
Ideaya’s Uveal Melanoma Drug Exceeds Success Benchmark in Late-Stage Trial
Ideaya BioSciences reported that its late‑stage drug for uveal melanoma met its pre‑specified success benchmark, achieving a 27% overall response rate versus the 20% target. The trial also demonstrated a manageable safety profile, reinforcing confidence in the therapy’s risk‑benefit balance. With these data, Ideaya plans to submit an accelerated filing to the U.S. FDA later this year. The result positions the company to potentially become the first to market a dedicated treatment for metastatic uveal melanoma.

Structural Elucidation and Antidiabetic Activity of Polysaccharides From the Parasitic Plant Orobanche Cumana
Researchers isolated three polysaccharide fractions from the parasitic plant Orobanche cumana and identified the alkaline‑extracted fraction OCP‑3 as a low‑molecular‑weight rhamnogalacturonan‑I‑rich polysaccharide. OCP‑3 showed strong antioxidant activity and inhibited key carbohydrate‑digesting enzymes, with IC₅₀ values of 98.5 µg mL⁻¹ for α‑amylase and...

BIO Coffee Chat Explores Venture Philanthropy
The Biotechnology Innovation Organization (BIO) hosted a Patient Advocacy Coffee Chat highlighting the growing influence of venture philanthropy in biotech. Patient groups are now acting as investors, using capital and disease expertise to de‑risk early‑stage programs and guide trial design....

This Method to Reverse Cellular Aging Is About to Be Tested in Humans
Researchers at the Whitehead Institute have engineered a three‑gene cocktail that partially reprograms aged retinal nerve cells, reversing age‑related damage in mouse eyes. The breakthrough underpins Life Biosciences' first human clinical trial, which will deliver the Yamanaka factors—minus the oncogenic...

A New Wave of Immunotherapy Is Eliminating Cancers
Immunotherapy, especially checkpoint inhibitors like dostarlimab, is delivering unprecedented tumor regressions, with recent trials reporting complete remission in 84% of participants. The approach offers non‑surgical, low‑toxicity alternatives, as illustrated by patients such as Maureen Sideris whose esophageal cancer vanished after...
Telix and Regeneron Sign $4.3bn Deal to Co-Develop Radiopharmaceutical Therapies
Telix and Regeneron Pharmaceuticals have signed a partnership valued at up to $4.3 billion to co‑develop radiopharmaceutical therapies for solid tumours. Regeneron will pay $40 million upfront for access to Telix’s manufacturing platform and can expand to four additional programmes, while Telix...
ViraHInter: A Dual-Modal Artificial Intelligence Framework for Predicting Virus-Host Interactions
ViraHInter is a dual‑modal deep‑learning framework that predicts virus‑host protein interactions by combining structure‑generation and sequence‑embedding branches. The system outperforms leading models such as RoseTTAFold2‑PPI and AlphaFold 3 on pathogenic coronaviruses and influenza A viruses, even under severe class imbalance. It uncovered...

FDA Grants Accelerated Approval to Tividenofusp Alfa for Neurologic Hunter Syndrome
The U.S. Food and Drug Administration granted accelerated approval to tividenofusp alfa, sold as Avlayah, as the first enzyme‑replacement therapy that crosses the blood‑brain barrier for neurologic Hunter syndrome. The drug combines iduronate‑2‑sulfatase with a transport protein to reach both...

Connected Medical Devices: Smarter Care Starts Here
Connected medical devices (CMD) are becoming essential for gathering real‑world patient data and supporting decentralized clinical trials. Their integration—from wearable biosensors to continuous glucose monitors—requires dedicated service lines that manage calibration, storage, and data flow. Companies such as Marken are...

Predictive Analytics in Pharma Turns Lab Data Into Launch Strategies
Pharmaceutical firms pour over $300 billion into R&D each year, yet only about 12% of clinical‑trial candidates secure FDA approval. As therapies become more precise, identifying eligible patients and the physicians who treat them grows increasingly complex. Quest Diagnostics highlights that...

Rethinking Dermatology Trial Design for Late-Stage Success
Advances in immunology have spurred many new dermatology therapies, but late‑stage trial failures often stem from outdated trial designs. Traditional short‑term efficacy endpoints like PASI or EASI miss critical data on durability, patient‑reported outcomes, and long‑term safety. Experts advocate incorporating...

Eco-Friendly Synthesis and Characterization of Eggshell-Derived Calcium-Deficiency Bone-Like Hydroxyapatite
The study introduces a two‑step thermal method that leverages the intrinsic pH 10 of calcined eggshell powder to synthesize phase‑pure calcium‑deficiency hydroxyapatite (CDHA) without added NaOH, KOH, or phosphoric acid. First, eggshells are calcined at 900 °C to form CaO; then the...

Regeneron and Sanofi Report the EC Approval of Dupixent (Dupilumab) for Chronic Spontaneous Urticaria (CSU)
Regeneron and Sanofi announced that the European Commission has approved Dupixent (dupilumab) for moderate‑to‑severe chronic spontaneous urticaria in children aged 2‑11 who remain symptomatic despite antihistamines. The decision follows two Phase‑III studies—LIBERTY‑CUPID and CUPIDKids—that demonstrated significant reductions in UAS7 scores...
MGI Tech Celebrates 10 Years of Innovation, Empowering 3,560 Users Across Six Continents
MGI Tech marks its 10th anniversary, now supporting 3,560 users and 5,300 installations across six continents. The company’s flagship T20×2 platform has driven whole‑genome sequencing costs below $100 per genome, while newer T1+ and T7+ systems deliver terabyte‑scale data in...

Boehringer Ingelheim Introduces LENZELTA®: A New Vaccine Advancing Mastitis Prevention in Dairy Cows
Boehringer Ingelheim has launched LENZELTA®, a new mastitis vaccine for dairy cows that requires two doses during the dry‑off period. The oil‑free formulation targets Staphylococcus aureus and Escherichia coli, delivering the earliest onset of immunity and protection lasting up to...

Miracell Reports US FDA 510(k) Clearance for SMART M-CELL PRP and Bone Marrow Concentration Systems
Miracell announced that the U.S. Food and Drug Administration granted 510(k) clearance for its SMART M-CELL PRP Concentration System and Bone Marrow Concentration System, including the related kits. The clearance was based on substantial equivalence to the previously cleared SmartPReP...

A Pioneer Compilation on Ibrutinib-Loaded Hybrid Nanoformulations for Different Types of Cancer
Researchers Pandey, Gautam, and Singh review hybrid nanoformulations that encapsulate ibrutinib, a BTK inhibitor used for chronic lymphocytic leukemia and mantle‑cell lymphoma. The paper details how polymeric nanoparticles, liposomes, dendrimers and SNEDDS improve ibrutinib’s solubility, bioavailability, and pharmacokinetics while reducing...

Profile of Helen M. Piwnica-Worms
Helen M. Piwnica‑Worms, a veteran cancer biologist, has spent decades dissecting how tumor cells bypass cell‑cycle checkpoints and develop drug resistance. Her early work clarified the role of tyrosine phosphorylation in src and cdc2 regulation, laying groundwork for modern checkpoint...

EU Asked to Include Microbial Proteins & Fermentation in Upcoming Biotech Act
Climate advocacy group WePlanet is urging the European Union to explicitly include advanced fermentation—single‑cell proteins, mycelium and precision‑fermentation products—in the upcoming Biotech Act II, slated for Q3 2026. The brief argues that Europe’s food system is overly dependent on imported soy and...

FDA’s CRL Transparency Policy Is Boosting Biopharma Accountability
More than a year after the FDA launched its radical transparency agenda, the agency has released over 200 complete response letters (CRLs) covering 2020‑2024 and added historic letters back to 2002. Public availability forces biotech firms to align their disclosures...

Weight-Loss Drugs and Mars Bars: Novo Nordisk’s Comeback Bid
Novo Nordisk is launching a next‑generation obesity drug to revive growth after a slowdown in its diabetes franchise. The company projects $5 billion in sales from the new semaglutide‑based therapy by 2028, targeting a global obesity market estimated at $250 billion. To...

Your Scale, Your Terms: How Modular Bioreactors Are Redefining Capacity & Manufacturing Strategies
AGC Biologics is championing a scale‑out strategy that uses modular single‑use bioreactors instead of traditional stainless‑steel scale‑up. Its proprietary 6Pack System™ links up to six 2,000‑liter disposable reactors, delivering flexible capacities from 2 kL to 12 kL while preserving process parameters. The approach...

Weizmannia Coagulans BC99 Presents Promising Probiotic Strategy for Chronic Constipation
A double‑blind, placebo‑controlled trial of 88 adults showed that daily intake of Weizmannia coagulans BC99 (10 billion CFU) for eight weeks markedly improved bowel‑movement frequency, stool form and psychological well‑being. Participants receiving BC99 experienced faster colonic transit, higher levels of motility‑promoting peptides and...

Stanford Scientists Discover “Natural Ozempic” Without Side Effects
Stanford Medicine scientists have identified a naturally occurring 12‑amino‑acid peptide, dubbed BRP, that mimics the appetite‑suppressing effects of semaglutide (Ozempide) in animal models. In lean mice and minipigs, a single injection cut food intake by up to 50% and daily...

Pleiotropic Modulation of the Gut-Brain-Lung Axis by Ketamine and Its Enantiomers
A new review examines how ketamine and its enantiomers reshape the gut‑brain‑lung axis by modulating microbiota, microbial metabolites, and immune‑cell trafficking. Both arketamine (R‑ketamine) and esketamine (S‑ketamine) reduce systemic inflammation, but they differ mechanistically: arketamine leverages vagus‑mediated gut‑brain signaling, while...

Singapore-Listed IX Biopharma Bets Big on Non-Opioid Pain Relief
Singapore-listed IX Biopharma has advanced its sublingual ketamine wafer, Wafermine, through Phase 2 trials and secured a $40.95 million contract from the U.S. Department of Defense to fund Phase 3 development and an Emergency Use Authorization. The FDA has accepted the Phase 2 data,...

Bladder Toxicity Risk Appears Low for Psychiatric Ketamine Patients, Though Data Is Limited
A systematic review of 27 clinical studies found that short‑term ketamine and esketamine treatments for psychiatric disorders do not significantly increase bladder or urinary tract toxicity compared with placebo. Reported urinary symptoms ranged from 0 % to 25 % and were generally...

CDC Caught Burying Report on Real Effects of COVID Vaccine
A CDC report led by acting director Jay Bhattacharya found that COVID‑19 vaccines cut urgent‑care visits by 50% and hospitalizations by 55% for healthy adults. The study, slated for the Morbidity and Mortality Weekly Report on March 19, was delayed over...

StockWatch: IPO Market Shows Sign of Life with Avalyn Filing
Avalyn Pharma, a Boston‑based biotech, filed an S‑1 on Wednesday seeking up to $100 million to advance its inhaled antifibrotic pipeline for pulmonary fibrosis. The filing marks the first biotech IPO since Generate: Biomedicines raised $400 million and revives a market that...

As RSV Evolves, a Two‑pronged Antibody Cocktail Aims to Stay Ahead
Chinese researchers at Xiamen University have engineered a two‑antibody cocktail, 1A2 and 1B6, that targets separate, conserved regions of the RSV fusion protein. Preclinical tests in mice and cotton rats showed the combo neutralized both RSV A and B subtypes...

Two New Takes on Making a Type of Targeted Cancer Therapy Even Better
Two biotech startups announced fresh capital to boost next‑generation antibody‑drug conjugates (ADCs). Sidewinder Therapeutics raised a $137 million Series B, bringing total funding to $162 million, to develop bispecific ADCs that bind a tumor‑driving receptor and an internalizing receptor, aiming for tighter cancer...
Immune Cells in the Nose Slow Influenza Virus, Study Finds
A University of Gothenburg study reveals that CD4 memory T cells linger in the nasal lining and can quickly reactivate when influenza re‑enters the body, curbing viral replication. In mouse models these resident cells lowered viral loads and limited tissue...

Re: The Power of the Markets: The Scandal that Keeps on Taking
A letter to the BMJ criticizes the pharmaceutical industry’s reliance on experimental trials that deny patients post‑trial access to new drugs. It argues that powerful, profit‑driven groups manipulate regulations, limiting transparency and compromising the NHS’s ability to provide affordable treatments....

Python Blood Could Be the Key to Weight Loss with Zero Side Effects According to New Study
Researchers from Colorado, Stanford and Baylor identified a metabolite, para‑tyramine‑O‑sulfate (pTOS), that spikes a thousand‑fold in python blood after a large meal. When administered to mice, high doses of synthetic pTOS triggered weight loss without nausea or reduced energy. The...

AI Can Design and Run Thousands of Lab Experiments without Human Hands. Humanity Isn't Ready
In February 2026 OpenAI and Ginkgo Bioworks reported that GPT‑5 autonomously designed and ran 36,000 biological experiments through a robotic cloud laboratory, slashing protein‑production costs by about 40%. The AI‑driven loop—design, build, test, learn—turns biology into an engineering discipline, enabling...

GSK Sees Blockbuster Potential in Targeted Cancer Therapy After Promising Early Data
GSK’s experimental targeted therapy Mo‑rez showed early signs of efficacy, shrinking tumors in a majority of patients with hard‑to‑treat cancers. In a trial, 62% of platinum‑resistant ovarian cancer patients and 67% of endometrial cancer patients achieved at least a 30%...

GSK Plans Five Phase 3 Studies for Gynecological Cancer ADC From Hansoh
GlaxoSmithKline (GSK) announced it will initiate five Phase 3 clinical trials to evaluate an antibody‑drug conjugate (ADC) for gynecological cancers, a candidate licensed from Chinese partner Hansoh Pharma. Early-stage data presented on Sunday showed encouraging tumor responses in ovarian and endometrial...

Why Ozempic Doesn’t Work for Everyone: Scientists Just Found a Hidden Reason
Researchers at Stanford Medicine and international partners identified a genetic basis for reduced effectiveness of GLP‑1 receptor agonists, such as Ozempic, in about 10% of the population. The study links specific PAM gene variants to a newly described GLP‑1 resistance,...

Ultragenyx Pharmaceutical: The Market Is Overlooking 2026 Catalysts
Ultragenyx Pharmaceutical (RARE) remains a speculative conviction hold as its commercial base funds a late‑stage rare‑disease pipeline. The company expects multiple 2026‑2027 catalysts—including GTX‑102, DTX401 and UX111—to drive revenue growth. Conservative U.S.–only modeling projects $1.5 B‑$2.4 B peak annual revenue from these...
The Biopharma Senior Associate Whose Career Was Fuelled by FUEL
Luke Sheppard, a senior associate in syringe manufacturing at Amgen, credits the company’s FUEL graduate programme and early internships for propelling his career in Ireland’s thriving biopharma sector. He describes a role that blends operations support, project work, and floor‑level...

The Largest Metabolomics Study Ever Just Pointed To A New Future In Medicine
The UK Biobank has finished the world’s largest metabolomics effort, profiling roughly 250 blood metabolites in half a million volunteers. Conducted by Nightingale Health over several years, the dataset includes repeat samples for 20,000 participants, allowing longitudinal analysis. By layering...
Per- and Polyfluoroalkyl Substances Are Associated with Reduced Cumulus Cell MFN1 Expression and Lower Oocyte Maturation Rates
A cross‑sectional study of 57 IVF patients found per‑ and polyfluoroalkyl substances (PFAS) in all follicular fluid samples, with perfluorooctanesulfonic acid (PFOS) present at measurable levels. PFOS showed a moderate inverse correlation (‑0.45) with cumulus‑cell MFN1 expression, a gene critical...

Hypsizygus Marmoreus Polysaccharides Protect Against Cisplatin-Induced Intestinal Mucositis via Modulation of Gut Microbiota, Inflammation, and Intestinal Barrier Function
Researchers extracted polysaccharides from the edible mushroom Hypsizygus marmoreus (HMP) with a 16% yield and characterized them as high‑molecular‑weight, glucose‑rich polymers. In a mouse model of cisplatin‑induced intestinal mucositis, oral HMP (10 mg kg⁻¹) mitigated weight loss, diarrhea, and immune‑organ atrophy while...

Hackers Meet Their Match: New DNA Encryption Protects Engineered Cells From Within
Researchers published a new DNA‑level encryption system that scrambles engineered cells' genetic code, making it unreadable until a precise chemical sequence restores it. The lock uses a series of nine chemicals arranged as a two‑digit keypad, yielding 45 possible inputs,...
New Drug Combination Doubles Down on Alzheimer's Treatments
University of Waterloo researchers have shown that pairing existing anti‑amyloid antibodies with nutraceutical small molecules such as resveratrol and curcumin dramatically reduces amyloid plaque formation in preclinical models. The combination neutralized protein clumping and allowed a 30 % reduction in antibody...

IDEXX (IDXX) Launches Canine Cancer Detection Panel in UK
IDEXX Laboratories announced the UK launch of its IDEXX Cancer Dx Panel, a blood‑based test for early detection of lymphoma in dogs. The assay delivers results in 3‑5 business days and boasts 79% sensitivity and 99% specificity, including B‑cell and...
Insmed (INSM): Top QQQ Stock to Buy Now
Insmed (NASDAQ: INSM) reported that its Phase 2b CEDAR study of brensocatib in moderate‑to‑severe hidradenitis suppurativa failed to meet both primary and secondary efficacy endpoints. The trial, which enrolled 214 patients across 72 sites, showed the drug underperformed placebo, with a...
Experimental Drug Cuts Parkinson's-Linked Protein up to 60% in Early Trial
Biogen’s experimental antisense oligonucleotide BIIB094 achieved up to a 59 % reduction in LRRK2 protein in a first‑in‑human Parkinson’s trial. The phase 1 study enrolled 82 patients, delivering the drug intrathecally in single‑dose and four‑dose cohorts, and reported only mild to moderate...
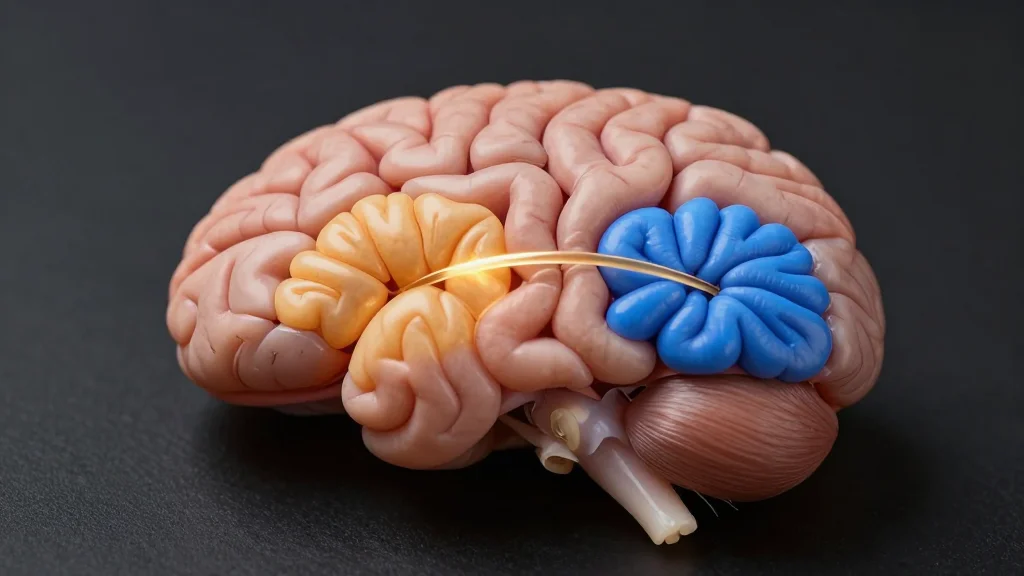
New Biological Marker of Early-Stage Alzheimer's Disease Uncovered
Researchers from Shenzhen MSU‑BIT University and collaborators have identified a disrupted connection between the piriform cortex (PCx) and infralimbic (IL) cortex as an early biological marker of Alzheimer’s disease. Using fMRI in humans and optogenetic, single‑cell RNA‑seq studies in 5xFAD...
Eli Lilly Isn't Replacing Zepbound -- It's Building an Obesity Empire
Eli Lilly is building an obesity empire by adding retatrutide and the oral GLP‑1 pill Foundayo to its Zepbound franchise. Retatrutide delivered a 28.7% mean weight loss in a 68‑week Phase 3 trial, outpacing Zepbound’s 20.2% result. The company is targeting the...