a single injection. The approach uses a controlled‑release particle system to deliver an approved drug...

Comparative Assessment of Brassica Nigra Seed and Its Sprout Ethanolic Extracts Against Paracetamol-Induced Hepatotoxicity in Rats: Insight Into Antioxidant and...
Researchers compared ethanolic extracts of Brassica nigra (black mustard) seeds and sprouts for protecting rat livers against a high‑dose paracetamol challenge. Rats received 500 mg/kg body weight of each extract for 21 days, with silymarin (100 mg/kg) as a reference drug. Both extracts...

Solvent‐Free Preparation of Pharmaceutically Active Diindolylmethane Derivatives by Stable Triple‐Interpenetrated Heterometallic‐Organic Frameworks With Sc‐Pd/Pt Synergistic Sites
Researchers reported a one‑pot synthesis of triple‑interpenetrated heterometallic‑organic frameworks (HMOFs) containing Sc‑Pd or Sc‑Pt synergistic sites. The resulting ScPd‑INA, ScPt‑INA and ScPt‑MCA catalysts achieved up to 99% conversion in solvent‑free Friedel‑Crafts alkylation of diindolylmethane (DIM) precursors. Single‑crystal X‑ray diffraction confirmed...
STAT+: Her Daughter Mila Got a Bespoke Medicine. Now She’s Starting a New Biotech to Make More
Julia Vitarello, whose eight‑year‑old daughter Mila was treated with a tailor‑made gene therapy, announced she is launching a new biotech to scale individualized medicines. Her previous company, EveryONE Medicines, folded after FDA guidance on custom therapies proved insufficient for investors....

Orf Genetics on Cutting Growth Factor Costs and Supplying Cultivated Meat Sector
Orf Genetics, an Icelandic biotech firm, is cutting cultivated‑meat growth‑factor costs by producing them in barley instead of traditional E. coli systems. The barley‑based platform delivers comparable bioactivity at a fraction of the price and is already used by roughly 150...

LEO Pharma to Acquire Replay for Rare Skin Disease Gene Therapy Platform
LEO Pharma has signed a definitive agreement to acquire gene‑therapy specialist Replay for an upfront cash payment of $50 million, with additional milestone fees and tiered single‑digit royalties. Replay brings a herpes simplex virus (HSV) platform that delivers large therapeutic genes...

Scientists Build Drug-Carrying DNA Robots to Target Diseases
Scientists have engineered microscopic DNA robots that can carry therapeutic payloads and seek out viruses, acting as nano‑surgeons within the bloodstream. By applying origami‑inspired rigid joints and flexible components, the robots achieve nanometer‑scale precision. Movement is programmed through DNA strand...
Pinnacle Food Group Eyes Open-Source Precision Fermentation Hub in Hong Kong
Canadian biotech firm Pinnacle Food Group has signed a non‑binding MoU with the Open Yeast Collection and Bioboost Synbio Consulting to explore an Open Yeast Platform hub in Hong Kong‑Shenzhen Innovation and Technology Park. The hub would combine an open‑access...
Beacon Biosignals Is Mapping the Brain During Sleep
Beacon Biosignals has launched an FDA‑cleared, lightweight EEG headband that records clinical‑grade brain activity while users sleep at home. The device’s machine‑learning platform extracts detailed sleep‑stage metrics and subtle architecture changes, supporting more than 40 global clinical trials for conditions...
Food Timing May Shape How T Cells Respond to Infection and Therapy
A Nature study shows that lipids released after a meal rapidly reprogram T‑cell metabolism, boosting glucose uptake, mitochondrial mass and cytokine production. Researchers observed these effects in both human donors and mice, with fed‑state T cells showing enhanced proliferation and...
![[Comment] GLP-1 Therapies: An Emerging Approach for Alcohol Reduction?](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/f628ae5758874c46e9810dd10e2c007b.webp)
[Comment] GLP-1 Therapies: An Emerging Approach for Alcohol Reduction?
Alcohol use disorder (AUD) remains one of the world’s most prevalent yet undertreated conditions, with fewer than 2 % of affected Americans receiving an FDA‑approved medication. Recent randomized trials of once‑weekly GLP‑1 receptor agonists, especially semaglutide, have demonstrated statistically significant reductions...
![[Comment] HPV Vaccine Scale-Up Is Key to Curb Rising Cervical Cancer Inequalities](https://hixhlmpcokxhartfkpyi.supabase.co/storage/v1/object/public/images/thumbnails/d5dae544601ad09d68c3dce84ec5011b.webp)
[Comment] HPV Vaccine Scale-Up Is Key to Curb Rising Cervical Cancer Inequalities
Despite advances in high‑income nations, cervical cancer deaths remain heavily concentrated in low‑ and lower‑middle‑income countries, where screening is scarce. Modeling studies show that scaling up HPV vaccination, especially with single‑dose regimens, could dramatically narrow these gaps. However, political and...

New Genetic Risk Report Reveals Hidden Heart Disease Risk Before Symptoms Appear
A JACC study validated an integrated polygenic risk‑score (PRS) panel for eight cardiovascular conditions using 245,394 All of Us participants and 53,306 Mass General Brigham Biobank members. The report stratifies risk, with the top 10% showing a 41‑fold odds for...

Gentler Treatment Improves Survival in Children with Relapsed Leukemia
A UK‑wide trial (UKALL Rel2020) tested a gentler regimen for children and young adults with relapsed acute lymphoblastic leukemia, using reduced‑intensity chemotherapy followed by the targeted immunotherapy blinatumomab. The study enrolled 188 patients across 25 centres and achieved a 92%...
Leishmanicidal Efficacy of Cold Atmospheric Multiple Plasma Jet Against Leishmania Major in a Murine Model: Effects on Parasite Burden, Cytokine...
The study evaluated a cold atmospheric multiple plasma jet (CAMPJ) as a novel therapy for cutaneous leishmaniasis in BALB/c mice. Topical applications of 5, 10 and 15 minutes, administered twice weekly for three weeks, markedly reduced lesion size and splenic...

FDA, up Close and Personal: A Banker’s Recalibration
Former FDA senior advisor Peter Reikes argues that the agency’s perceived slowness stems from its core principles of independence, caution, and process discipline rather than dysfunction. He notes that despite recent leadership turnover and political pressure, a deep bench of...

The CRISPR Medicine That Cured a Child: How Yuri Milner’s Breakthrough Prize Brought Gene Therapy to Hollywood
At the 2026 Breakthrough Prize ceremony, Baby KJ was celebrated after receiving Casgevy, the first FDA‑approved CRISPR gene‑editing therapy for sickle cell disease and beta‑thalassemia. The $3 million prize honored researchers Stuart Orkin, Swee Lay Thein and others whose work on the BCL11A...

This Organoid Can Menstruate — and Shows How Tissue Can Repair Itself
Researchers at the Friedrich Miescher Institute have engineered 3‑D endometrium organoids that can undergo a full menstrual cycle, shedding and regenerating tissue without scarring. By exposing the organoids to estrogen and progesterone and then withdrawing the hormones, they induced hormonal...

GLP-1s Reduce Heavy Drinking Days in Patients With Obesity, Alcohol Use Disorder
A randomized, double‑blind trial in Copenhagen found that once‑weekly semaglutide 2.4 mg significantly reduced heavy‑drinking days in patients with alcohol use disorder and obesity. Over 26 weeks, the semaglutide group saw a 41.1‑percentage‑point drop in heavy‑drinking days versus 26.4 points for...

Remembering J. Craig Venter: A Relentless Scientist Who Changed Biotech — and Was All Too Easily Misunderstood
J. Craig Venter, the pioneering genomics entrepreneur who died at 79, reshaped biotechnology by accelerating the human genome sequence and building the first synthetic cell. His private‑sector efforts, including Celera and Synthetic Genomics, turned DNA sequencing into an industrial process. Venter’s...
Single-Vesicle Profiling Could Push Liquid Biopsies Toward Everyday Clinical Use
Researchers from Incheon National University and the University of Pennsylvania reviewed cutting‑edge single‑extracellular vesicle (EV) profiling technologies that isolate and analyze vesicles one at a time. The review, published in TrAC Trends in Analytical Chemistry, highlights substrate‑based, droplet‑based and solution‑based...

The BioPharm Brief: Metabolic Phase III Progress, HER2 Oncology Momentum, and US Manufacturing Expansion Drive Industry Scale
Zealand Pharma and Roche are moving petrelintide, an amylin analog, into Phase 3 trials for chronic weight management, with enrollment slated for the second half of 2026 after earlier studies showed double‑digit weight loss and tolerability comparable to placebo. The FDA...
CRISPR Speed Patterns Can Identify Multiple Viruses and Variants Simultaneously
KAIST researchers and partners have unveiled a CRISPR‑Cas13 diagnostic that reads the enzyme's reaction speed to identify multiple viruses and variants in a single test. By encoding kinetic patterns as a barcode, the method distinguishes pathogens without needing separate gene...

Drug Trials Snapshots: BIMZELX
UCB’s BIMZELX, a dual IL‑17A/F monoclonal antibody, received FDA approval on Oct 17 2023 for adults with moderate‑to‑severe plaque psoriasis. The approval was based on two phase‑3 placebo‑controlled trials that enrolled 839 patients across 13 countries, with a demographic profile of 72%...

Drug Trials Snapshots: AGAMREE
Santhera Pharmaceuticals received FDA approval on October 26, 2023 for AGAMREE (vamorolone), a corticosteroid designed to treat Duchenne muscular dystrophy (DMD) in patients aged two years and older. The approval rests on a multinational, double‑blind, 24‑week trial of 121 male patients that...

Drug Trials Snapshot: TALVEY
TALVEY (talquetamab‑tgvs), a bispecific antibody from Janssen Biotech, received FDA approval on August 10 2023 for adult patients with relapsed or refractory multiple myeloma who have exhausted at least four prior therapies. The pivotal trial enrolled 339 participants across 47 sites, with...

Drug Trials Snapshots: OJJAARA
OJJAARA (momelotinib) received FDA approval on September 15 2023 for adults with myelofibrosis‑related anemia. The approval rests on two pivotal trials—MOMENTUM, which enrolled JAK‑inhibitor‑experienced patients, and SIMPLIFY‑1, which enrolled JAK‑naïve patients. In MOMENTUM, 25% of OJJAARA recipients achieved a ≥50% reduction in...

There Are Two GLP-1 Side Effects Your Doctor Doesn’t Know About, and They Can Affect Your Workouts
A new *Nature Health* study used AI to scan 400,000 Reddit posts, finding that roughly 70,000 users were taking GLP‑1 drugs such as Ozempic, Wegovy, or Zepbound. While nausea and fatigue remain the most common side effects, about 4% of...

RNA-Built Droplets Create Customizable Organelles Inside Living Cells
UCLA researchers have engineered programmable artificial organelles by assembling RNA nanostars into droplet‑like condensates inside living cells. The RNA sequences encode assembly instructions, allowing precise control over condensate size, composition, and subcellular location. Published in Nature Nanotechnology, the study demonstrates...

Gene Circuits Reshape DNA Folding and Affect How Genes Are Expressed, Study Finds
MIT researchers published in Science that the physical arrangement of genes—termed "gene syntax"—dramatically reshapes DNA supercoiling and alters transcription. Divergent gene pairs boost expression of both genes, while tandem pairs cause the upstream gene to suppress the downstream one, producing...

Drug Trials Snapshots: ELREXFIO
Pfizer’s ELREXFIO (elranatamab‑bcmm) received accelerated FDA approval on Aug 14 2023 for relapsed/refractory multiple myeloma after at least four prior therapies. The approval rests on a single‑arm trial of 187 patients, with 97 efficacy‑eligible participants showing a 57.7% objective response rate, including...