
Truist Reaffirms Buy on Eli Lilly (LLY) After Trial Success
Truist Securities reaffirmed its Buy rating on Eli Lilly, keeping a $1,281 price target after the company reported positive Phase 3 data for its insulin candidate Foundayo. The ACHIEVE‑4 trial involving over 2,700 type 2 diabetes patients demonstrated non‑inferiority to insulin glargine and a 57% reduction in all‑cause mortality, with no liver safety signals across the broader Phase 3 program. Lilly plans to submit a U.S. NDA for Foundayo by the second quarter of 2026, though the FDA has asked for additional post‑marketing studies. The results bolster Lilly’s diabetes franchise and support the analyst’s optimistic outlook.

Scientists Cautiously Suggest GLP-1s Are Safe to Use Around Pregnancy
A systematic review of more than 49,000 pregnancies over two decades found that exposure to GLP‑1 receptor agonists such as Ozempic, Wegovy and Mounjaro around conception does not increase the risk of major birth defects. The meta‑analysis of ten cohort...
New Test Promises to Detect Cancer Earlier, From Tiny Particles in Bodily Fluids
Researchers at the University of Calgary have unveiled EXOSense, a patent‑pending platform that electrically isolates small extracellular vesicles from blood or urine for cancer screening. These vesicles carry molecular signatures that appear long before conventional biomarkers, offering a potential route...
Development Analysis and Strategic Insights From a 505(b)(2) Reformulation Product: Enzalutamide
Astellas used the 505(b)(2) pathway to reformulate enzalutamide (XTANDI®) from a 40 mg lipid‑filled soft capsule into 40 mg and 80 mg film‑coated tablets, aiming to cut pill burden. Five biopharmaceutic studies demonstrated comparable AUC between the two dosage forms, while the tablet...

Effect of Virtual Reality on Acute Stress Response and Discomfort During Vacuum-Assisted Closure (VAC) Dressing Changes: A Protocol for Randomized...
A randomized controlled trial will evaluate immersive virtual reality (VR) as a non‑pharmacologic method to lessen acute stress during vacuum‑assisted closure (VAC) dressing changes. Participants are split 1:1 between VR and standard care, with primary endpoints including heart rate, blood...

'Build AI that Can Accurately Represent the Full Complexity of Biology': Mark Zuckerberg Wants to Cure All Diseases but Needs...
Meta billionaire Mark Zuckerberg is channeling $500 million into Biohub’s Virtual Biology Initiative to amass massive cellular datasets for AI modeling. The funding splits into $100 million for global data collection and $400 million for advanced imaging and engineering tools. Partnerships with the...
Human Organ Chip Systems Reshape Drug Development
Harvard’s Wyss Institute, led by Dr. Donald Ingber, has spent over a decade perfecting Human Organ Chip systems that mimic organ-level functions in a thumb‑drive‑sized device. Recent FDA and NIH policy shifts endorse these chips as viable alternatives to animal...

Could Ozempic Help With Alzheimer’s Disease? Scientists Are Taking a Closer Look
A new Anglia Ruskin University review of 30 preclinical studies suggests GLP‑1 drugs such as Ozempic and Wegovy can lower amyloid‑beta and tau, the hallmark proteins of Alzheimer’s disease. Twenty‑two studies reported reduced amyloid‑beta and nineteen showed decreased tau, with liraglutide...
Boosting One Protein Helps the Brain Fight Alzheimer’s
Researchers at Baylor College of Medicine discovered that increasing the protein Sox9 in astrocytes enables mouse models of Alzheimer’s disease to clear existing amyloid plaques and retain memory performance. The study, published in Nature Neuroscience, showed that elevated Sox9 enhances...

Your Next Dog May Live Longer
Longevity biotech Loyal, founded by Celine Halioua, has received FDA clearance to market a daily pill that improves insulin sensitivity and could extend dogs’ lifespans. The agency deemed the drug likely effective based on a small study of about 50...
Mcu Controls Bone Growth Through Mitochondrial Calcium
Researchers have identified the mitochondrial calcium uniporter (Mcu) as a key regulator of bone formation, showing that its activity controls mitochondrial calcium uptake and directs mesenchymal stem cells toward the osteoblast lineage. Genetic loss of Mcu in mouse models reduced...

The Company that Built TikTok’s Algorithm Is Now Designing Drugs for Diseases Pharma Called Undruggable
ByteDance’s Anew Labs showcased its first AI‑designed therapy, a small‑molecule inhibitor of IL‑17, at the American Association of Immunologists meeting in Boston. The molecule targets a protein‑protein interaction long deemed undruggable, suggesting generative AI can breach a major pharmaceutical barrier....
Precise Spatiotemporal Cardiac Repair and Regeneration
Researchers are advancing spatiotemporal drug delivery systems (DDS) that synchronize therapeutic release with the heart’s natural healing stages after myocardial infarction. These platforms integrate bioelectrical scaffolds, programmable degradation, and cell‑free vesicle carriers to provide phase‑specific immunomodulation, angiogenesis, and antifibrotic treatment....

FDA Approves Once-Daily Idvynso Tablet for Treating HIV
The FDA has approved Merck’s Idvynso, a once‑daily, two‑drug tablet combining doravirine and islatravir, for HIV‑1‑infected adults who are virologically suppressed. The regimen replaces existing antiretroviral therapy and is tenofovir‑free, targeting patients without prior treatment failure. Approval rests on two...

Dietary Fats Shape Pancreatic Cancer Risk via Ferroptosis
Researchers at Yale School of Medicine published a study in Cancer Discovery showing that the type of dietary fat, not just total fat, influences pancreatic ductal adenocarcinoma development in mice. Diets high in oleic acid, a monounsaturated fat common in...
Early Detection of Keratoconus Enhanced by Light Polarization and AI
Researchers at Seoul National University have unveiled a portable eye‑screening system that combines polarization‑sensitive imaging with a deep‑learning algorithm to spot keratoconus at its earliest stages. The device captures subtle birefringence patterns in the cornea, which the AI model classifies...
Innovative Nanoparticle Technique Advances Early Detection of Pancreatic Cancer
Researchers have unveiled a novel nanoparticle‑based imaging agent that markedly improves early detection of pancreatic cancer. In pre‑clinical trials the probe identified tumors as small as 2 mm, delivering a 30% sensitivity boost over conventional CT scans. The technology earned FDA...

The Biopharm Brief: Microbiome Fast Track, Leo Pharma Gene Therapy Deal, AAV Automation Advances
The FDA has awarded Fast Track designation to a live biotherapeutic aimed at restoring gut microbiota in moderate‑to‑severe ulcerative colitis, accelerating its development path. Leo Pharma is buying Replay’s gene‑therapy program to broaden its rare‑dermatology portfolio, marking a shift toward...

Artificial Intelligence for Drug Development
The FDA’s Center for Drug Evaluation and Research (CDER) is formalizing its AI strategy through a draft 2025 guidance, an AI Council created in 2024, and extensive stakeholder engagement. CDER has reviewed more than 500 AI‑enabled drug submissions since 2016...

External Engagements with FDA for Artificial Intelligence in Drug Development
The FDA has published a comprehensive menu of early‑engagement options for sponsors leveraging artificial intelligence across the drug development lifecycle. Sponsors can request formal meetings for IND or pre‑IND programs, or use targeted programs such as C3TI for trial design,...

Arvinas’ ‘Protac’ Breast Cancer Drug Cleared by FDA
The FDA approved Arvinas and Pfizer’s Veppanu, a PROTAC drug, as a second‑line therapy for metastatic ER‑positive, HER2‑negative breast cancer with ESR1 mutations. Veppanu is the first protein‑degrading medicine cleared in the U.S., targeting estrogen receptors for destruction. While the...

UniQure Pushes Forward As FDA Rare Disease Controversies Continue
UniQure, the gene‑therapy pioneer at the heart of FDA Commissioner Marty Makary’s rare‑disease controversy, is pressing forward with a formal request for its Huntington’s disease treatment. The company will present data at a Type B meeting with the FDA in the...

The World Wants to Eliminate Cervical Cancer - How Australian Scientists Led the Way
Australian scientists pioneered the Gardasil HPV vaccine, enabling the world’s first national vaccination programme in 2007 and positioning Australia to eliminate cervical cancer by 2035. The combined strategy of >80% vaccination of adolescents, HPV‑based screening every five years, and self‑sampling...
DNA-Containing Extracellular Vesicles Boost Antitumor Responses in Mice
Researchers at Weill Cornell Medicine discovered that extracellular vesicles released by activated T cells contain DNA that can be transferred to dendritic and tumor cells, enhancing antigen processing and presentation. In mouse models of glioblastoma, pancreatic and triple‑negative breast cancer,...

It’s Time to Take Genetic Testing Off the Pedestal
Genomic testing, once a specialist‑only tool, is now technologically mature and affordable, yet it remains underused in routine care. Advances in sequencing speed, AI‑driven interpretation, and large data sets have removed most technical barriers. Patients are increasingly seeking molecular insights,...

The Next Alzheimer’s Breakthrough Will Take More Than Just Science
Alzheimer’s research has moved from theory to treatment as anti‑amyloid antibodies like Lecanemab and Donanemab receive regulatory approval and begin reaching patients. These drugs can clear existing amyloid plaques and modestly slow cognitive decline, extending the disease trajectory from roughly...
Restoring Protein Recycling Reverses T-Cell Exhaustion in Mice
Scientists at UC San Diego discovered that impaired protein recycling drives T‑cell exhaustion in mice. Restoring the activity of specific E3 ligases—NEURL3, RNF149, and WSB1—reestablished proteostasis, cleared misfolded proteins, and revived T‑cell anti‑tumor function. The findings, published in Cell, suggest...

Moderna in Talks with FDA over Phase 4 Covid Vaccine Data
Moderna is actively collaborating with the U.S. Food and Drug Administration to submit Phase 4 post‑marketing data on its COVID‑19 vaccines. The company hopes the additional safety and efficacy evidence will persuade regulators to broaden the current, narrowed product labels that...

AI, Gene Therapies Drive Market Trends in Eye Care
In 2026 the ophthalmology market is being reshaped by gene‑therapy breakthroughs and the emergence of agentic AI, according to Boston Consulting Group’s Long Sha. Gene‑based treatments are moving beyond rare retinal disorders into chronic conditions such as wet age‑related macular...
Common Cholesterol Medications Do Not Alter Long-Term Dementia Risk
A massive target‑trial emulation study of more than 320,000 older adults found that statin use does not change long‑term risk of dementia. While statin users showed a 46% spike in dementia diagnoses during the first year after initiation, researchers attribute...

Ensorcell Debuts VersaWeld Platform, Wins Best New Product at INTERPHEX 2026
Ensorcell introduced the VersaWeld sterile tube welding platform at INTERPHEX 2026, marking its entry into cell and gene therapy (CGT) manufacturing equipment. The system automates thermoplastic tubing welding in closed‑system environments, delivering consistent weld integrity across multiple tube sizes and materials...

Amgen Launches Late-Stage Obesity Trial in Patients Who Switch From Rival Drugs
Amgen is initiating three Phase III trials for its long‑acting obesity injection MariTide, including a pivotal study that enrolls about 1,200 patients switching from Eli Lilly’s semaglutide or Novo Nordisk’s tirzepatide. The primary goal is a minimum 10% body‑weight loss after 68 weeks,...

F.D.A. Grants Early Access to Promising Drug for Pancreatic Cancer
On May 1, the FDA granted expanded‑access permission for daraxonrasib, an experimental oral drug from Revolution Medicines, allowing patients with previously treated metastatic pancreatic cancer to obtain the therapy outside clinical trials. The drug, taken as three pills daily, has produced...

Regulatory Actions for May 1, 2026
BioWorld released a comprehensive “Regulatory Actions for May 1, 2026” roundup, collating FDA, EMA and other global regulator updates across biopharma, medical technology, and policy domains. The collection links to data snapshots, infographics on digital analysis, and special reports covering topics such...

FDA Permits Expanded Access for Investigational Pancreatic Cancer Drug
The FDA issued a “safe to proceed” letter to Revolution Medicines, enabling an expanded access protocol for its experimental pancreatic cancer drug daraxonrasib. The request, received on April 28 and signed on April 30, targets patients with previously treated metastatic pancreatic ductal...

Redo TAVR: Supra-Annular, Intra-Annular Valves Linked to Comparable Outcomes
A study of 172 redo transcatheter aortic valve replacement (TAVR‑in‑TAVR) procedures from the international PANDORA registry shows comparable one‑year outcomes regardless of whether the initial and second valves are supra‑annular or intra‑annular. The median interval between the index and redo...

Acelyrin Founder Shao-Lee Lin Emerges at Cue, as It Becomes Latest to License From China
Cue Biopharma announced a strategic reboot, appointing Acelyrin founder Shao‑Lee Lin to its executive team and unveiling a new allergy drug candidate sourced from research labs in Taiwan and mainland China. The Boston‑based company, previously focused on T‑cell immunotherapies, will...

Microbiome-Based Therapy Gains FDA Fast Track in Ulcerative Colitis
Belgium‑based MRM Health announced that its investigational microbiome‑based therapy MH002 has been granted FDA fast track designation for mild‑to‑moderate ulcerative colitis. MH002 is a live biotherapeutic composed of six defined commensal bacterial strains designed to restore gut microbial balance and...

Policy Watch: FDA Looks to Expand Real-Time Drug Clinical Trials
The FDA has launched an AI‑driven pilot that streams clinical‑trial data in real time for AstraZeneca and Amgen’s lymphoma and lung‑cancer studies, and is now seeking public input to broaden the approach. The Ninth Circuit Court declined to compel the...
Gut Microbe’s Sulfated Bile Acid Eases Pediatric Sepsis
Researchers identified deoxycholic acid 3‑sulfate (DCA‑3S) as a gut‑derived metabolite that mitigates pediatric sepsis. Metabolomic and metagenomic analyses revealed Enterococcus raffinosus as the primary producer, accounting for over 80 % of DCA‑3S synthesis. In mouse models, DCA‑3S restored intestinal barrier integrity and dampened...
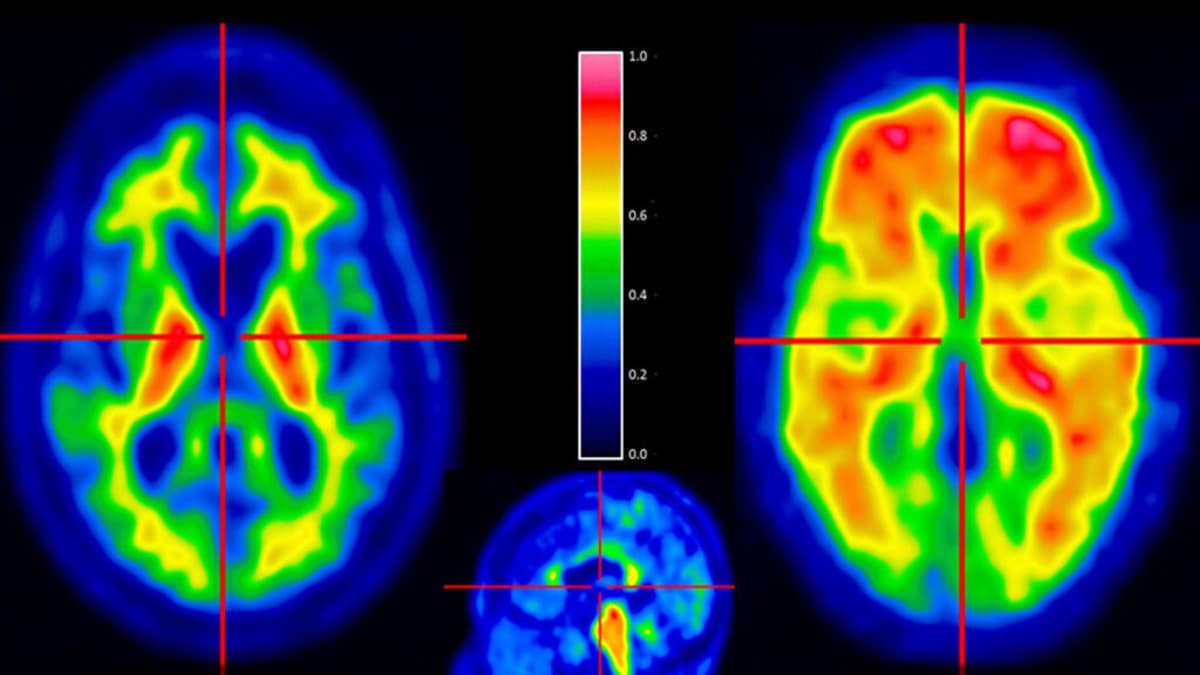
STAT+: Axsome Wins FDA Nod for Alzheimer’s Agitation
Axsome Therapeutics announced that the U.S. Food and Drug Administration has granted a regulatory nod for its investigational therapy aimed at treating agitation in Alzheimer’s disease. The agency’s decision clears the path for accelerated clinical development, potentially moving the drug...

Novel Pulsed Field Ablation Technology ‘Works’
A first‑in‑human trial of Pulse Biosciences' nanosecond pulsed‑field ablation (CellFX nsPFA 360) treated 177 patients with paroxysmal atrial fibrillation. The catheter delivered >10,000 V nanosecond pulses, achieving 100% acute lesion success and 91% durability at 2‑3 months. At one year, 89.7% of patients remained free...
Peptides Are Unproven as Health Aids. FDA May Unleash Them Anyway
The FDA is poised to broaden access to injectable peptides by allowing compounding pharmacies to produce them and by considering their inclusion in oral dietary supplements. Health and Human Services Secretary Robert F. Kennedy Jr. has publicly opposed what he...

Drug Trials Snapshot: TRYNGOLZA
TRYNGOLZA (olezarsen), an APOC‑III‑directed antisense oligonucleotide, received FDA approval on December 19, 2024 for adults with familial chylomicronemia syndrome (FCS). In the pivotal Phase 3 trial (NCT04568434), 66 patients from 11 countries received monthly 80 mg injections or placebo for 53 weeks. The drug achieved...
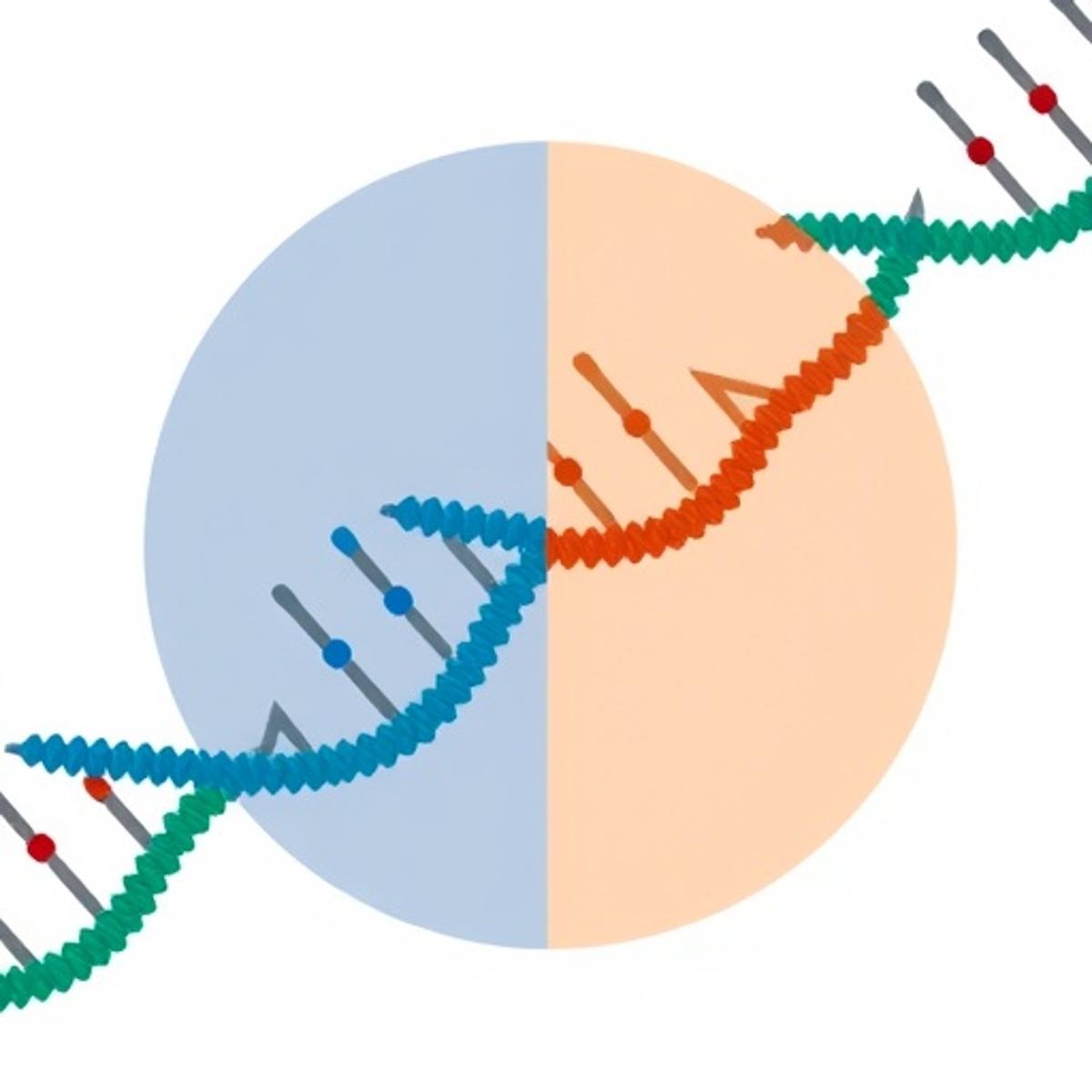
Programmable RNA Targeting via DNA-Guided CRISPR-Cas12a
A team of molecular biologists has reengineered the CRISPR‑Cas12a nuclease to cleave RNA using a DNA guide, creating a programmable RNA‑targeting platform. The DNA‑guided Cas12a system achieved up to 90% knockdown of endogenous transcripts in human cell lines and functioned...

Drug Trials Snapshot: ALYFTREK
Vertex Pharmaceuticals received FDA approval for ALYFTREK, a triple‑combination CFTR modulator, on Dec. 20, 2024. The drug targets cystic fibrosis patients aged six and older with at least one F508del or other responsive mutation. Approval was based on two 52‑week, non‑inferiority trials...

Drug Trials Snapshot: ROMVIMZA
Deciphera's ROMVIMZA (vimseltinib) received FDA approval on Feb 17 2025 for adult tenosynovial giant‑cell tumor (TGCT) when surgery is unsuitable. Approval was based on a global phase II trial of 123 patients, showing a 40% overall response rate versus 0% with placebo...

Summit Slips on Ivonescimab's Apparent Interim Miss in Sign of Investor Frustration
Summit Therapeutics’ shares plunged almost 20% after an interim analysis of its Phase 3 trial of ivonescimab showed a statistical shortfall in a key efficacy endpoint. The miss was limited to a subgroup analysis, but investors interpreted it as a signal...

Amgen Files Update to Tavneos Label as FDA Escalates Push to Withdraw
Amgen filed a supplemental application to revise the prescribing information for its rare‑disease drug Tavneos (avacopan) as the FDA intensifies its effort to withdraw the product. The label update seeks to add new safety warnings and monitoring requirements after the...

Amgen Positions MariTide as Potential ‘Best Monthly’ Obesity Drug
Amgen is positioning its bispecific antibody‑peptide MariTide as the premier monthly or less‑frequent obesity treatment. The company launched a Phase 3 MARITIME‑Switch study to evaluate switching patients from weekly GLP‑1 injections to four‑to‑six monthly doses. Phase 2 data showed up to a...