Long-Term Leukemia Trial Reveals MRD-Triggered Treatment May Slow or Prevent Relapse
The RELAZA2 trial has released its long‑term data, confirming that azacitidine administered at the first sign of measurable residual disease (MRD) can significantly delay relapse in myelodysplastic syndromes (MDS) and acute myeloid leukemia (AML). As the world’s first prospective MRD‑triggered study, it shows that early molecular intervention can shift MRD from a prognostic marker to a therapeutic decision point. The multicenter effort, spanning over a decade, demonstrates the feasibility of coordinated, high‑sensitivity monitoring across leading German leukemia centers. Researchers view the results as a launchpad for broader MRD‑guided strategies.
Experimental Pill Promises New Hope for Deadly Pancreatic Cancer
Researchers reported that the experimental pill daraxonrasib, which blocks mutated KRAS proteins, nearly doubled median survival for patients with advanced pancreatic cancer to 13.2 months versus 6.7 months on chemotherapy. The drug showed fewer severe side effects and improved quality...
RASolute 302 Brings a “Transformative" Moment in Pancreatic Cancer: A 60% Improvement in Overall Survival
Revolution Medicines’ RASolute 302 phase‑3 trial showed that the RAS(ON) inhibitor daraxonrasib improved overall survival by 60% in patients with previously treated metastatic pancreatic ductal adenocarcinoma. Median overall survival more than doubled to 13.2 months versus 6.6 months on standard chemotherapy,...
STAT+: For Prostate Cancer Patients Set on Surgery, New Hormone Regimen May Improve Outcomes, Study Finds
A phase‑3 PROTEUS trial found that administering two hormone therapies before and after prostatectomy outperformed a single‑hormone regimen in high‑risk, early‑stage patients. The dual approach reduced biochemical recurrence and improved margin‑negative resection rates. Oncologists view the data as a potential...

NeuExcell Therapeutics Announces Encouraging Data of NXL-004 in Recurrent Malignant Glioma at 2026 ASCO
NeuExcell Therapeutics announced that its first‑in‑human gene therapy NXL‑004 for recurrent malignant glioma earned a Rapid Oral presentation slot at ASCO 2026. The AAV‑NeuroD1 platform was tested in eleven patients, showing a favorable safety profile with no drug‑related serious adverse...
Real-World Predictors of Survival and Response in Advanced Melanoma
Immunotherapy has transformed advanced melanoma care, yet many patients fail first‑line treatment. Researchers applied machine‑learning to a large, real‑world electronic health record database to build interpretable models for overall survival, progression‑free survival, and response. ECOG performance status, PD‑L1‑based first‑line therapy,...
HIV in South Africa: Why Rolling Out a Groundbreaking New Shot Will Miss a Critical Group of Men
The U.S. shipped the first batch of lenacapavir, a long‑acting injectable HIV‑prevention shot, to South Africa in early April 2026, with rollout slated for June. Clinical trials show close to 100% efficacy with just two doses per year. The national...
COXFA4L2 Boosts Cytochrome C Oxidase in Leigh Syndrome
A new Nature Communications study reveals that the mitochondrial protein COXFA4L2 is up‑regulated in cells with COXFA4 mutations, preserving cytochrome c oxidase activity in Leigh‑like encephalopathy. Cryo‑EM shows COXFA4L2 integrates into complex IV, maintaining electron transfer despite the genetic defect. Functional assays...

Predicting Drug Side Effects via LLM Pharmacology
A new study published in Scientific Reports introduces PromptSE, a framework that uses large language models (LLMs) to predict drug side effects from textual pharmacological descriptions. By converting chemical and mechanistic data into prompts, PromptSE outperforms traditional classification models in...
Targeting BCL2: New Hope for Pancreatitis Therapy?
Researchers have identified the anti‑apoptotic protein BCL2 as a therapeutic target for acute pancreatitis, a condition that currently lacks disease‑modifying drugs. Preclinical studies using a selective BCL2 inhibitor demonstrated a marked reduction in pancreatic inflammation and cell death. Building on...

ARACOG Trial Links Enzalutamide to Greater Cognitive Decline Compared With Darolutamide: Alicia Morgans, MD, MPH
The phase‑2 ARACOG trial directly compared the cognitive impact of two androgen‑receptor pathway inhibitors in advanced prostate cancer. At 24 weeks, patients on darolutamide experienced a 15.8% decline on a neuropsychological test versus a 36.1% decline with enzalutamide. The advantage...

Eikon Therapeutics Presents Data on Clinical-Stage Programs at the 2026 Annual Meeting of the American Society of Clinical Oncology
Eikon Therapeutics presented six ASCO 2026 abstracts highlighting late‑stage data for its lead candidates. In the Phase 2 TeLuRide‑005 trial, the TLR7/8 dual agonist EIK1001 combined with pembrolizumab and chemotherapy achieved a 63.1% objective response rate and a 90.8% disease‑control rate...

Global Data for BioNTech and Bristol Myers Squibb’s PD-L1xVEGF-A Bispecific Pumitamig Shows Encouraging Efficacy in Patients with Non-Small Cell Lung...
BioNTech and Bristol Myers Squibb reported interim Phase 2 data from the global ROSETTA Lung‑02 trial of their bispecific PD‑L1×VEGF‑A immunomodulator pumitamig combined with chemotherapy in treatment‑naïve advanced NSCLC. Among 40 evaluable patients, the regimen yielded a confirmed objective response rate...
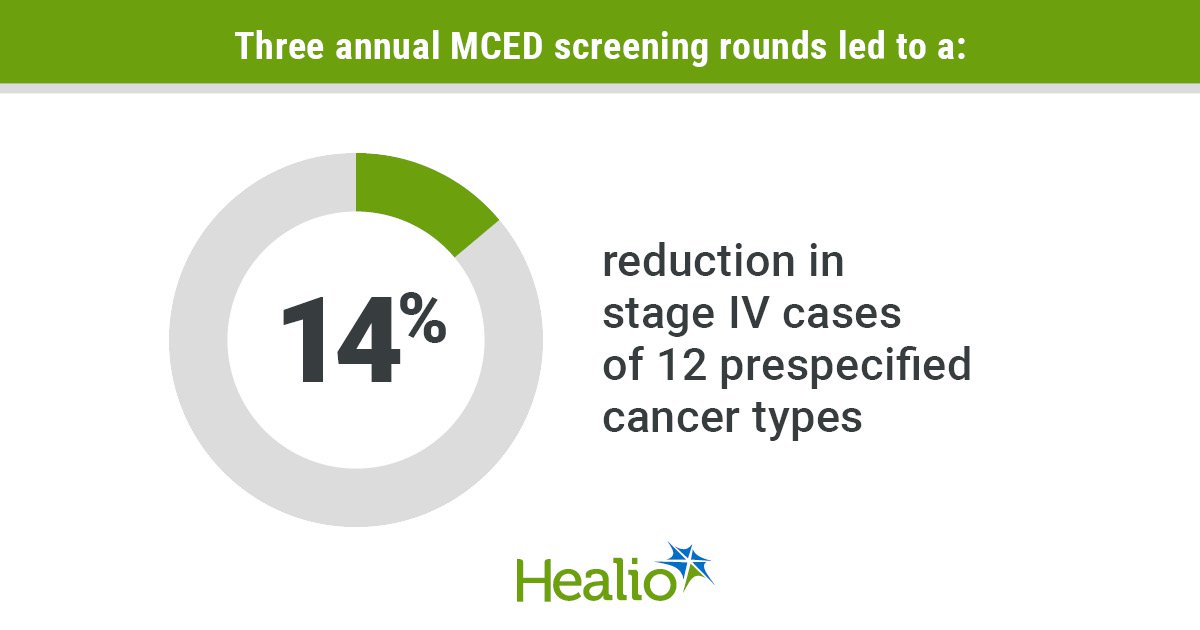
Trial of Early Cancer Detection Test Misses Primary Endpoint
The NHS‑Galleri randomized trial evaluated a multicancer early detection (MCED) blood test in 142,942 adults aged 50‑77. After three annual screens, the study failed to meet its primary endpoint of reducing combined stage III/IV diagnoses for 12 prespecified cancers. However, secondary...
Tezepelumab Helps Severe Asthma Patients Reduce Oral Steroids over 28 Weeks
A Phase III SUNRISE trial published in The Lancet Respiratory Medicine shows tezepelumab enables severe asthma patients to sharply cut their reliance on oral corticosteroids. Over 28 weeks, 69% of participants on the drug achieved at least a 50% dose reduction...

ITM Announces Phase 3 COMPETE Patient-Reported Quality of Life Data with N.c.a. ¹⁷⁷Lu-Edotreotide (ITM-11) Vs. Everolimus at ASCO 2026
ITM Isotope Technologies presented Phase 3 COMPETE patient‑reported quality‑of‑life data at ASCO 2026, showing that non‑carrier‑added ¹⁷⁷Lu‑edotreotide (ITM‑11) outperformed everolimus in gastroenteropancreatic neuroendocrine tumors (GEP‑NETs). In a cohort of 309 patients, the radiopharmaceutical maintained QoL scores (+0.9) while everolimus saw a decline...
Melanoma's Progress, Persistent Gaps, and the Toxicity Criteria That Needed to Change: Igor Puzanov, MD
Immunotherapy has cut U.S. melanoma deaths roughly in half, dropping from about 15,000 to 7,700 annually, according to Roswell Park’s Igor Puzanov. He warns that a subset of tumors undergo epithelial‑mesenchymal transition (EMT), shedding immune‑visible markers and escaping checkpoint inhibitors. To...

Regimen May Become ‘a Standard’ for Aggressive Lymphomas
A phase‑3 frontMIND trial showed that adding tafasitamab and lenalidomide to standard R‑CHOP cuts the risk of progression or death by 25% in newly diagnosed high‑risk diffuse large B‑cell lymphoma (DLBCL) and high‑grade B‑cell lymphoma. The experimental arm achieved 71%...

Scientists Got Mouse Eyes to Perform Photosynthesis — and No, They Didn't Turn Green
Scientists have engineered eye drops containing spinach thylakoid grana that enable mouse eyes to perform light‑driven photosynthetic reactions. The formulation, dubbed LEAF, produces the antioxidant NADPH, reducing inflammation and restoring tear production in a dry‑eye mouse model. After five days,...

STAT+: Revolution Medicines Starts Shipping Experimental Pancreatic Cancer Drug
Revolution Medicines has begun shipping its experimental pancreatic cancer therapy, daraxonrasib, to physicians through an FDA‑authorized early‑access program. The rollout follows Phase 3 data released in mid‑April showing patients lived nearly twice as long as those on standard chemotherapy—the longest survival...

Trial of Multi-Cancer Blood Test Among 142,000 NHS Patients Fails to Meet Main Aim
A randomized controlled trial of Grail's Galleri multi‑cancer blood test involving 142,942 NHS patients failed to meet its primary goal of reducing late‑stage cancer diagnoses. Participants were split evenly between Galleri screening and standard care, with blood drawn annually for...
DNA Framework Nucleator‐Enabled Intelligent Hydrogel Interfaces on Living Cells
Researchers introduced a DNA framework nucleator (DFN) that creates ordered hydrogel interfaces on living cell membranes. The rigid tetrahedral DNA scaffold directs localized branched hybridization chain reactions, delivering an ATP‑responsive hydrogel with ~90.7% efficiency—2.9‑fold higher than flexible dsDNA nucleators. The...

China’s Rise in Drug Development Looms Over U.S.
At this year’s American Society of Clinical Oncology (ASCO) meeting in Chicago, a clinical trial conducted solely in China was featured as one of five headliners, underscoring China’s rapid ascent in biotech. Over the past few years, China’s biotech sector...
Pembrolizumab Combination Therapy Improves Survival in Recurrent Endometrial Cancer Patients
The phase 3 NRG‑GY018 trial presented at ASCO 2026 confirms that adding pembrolizumab to carboplatin‑paclitaxel extends overall survival in advanced or recurrent endometrial cancer. In the mismatch‑repair‑deficient (dMMR) cohort, 48‑month survival rose to 79% versus 60% with chemotherapy alone (HR 0.56). In...

Busy June at FDA with 11 Upcoming PDUFA Dates
The FDA has a packed June agenda with 11 PDUFA decision dates, covering at least eight new drug approvals and three new indication expansions. Among the high‑profile reviews is Ionis Pharmaceuticals’ antisense therapy Tryngolza, currently approved for familial chylomicronemia syndrome....

The CFO Helping to Bring His Biotech “to the Finish Line”
CFO Robert Hoffman joined CytoDyn in May 2025 and quickly tackled its capital shortfall while driving cost discipline. He secured a $30 million standby equity purchase agreement with Yorkville Advisors and closed a $17.5 million private placement, while renegotiating a $57 million convertible...

Teclistamab Extends Remission in Relapsed Myeloma, with 70% Progression-Free at 18 Months
A Phase III MajesTEC‑9 trial of the bispecific antibody teclistamab showed that 70% of relapsed multiple myeloma patients remained progression‑free after 18 months, far outpacing the 27% rate for standard therapies. Nearly two‑thirds of participants achieved complete remission, many reaching MRD‑negative status....
At ASCO, Merck Makes Case for a ‘Cornerstone’ Cancer Drug
Merck is positioning sacituzumab tirumotecan (sac‑TMT), an ADC discovered by China’s Kelun‑Biotech, as a potential cornerstone therapy as its blockbuster Keytruda nears patent expiry. The drug entered a 17‑study Phase 3 program and showed a 65% reduction in disease progression or...

Linvoseltamab Data ‘Encouraging’ in Relapsed AL Amyloidosis
Regeneron's bispecific antibody linvoseltamab showed rapid and deep activity in a phase 1/2 trial for relapsed or refractory systemic AL amyloidosis, with all 20 patients achieving an objective response and 90% attaining a hematologic complete response. The safety profile was manageable,...
Depemokimab Reduces Exacerbations in Type 2 Asthma, CRSwNP Over 2 Years
Depemokimab, GSK's ultra‑long‑acting biologic, achieved a 51% overall reduction in asthma exacerbations and a 69% reduction in patients with chronic rhinosinusitis with nasal polyps (CRSwNP) in the phase 3 SWIFT‑1/2 trials. The drug was administered subcutaneously at 100 mg every 26 weeks, with...
Resmetirom Cuts CV Risk in MASH: Meena Bansal, MD
Resmetirom, a thyroid hormone receptor‑β agonist, showed in secondary analyses of the phase 3 MAESTRO‑NASH and MAESTRO‑NAFLD‑1 trials that it reduces LDL‑C, apolipoprotein B and lipoprotein(a) in patients with metabolic dysfunction‑associated steatohepatitis (MASH) even when they are already receiving statins. The lipid...

Pfizer, Innovent Ink Up-to-$10.5B+ Cancer Treatment Collaboration
Pfizer and China’s Innovent Biologics have signed a global licensing and collaboration deal to co‑develop and co‑commercialize 12 early‑stage antibodies and antibody‑drug conjugates (ADCs) targeting cancer. Pfizer will pay Innovent $650 million upfront and up to $9.85 billion in milestone payments, plus...

‘The Most Significant Change in 20 Years’: Cancer Centers Prepare for Daraxonrasib Demand
U.S. health systems are seeing a surge in demand for daraxonrasib after the FDA issued a May 1 expanded‑access letter. The drug, a KRAS‑G12C inhibitor from Revolution Medicines, showed a 35% response rate and median overall survival of 13.1 months in...

‘Like Christmas’: Woman’s Relief After Test Finds She Can Skip Chemotherapy
A multinational Optima trial involving 4,429 breast‑cancer patients showed that the Prosigna genomic test can reliably identify women who may forgo chemotherapy. The five‑year disease‑free survival was 93.7% for patients who skipped chemo, statistically indistinguishable from the 94.9% rate in...
RNA Therapy for Genetic Heart Failure Moves Closer to Patients After Lab Gains
Researchers at University Medical Center Groningen demonstrated that RNA therapy targeting the PLN R14del mutation reduces protein aggregation and restores cellular function in patient‑derived heart cells. Using induced pluripotent stem cell‑derived cardiomyocytes, the treatment reversed phosphoproteomic abnormalities linked to calcium regulation....

New Approach Methodologies (NAMs)
The FDA announced that it has achieved its Year 1 objectives for New Approach Methodologies (NAMs), dramatically reducing reliance on animal testing in drug development. By focusing first on monoclonal antibodies, the agency outlined a step‑wise roadmap that validates AI‑driven models,...

Putin’s $26 Billion Longevity Push
Russian President Vladimir Putin has earmarked $26 billion for a national longevity program that targets organ bioprinting and xenotransplantation in genetically engineered mini‑pigs. State scientists claim early successes, such as printed cartilage and a mouse thyroid, with a goal of human organ...

Illumina Announces MRD Kit Ahead of ASCO Meeting
Illumina announced a molecular residual disease (MRD) kit ahead of the ASCO meeting, offering solid‑tumor and blood‑cancer genomic profiling on NovaSeq systems. The kit delivers whole‑genome sequencing sensitivity down to 10 ppm and 99.5% analytical specificity, with an end‑to‑end workflow completed...

ASCO: BMS Hails "Compelling" Phase 3 Celmod Readout
Bristol Myers Squibb presented phase 3 data for mezigdomide, a cereblon E3 ligase modulator, combined with carfilzomib and dexamethasone in relapsed/refractory multiple myeloma. The SUCCESSOR‑2 trial showed a 52% reduction in progression or death risk and a median progression‑free survival of...

Cracked Gaskets and Rust-Like Residues Trigger FDA Warning Letter to Alchymars
The U.S. Food and Drug Administration issued a second warning letter to Indian API maker Alchymars ICM SM Private Limited after an unannounced inspection uncovered cracked gaskets, rust‑like residues and wet paint on equipment used for U.S. drug production. The...

STAT+: Pharmalittle: We’re Reading About Replimune Drug Getting Third Try at FDA Approval, a Pfizer Deal in China, and More
An FDA advisory panel recommended that this fall’s COVID‑19 vaccines be updated to target the fast‑growing XFG (“stratus”) variant. Pfizer signed a global licensing and collaboration deal with China’s Innovent Biologics worth up to $10.5 billion to co‑develop 12 early‑stage cancer...

Replimune Gives Cancer Immunotherapy a Third Try After FDA Leadership Shakeup
Replimune is filing a third biologics license application for its oncolytic melanoma therapy RP1, now paired with Bristol Myers Squibb’s PD‑1 inhibitor Opdivo. The FDA has labeled the resubmission an urgent matter and will prioritize its review. Earlier submissions were rejected...

Can AI Crack Pharma’s Drug Repurposing Goldmine?
The FDA has opened a public comment period, ending June 11, to gather ideas on how to accelerate drug repurposing using existing therapies. It aims to identify candidates that already meet evidence standards or show early efficacy, reducing the need for...

Replimune to Resubmit Twice-Rejected Drug for Approval After FDA Shakeup
Replimune announced it will resubmit its melanoma drug to the FDA after two prior rejections under former Commissioner Marty Makary. The agency’s recent leadership turnover has led to an agreement on a path forward, with the FDA promising an urgent...
Targeted Therapy Shows Superior Results Over Chemotherapy in Treating Difficult Lung Cancer, ASCO Reports
At the American Society of Clinical Oncology (ASCO) meeting, researchers presented data showing a novel targeted therapy outperformed standard chemotherapy in patients with advanced non‑small cell lung cancer (NSCLC) that are difficult to treat. The trial reported a median progression‑free...

Can WEE1 Inhibitors Finally Make Replication Stress Druggable?
Targeting replication stress through WEE1 inhibition is re‑emerging after early setbacks. First‑generation inhibitor adavosertib demonstrated tumor responses in ovarian and uterine cancers but was limited by dose‑limiting toxicity. Newer agents such as APR‑1051, azenosertib and zedoresertib are being tested in...
New Study Uncovers How Bacteria Seize a Rare Sugar Molecule
A multidisciplinary team has identified a previously unknown bacterial transporter that snatches a rare sugar molecule, N‑acetyl‑neuraminic‑acid analog, from host environments. Cryo‑EM imaging revealed the transporter’s architecture at 2.8 Å, pinpointing the binding pocket and key amino‑acid residues. Functional assays showed...

Merck Secures the US FDA Breakthrough Therapy Designation for Calderasib to Treat Newly Diagnosed Metastatic KRAS G12C-Mutant NSCLC
The U.S. FDA granted Merck’s KRAS‑G12C inhibitor calderasib (MK‑1084) breakthrough therapy designation for first‑line treatment of newly diagnosed metastatic non‑small cell lung cancer (NSCLC) that harbors a KRAS G12C mutation and expresses PD‑L1 (TPS ≥ 1%). The designation is backed by Phase I...

Tomato Gene Discovery Opens Door to Heat-Tolerant Varieties
Researchers at Japan's University of Tsukuba identified the SlIAA9 gene as a key brake on heat tolerance during tomato seed germination. Loss‑of‑function mutants lacking SlIAA9 germinated at near‑normal rates even under prolonged high‑temperature exposure and produced vigorous seedlings. Molecular analysis...
GSK’s Nucala Secures NICE COPD Endorsement
NICE has added GSK’s Nucala (mepolizumab) to its list of approved biologic add‑on treatments for chronic obstructive pulmonary disease (COPD) in patients with elevated eosinophils. The endorsement is based on three Phase III trials that demonstrated up to a 21 % reduction...