
Biotech Has a New Startup Model: Small Team, Big Check and Chinese Assets
A wave of biotech startups is emerging that forgos deep‑science platforms in favor of lean teams, mega‑size venture checks and strategic Chinese assets such as patient data, manufacturing capacity, and regulatory pathways. These companies raise $150‑$250 million in Series A or B rounds, often led by global VC firms seeking rapid market entry. By anchoring operations in China, they tap lower R&D costs and faster clinical trial enrollment while maintaining a U.S.‑centric commercial outlook. The model challenges the traditional, science‑first biotech playbook and could reshape funding dynamics across the sector.

UCB to Acquire Maker of Antibody Treatments for Autoimmune Diseases
UCB announced a definitive agreement to acquire a privately held biotech that develops monoclonal antibody treatments for autoimmune diseases. The target company’s portfolio includes candidates for rheumatoid arthritis, lupus and multiple sclerosis, complementing UCB’s existing immunology assets. Financial terms were...
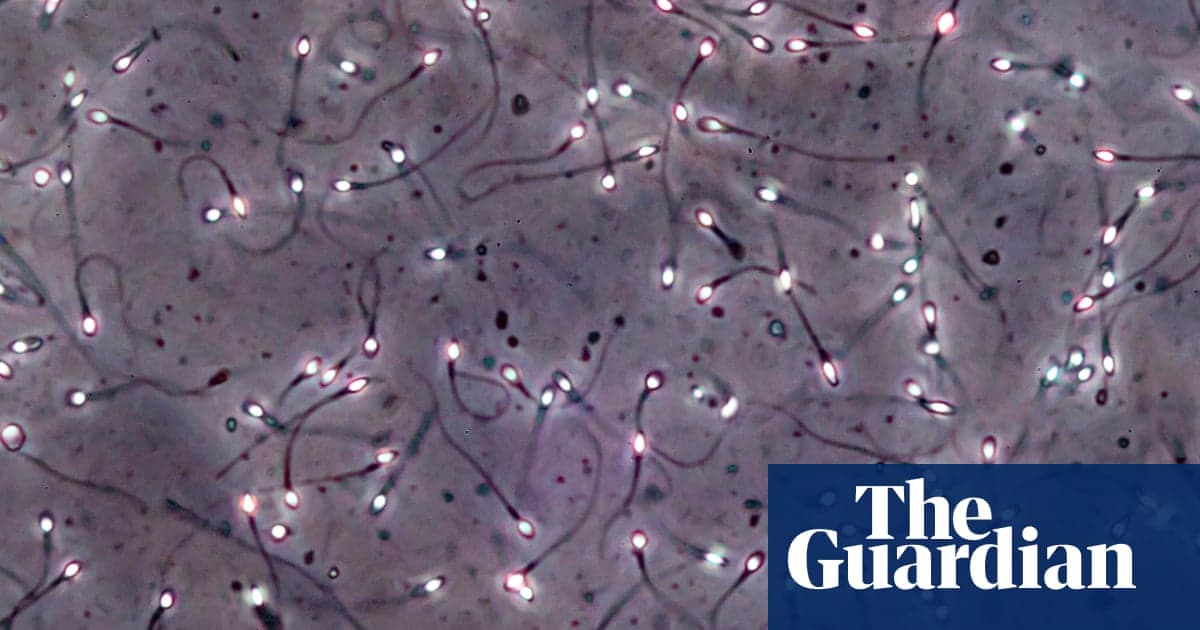
Man Produces Sperm From Testicular Tissue Frozen as a Child in Breakthrough Trial
A 27‑year‑old man has produced mature sperm after his prepubertal testicular tissue, frozen at age 10 before chemotherapy for sickle‑cell disease, was re‑transplanted 16 years later. This is the first documented restoration of sperm production from cryopreserved prepubertal tissue in...

Choosing the Right CDMO for Long-Term Stability
Biotech firms must scrutinize contract development and manufacturing organizations (CDMOs) before committing to multi‑year projects. Dr. Patrick Meyer of Rentschler Biopharma outlines criteria such as transparent communication, accurate timelines, and a proven commercialization record. Technical expertise and a resilient supply chain...
Gene Therapy’s Evidence Problem—Lessons From Recent FDA Decisions
The FDA recently rejected REGENXBIO’s gene‑therapy candidate RGX‑121, citing an unvalidated biomarker as the primary endpoint and reliance on an external natural‑history control. The decision highlights a broader pattern of mixed regulatory outcomes for advanced therapeutics, with approvals like Sarepta’s...
Glowing Nanoparticles Exposed Hidden Cancer-Protein Behavior that Could Reshape Drug Screening
A Broad Institute team led by Sam Peng introduced upconverting nanoparticle probes that remain luminescent for minutes to hours, enabling continuous single‑molecule imaging of cancer‑related receptors in living cells. Using these probes, they captured real‑time dimerization dynamics of EGFR, HER2...

Protein Degraders Gain Speed as Arvinas Scores Landmark Approval
Arvinas received FDA approval for Veppanu, the first PROTAC therapy, marking a milestone for protein degraders. The drug showed modest benefit in a phase 3 trial, with the clearest effect in patients carrying ESR1 mutations, prompting the company to lay off...

Needle-Free Diabetes Care: 6 Devices that Painlessly Monitor Blood Sugar
Needle‑free glucose monitors are moving from research labs to commercial shelves, with six innovative devices highlighted for their non‑invasive approaches. Abbott’s FreeStyle Libre remains the market leader for interstitial sensing, while newcomers such as Occuity Indigo, D‑Pocket, Light Touch Technology,...

PCI Adds US Sterile Fill/Finish Capabilities Through $1B Global Expansion Drive
PCI Pharma Services announced a $1 billion global expansion, allocating $100 million to upgrade its San Diego campus with a high‑speed isolator line that will more than double its prefilled syringe capacity. The company also launched a GMP‑ready isolator vial and lyophilization line...

Regulatory Submissions with Real-World Evidence: Successes, Challenges, and Lessons Learned - 09/23/2025
In a September 23, 2025 speech, FDA Principal Deputy Commissioner Sara Brenner outlined how real‑world evidence (RWE) has been used to support regulatory decisions and announced the agency‑wide FDA‑RWE‑ACCELERATE initiative. She highlighted recent examples from CDER, CBER and CDRH, and introduced Sentinel 3.0,...
Speed 'Training' Prepares Bacteria for Complex Tasks, Like Munching Plastics
Researchers at the National University of Singapore unveiled Lytic Selection and Evolution (LySE), a phage‑based platform that can rapidly evolve large gene clusters up to 40 kb. In a proof‑of‑concept, LySE boosted a five‑gene pathway enabling E. coli to consume ethylene glycol,...

First FDA ODAC Meeting without Pazdur Highlights Conceptual Challenges with Trial
The FDA’s Oncology Drugs Advisory Committee (ODAC) convened for the first time in nine months to review two AstraZeneca cancer therapies. The panel voted 6‑3 against approving camizestrant, an oral SERD for HER2‑negative advanced breast cancer, citing the trial’s failure...

Sonire Therapeutics Initiates First U.S. Clinical Study of Ultrasound-Guided HIFU Therapy for Pancreatic Cancer
Sonire Therapeutics announced the launch of SUNRISE‑II, its first U.S. clinical trial evaluating a proprietary high‑intensity focused ultrasound (HIFU) system for pancreatic cancer. The study will enroll roughly 10 patients to assess safety and feasibility. The inaugural patient was treated...

Celcuity Reports the P-III (VIKTORIA-1) Trial Data on Gedatolisib Combination for HR+/HER2- PIK3CA Mutant Advanced Breast Cancer
Celcuity announced phase‑III VIKTORIA‑1 data showing that gedatolisib combined with fulvestrant, with or without palbociclib, achieved a statistically significant improvement in progression‑free survival (PFS) versus the alpelisib‑fulvestrant standard of care in HR+/HER2‑ advanced breast cancer patients harboring PIK3CA mutations. The...

Summit Shares Descend as PD-1/VEGF Asset Misses Early Survival Mark
Summit Therapeutics announced that its anti‑PD‑1/VEGF bispecific, ivonescimab, failed to meet the interim progression‑free survival threshold in the Phase 3 HARMONi‑3 trial for squamous NSCLC. The miss triggered a 26% plunge in Summit’s share price, closing at $16.12. An independent data...

Drugmaker AbbVie Chooses North Carolina for $1.4B Manufacturing Campus
AbbVie announced a $1.4 billion investment to build a 185‑acre manufacturing campus in Durham, North Carolina, targeting its immunology, neuroscience and oncology portfolios. The site will create 734 jobs over four years, incorporate AI‑driven production tools, and be operational by the...

UCB Captures Candid in $2.2B Autoimmune Deal as Pharma’s M&A Train Chugs Along
UCB announced a $2 billion acquisition of Candid Therapeutics, adding a pipeline of T‑cell engager drugs aimed at autoimmune and inflammatory diseases. The deal also includes up to $200 million in milestone payments and is expected to close by Q3 2026. Candid’s...

How a Vision-Restoring Gene Therapy Proved that We Can Treat Inherited Diseases
Luxturna, the first FDA‑approved gene‑augmenting therapy for inherited retinal disease, received the 2026 Breakthrough Prize after restoring sight to patients with Leber’s congenital amaurosis type 2. Developed by Spark Therapeutics founders Katherine High, Jean Bennett and surgeon Albert Maguire, the treatment...
Obesity Ends Oncology’s Long Reign as Top Contributor to Biopharma Pipeline Value
Deloitte’s 2025 pipeline analysis shows obesity overtaking oncology as the top value‑generating therapeutic area, accounting for 25% of total pipeline worth. Eli Lilly and Novo Nordisk dominate the obesity segment, together holding roughly 96% of its assigned value. GLP‑1/GIP agonists are driving...

OneSource Notches a Second Generic Semaglutide Approval in Canada, Through Partner
Canada approved its second generic version of Ozempic (semaglutide) through Apotex, which partnered with Orbicular Pharmaceutical Technologies and India‑based CDMO OneSource Specialty Pharma. OneSource handled end‑to‑end manufacturing, leveraging its FDA‑approved Bengaluru facility, while Orbicular led product development. The approval follows...
FDA Grants 510(k) Clearance to TaeWoong Medical for Spaxus EUS Stent
TaeWoong Medical received FDA 510(k) clearance for its Niti‑S Spaxus Stent, a fully covered, self‑expanding metallic device used in endoscopic ultrasound (EUS)‑guided drainage. The stent is approved for draining symptomatic pancreatic pseudocysts, walled‑off necrosis larger than 6 cm, and for gallbladder...

What Is Your AI Drug Repurposing Strategy Missing?
The article argues that AI‑driven drug repurposing for oncology often fails because models are fed fragmented, noisy data despite abundant datasets. It stresses that more data alone won’t improve outcomes; instead, high‑quality, curated, structured data—such as knowledge graphs linking genes,...

EU Backs Itvisma Gene Therapy for Spinal Muscular Atrophy
European regulators have issued a positive opinion on Itvisma, Novartis' onasemnogene abeparvovec gene therapy for spinal muscular atrophy (SMA). The Committee for Medicinal Products for Human Use (CHMP) recommended marketing authorisation on 23 April 2026, pending final approval by the European Commission....

Vantrexa Sets High Standards for Retatrutide Research Materials in Metabolic Studies
Vantrexa, a professional laboratory supplier, is now offering research‑grade Retatrutide (LY3437943), a triple‑agonist targeting GLP‑1, GIP and glucagon receptors, for metabolic studies. The company employs advanced solid‑phase peptide synthesis and rigorous in‑house plus third‑party HPLC/LC‑MS testing, achieving purity levels above...

Rhythm Pharmaceuticals Reports the EC Approval of Imcivree (Setmelanotide) for Acquired Hypothalamic Obesity
Rhythm Pharmaceuticals announced that the European Commission has granted marketing authorization for Imcivree (setmelanotide) to treat acquired hypothalamic obesity in patients aged four years and older. The approval is based on the Phase III TRANSCEND trial, which enrolled 120 participants...
Novo Nordisk to Launch Ozempic for Type 2 Diabetes in US
Novo Nordisk will launch an oral version of Ozempic in the United States, following FDA approval that marks the first oral GLP‑1 drug cleared for both blood‑sugar control and cardiovascular risk reduction. The tablets, available in 1.5 mg, 4 mg and 9 mg...

Novel Wheat Hybrids Increase Resistance to Major Fungal Disease by up to 70%
Researchers have transferred a novel resistance locus from the wild weed Elymus repens into wheat, creating hybrids that slash Fusarium head blight (FHB) infection by up to 70%. The new locus, named Fhb.Er‑1StL, showed a 69% reduction in diseased spikelets...

Arvinas and Pfizer Report the US FDA Approval of Veppanu (Vepdegestrant) for ESR1-Mutated Breast Cancer
The U.S. FDA has approved Veppanu (vepdegestrant) for adults with ESR1‑mutated ER+/HER2‑ advanced or metastatic breast cancer who have received at least one line of endocrine therapy. The approval follows the Phase III VERITAC‑2 trial, which compared Veppanu to fulvestrant...

65 % of Eligible Lung Cancer Patients Do Not Receive the Most Appropriate Targeted Therapies, Diaceutics Report Finds
Diaceutics’ 2026 Clinical Practice Gaps report shows that 65% of U.S. patients with advanced non‑small cell lung cancer still miss the most appropriate targeted therapy, a figure unchanged since 2019. While biomarker testing has improved, the biggest loss now occurs...

Artificial Retina Uses Biological Liquid Medium for Direct-to-Display ‘Vision’
Italian researchers led by Prof. Thomas M. Brown unveiled BIOPIX, a bio‑electronic hybrid retina emulator that embeds organic photodetectors in a liquid Ames medium. The proof‑of‑concept includes a 2 × 2 cone‑type array for colour and a 4 × 4 rod‑type array for grayscale,...

Novel Bioprinting Method Lays the Foundation for Personalised Regenerative Medicine
Researchers in Italy unveiled a solid‑electronics, liquid‑electrolyte sensor array that mimics retinal function, marking a proof‑of‑concept for bio‑integrated vision devices. A cell‑free hydrogel delivering extracellular vesicles showed efficacy in repairing intrauterine adhesions and restoring fertility in preclinical studies. An analysis...

Glowing Probe Detects Multiple Antibiotics with Just a Smartphone
Researchers in Italy unveiled a proof‑of‑concept sensor array that blends solid‑state electronics with a liquid electrolyte, effectively mimicking biological vision and allowing a smartphone to detect multiple antibiotics via fluorescence. In parallel, a heat‑resistant polyamide was engineered to emit pure...

FDA Action Alert: Argenx, AstraZeneca/Daiichi Sankyo, Biogen/Eisai and Cingulate
May’s FDA docket is light, but the decisions on four high‑profile drugs could reshape market dynamics. Argenx is seeking to broaden Vyvgart’s label to seronegative myasthenia gravis patients, potentially adding 11,000 new users. AstraZeneca and Daiichi Sankyo aim to secure...

Queensland Funds Research Into Cell-Based Therapies for Traumatic Brain Injury
The Queensland Government has pledged A$5.5 million (about US$3.6 million) over three years to the Cure TBI initiative, a research programme focused on cell‑based therapies for traumatic brain injury. Backed by the National Injury Insurance Scheme, Queensland (NIISQ), the effort will be...
Low-Dose Drug Cuts Breast Density up to 26% with Fewer Side Effects
A Karolinska Institutet study found that low‑dose endoxifen, the active metabolite of tamoxifen, reduces mammographic breast density by up to 26%—comparable to the 18.5% reduction seen with standard 20 mg tamoxifen—while causing far fewer serious side effects. In a randomized, placebo‑controlled...

Krystal Biotech Inc (KRYS) Q1 2026 Earnings Call Transcript
Krystal Biotech reported Q4 2025 net VYJUVEK revenue of $107.1 million, a 34% year‑over‑year increase, pushing cumulative sales since launch past $730 million. The company maintained a 94% gross margin and posted $51.4 million of net income for the quarter, with cash and...

ADC Therapeutics SA (ADCT) Q1 2026 Earnings Call Transcript
ADC Therapeutics reported Q1 2026 product revenue of $15.8 million, a decline from the prior year, while GAAP net loss narrowed to $41 million ($0.30 per share). Operating expenses fell 12.1% on a non‑GAAP basis, driven by lower R&D spend. The company...

Travere Therapeutics Inc (TVTX) Q1 2026 Earnings Call Transcript
Travere Therapeutics reported record commercial performance for FILSPARI, posting $322 million in 2025 sales—a 144% year‑over‑year increase—and a Q4 record of 908 new patient start forms. The company secured a new FDA PDUFA target date of April 13, 2026 for the FSGS indication...

NHS Cancer Jab Could Save Patients Hours in Hospital
NHS England is introducing an injectable form of Keytruda, the blockbuster immunotherapy, that can be given in one to two minutes instead of the traditional hour‑long infusion. About 14,000 cancer patients in England start Keytruda each year, and most are...

StockWatch: Patient Death, Rival’s Patent Challenge Sink Erasca Shares
Erasca (NASDAQ: ERAS) saw its shares plunge 53% after disclosing a patient death linked to its pan‑RAS drug ERAS‑0015 and a patent infringement claim from rival Revolution Medicine. The company reported unconfirmed overall response rates of 62%‑75% in KRAS G12X...
CD44+ Monocytes Drive Inflammation in Preemie Lung Disease
Researchers have identified CD44⁺ monocytes as a key driver of hyperinflammatory responses in bronchopulmonary dysplasia (BPD) among very premature infants. The study shows these cells are markedly elevated in BPD patients and release amplified cytokine storms when exposed to hyperoxic...

Synthetic Biology Promised to Rewrite Life—With the Death of Its Pioneer, J. Craig Venter, How Close Are Scientists?
J. Craig Venter’s 2010 breakthrough—creating a cell controlled by a fully synthetic genome—proved that DNA could be written like software, launching modern synthetic biology. Since then researchers have engineered microbes to produce medicines such as artemisinin, explored bio‑fuel production, and built...

Brain Complexity Enhances Premature Newborns’ Maturity Evaluation
Researchers have demonstrated that measuring brain signal complexity provides a reliable indicator of physiological maturity in premature newborns. Using high‑density EEG and advanced signal‑processing algorithms, the study linked specific complexity patterns to gestational age and future neurodevelopmental trajectories. The approach...
Axon Pathways Connect Small Gestational Age to Lung Restrictions
A 2026 Nature Communications study reveals that axon guidance pathways, traditionally linked to neural development, mediate the relationship between small for gestational age (SGA) birth and later spirometric restriction. Researchers analyzed genomic, transcriptomic and longitudinal lung‑function data from diverse cohorts...

Liquid Biopsy Predicts Response to Breast Cancer Immunotherapy
Researchers at Vanderbilt-Ingram Cancer Center demonstrated that serial liquid biopsies analyzing peripheral blood RNA can predict response to pembrolizumab in high‑risk early‑stage HER2‑negative breast cancer. The study examined 546 blood samples from 160 patients in the I‑SPY2 trial, showing transcriptional...

Spaceflight Is Hard on the Heart, yet Artificial Ones Grow Better in Space than on Earth
Researchers demonstrated that miniature human hearts grown from stem cells mature faster in microgravity than on Earth. The International Society for Heart and Lung Transplantation meeting highlighted data from ISS experiments showing a significant boost in organoid production without the...

The Biosimilar Market Isn’t Failing, It’s Finding Its Purpose
The biosimilar market is not collapsing; it is transitioning from a price‑driven scramble to a purpose‑focused strategy. Recent HHS data show that markets become uneconomic once five competitors vie for the same molecule, prompting a shift toward diversified product portfolios....
Blocking a Cellular Inflammation Process Could Result in Effective Therapy for Pancreatic Cancer
Scientists at The Wistar Institute and ChristianaCare identified a vulnerability in pancreatic cancer where defective mitochondria release double‑stranded RNA, triggering the TLR3/TRAF6 inflammatory pathway. The tumor cells become dependent on this inflammation for growth and survival, and blocking the pathway...

Stopping and Restarting Certain GLP-1s to Lose Weight May Make the Drug Less Effective
A preclinical study from the University of Pennsylvania found that stopping and restarting GLP‑1 weight‑loss drugs, such as semaglutide, markedly diminishes their efficacy. Overweight mice on a stop‑and‑start regimen regained weight during off periods and never recaptured their initial loss,...

The Bias in Medical Research: Africa Carries a Huge Disease Burden but Is Missing From Clinical Trials
A new analysis of 2,472 randomized controlled trials published between 2019 and 2024 reveals a stark under‑representation of Africa in top medical research. Only 3.9% of trials in the most prestigious general journals were conducted exclusively on the continent, and...